Gases I Physical Properties A Kinetic Molecular Theory





















- Slides: 26

Gases I. Physical Properties

A. Kinetic Molecular Theory b Particles in an ideal gas… • have no volume. • have elastic collisions. • are in constant, random, straightline motion. • don’t attract or repel each other.

B. Characteristics of Gases b Gases expand to fill any container. • random motion, no attraction b Gases are fluids (like liquids). • no attraction b Gases have very low densities. • no volume = lots of empty space

C. Temperature b Always use absolute temperature (Kelvin) when working with gases. ºF -459 ºC -273 K 0 32 212 0 100 273 373 K = ºC + 273

Converting Celsius to Kelvin • Gas law problems involving temperature will always require that the temperature be in Kelvin. (Remember that no degree sign is shown with the kelvin scale. ) • Reason? There will never be a zero volume, since we have never reached absolute zero. Kelvin = C + 273 °C = Kelvin - 273

Direct relationship b Direct : As one variable increases, the other increases by the same –Ex: double one variable, double the other – Quadruple one variable, quadruple the other

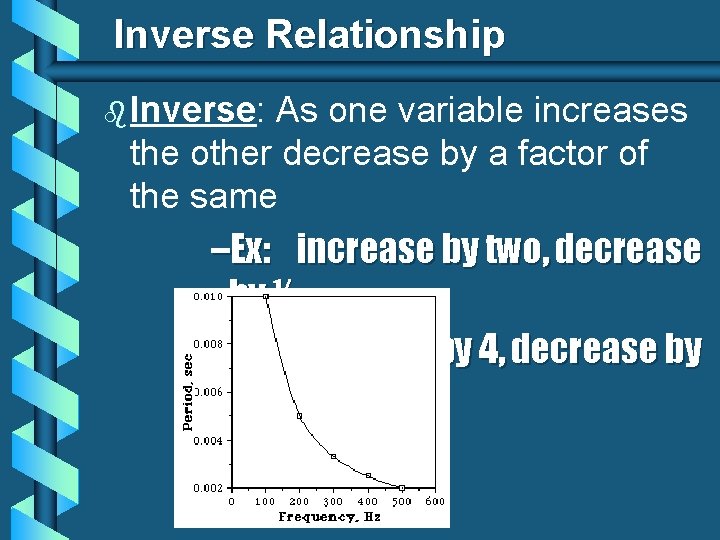
Inverse Relationship b Inverse: As one variable increases the other decrease by a factor of the same –Ex: increase by two, decrease by ½ – increase by 4, decrease by ¼

Gas Laws II. The Gas Laws a. BOYLES b. CHARLES c. GAY-LUSSAC

Robert Boyle (1627 -1691) • Boyle was born into an aristocratic Irish family • Became interested in medicine and the new science of Galileo and studied chemistry. • Wrote extensively on science, philosophy, and theology.

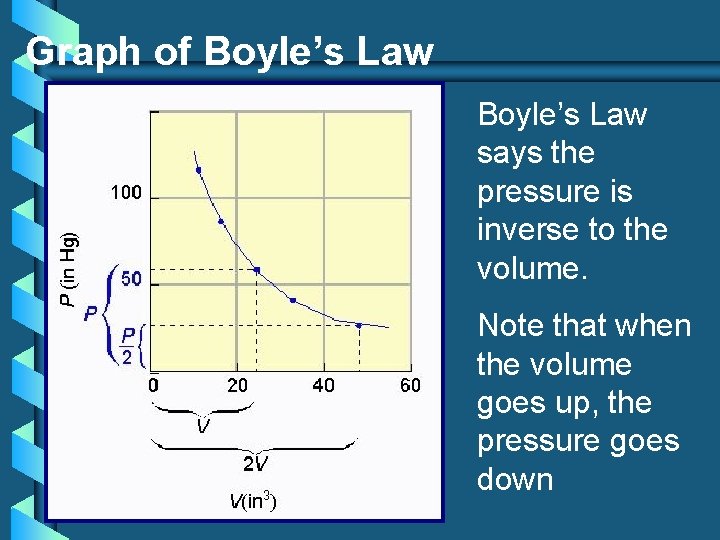
#1. Boyle’s Law - 1662 Gas pressure is inversely proportional to the volume, when temperature is held constant. Pressure x Volume = a constant Equation: P 1 V 1 = P 2 V 2 (T = constant)

Graph of Boyle’s Law says the pressure is inverse to the volume. Note that when the volume goes up, the pressure goes down

Boyle’s Law A bicycle pump is a good example of Boyle’s law. As the volume of the air trapped in the pump is reduced, its pressure goes up, and air is forced into the tire.

