GASES Myth Busters Fun With Gas Video Discovery

GASES! Myth. Busters: Fun With Gas : Video : Discovery Channel They’re all around you….

Observations of Gas n n n Gases have mass. Compare the mass of an empty balloon to the mass of one that’s filled with air. Gases expand to fill their containers. As the pressure on a gas increases its volume decreases. Gases are compressible. Different gases move through each other easily. The movement of one substance through another is called diffusion. Gases exert pressure. As temp increases, the pressure exerted by the gas increases.

Kinetic Molecular Theory n n n Gases consist of small particles, molecules or atoms that have mass. Gas particles are separated by large distances compared to their size. In this theory the volume of gas particles is considered 0. Gas particles are in constant motion. Gas particles do not exert attractive forces on each other. When gas particles collide energy may be transferred but total energy remains. n n n The observation that gases have mass. The compressibility of gases. Also explains the low density of gases. The ability of gases to expand rapidly. The fact that gases exert pressure. The observation that gases expand to fill their containers. The constant pressure of a gas sample at a given temperature.

KMT Gases that follow the assumptions outlined in the KMT are called IDEAL gases.

Real Gases In reality real gases deviate from ideal behavior because: 1) Real gas particles have small but significant volume. 2) Real gases exert some attractive force on each other. The two gases that come closest to ideal gas behavior due to their small size and attractive forces are H and He. n

KMT v. Real Gases KMT useful for predicting gas behavior. Real gases are most likely to behave ideally when: 1) Pressure is low because the particles are far apart. 2) Temperature is high because the kinetic energy of the particles overcomes their attractive forces. Conversely, gases deviate most from ideal behavior at high pressure and low temperature. n
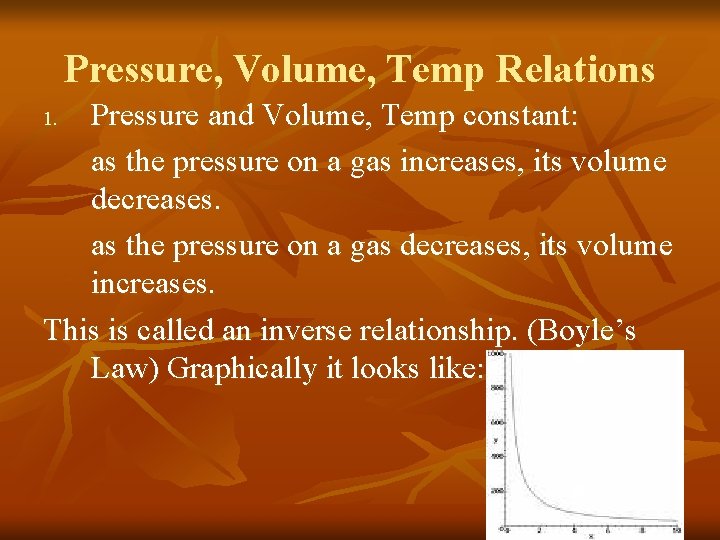
Pressure, Volume, Temp Relations Pressure and Volume, Temp constant: as the pressure on a gas increases, its volume decreases. as the pressure on a gas decreases, its volume increases. This is called an inverse relationship. (Boyle’s Law) Graphically it looks like: 1.

Pressure and Volume con. n n n When we decrease the volume of a sample of gas, the number of particles colliding with a given area increases. This results in an increase in pressure. AKA – Gas molecules collide with other gas molecules and walls which exerts a pressure/force on the walls. VTP (like VIP)
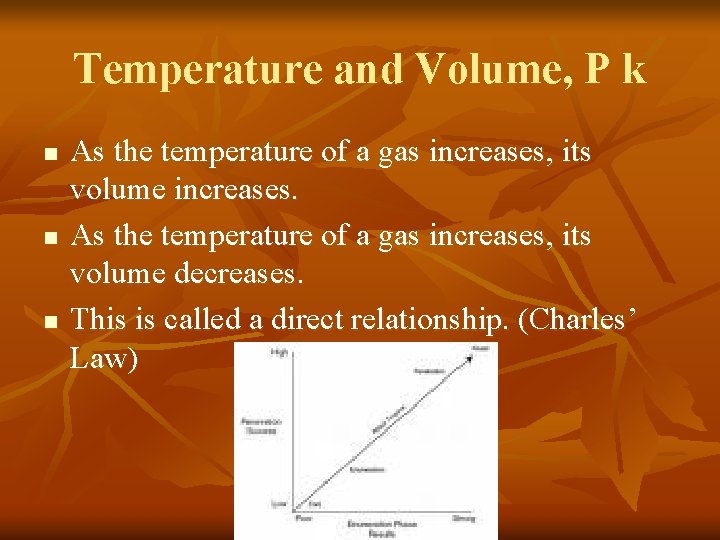
Temperature and Volume, P k n n n As the temperature of a gas increases, its volume increases. As the temperature of a gas increases, its volume decreases. This is called a direct relationship. (Charles’ Law)

Temperature and pressure, V k n n As the temp of a gas increases, its pressure increases. As the temp of a gas decreases, its pressure decreases. This is called a direct relationship. As temp increases the speed/velocity of the particles and the frequency of the collisions both increase. This results in an increase in pressure.

Combined Gas Law The Combined Gas Law can be used to predict the properties of gases that behave ideally. This occurs at conditions of increased temp and decreased pressure. n The combined gas law is given in Table T as P 1 V 1 = P 2 V 2 T 1 T 2 Note: Temp must be in K. n

Avogadro's Principle n n Equal volumes of gases at the same T and P contain equal #’s of particles. This allows use to determine how many moles of a gas we have based on volume.
- Slides: 12