Jacques Charles (1746 -1823) • • French Physicist Part of a scientific balloon flight on Dec. 1, 1783 – was one of three passengers in the second balloon ascension that carried humans This is how his interest in gases started It was a hydrogen filled balloon – good thing they were careful!

#2. Charles’s Law - 1787 The volume of a fixed mass of gas is directly proportional to the Kelvin temperature, when pressure is held constant. This extrapolates to zero volume at a temperature of zero Kelvin.

B. Charles’ Law b The volume and absolute temperature (K) of a gas are directly related • at constant mass & pressure V T

Joseph Louis Gay-Lussac (1778 – 1850) French chemist and physicist v Known for his studies on the physical properties of gases. v In 1804 he made balloon ascensions to study magnetic forces and to observe the composition and temperature of the air at different altitudes. v

#3. Gay-Lussac’s Law - 1802 • The pressure and Kelvin temperature o a gas are directly proportional, provided that the volume remains constant.

C. Gay-Lussac’s Law b The pressure and absolute temperature (K) of a gas are directly related • at constant mass & volume P T

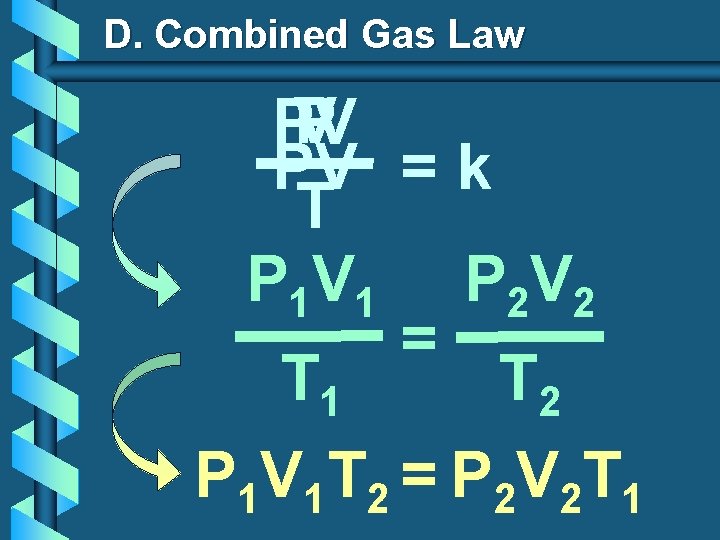
Combined Gas Law Why didn’t you just show us this in the first place? P 1 V 1 T 1 = P 2 V 2 T 2

D. Combined Gas Law P V PV PV = k T P 1 V 1 P 2 V 2 = T 1 T 2 P 1 V 1 T 2 = P 2 V 2 T 1

Try This one… b 500. 0 liters of a gas are prepared at 700. 0 mm Hg and 200. 0 °C. The gas is placed into a tank under high pressure. When the tank cools to 20. 0 °C, the pressure of the gas is 30. 0 atm. What is the volume of the gas?

Here ya go… P 1 V 1 T 1 = P 2 V 2 T 2 700 mm. Hg · 500 L = 30 mm. Hg · (x? ) 200˚C 20 ˚C Volume =

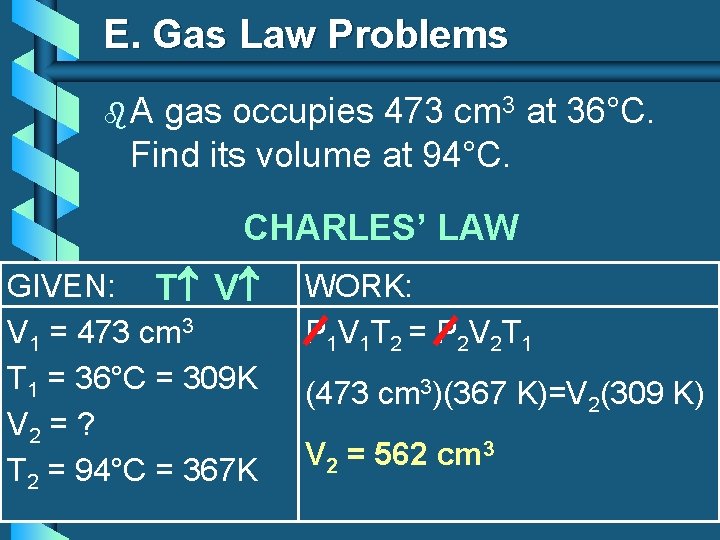
E. Gas Law Problems b. A gas occupies 473 cm 3 at 36°C. Find its volume at 94°C. CHARLES’ LAW GIVEN: T V V 1 = 473 cm 3 T 1 = 36°C = 309 K V 2 = ? T 2 = 94°C = 367 K WORK: P 1 V 1 T 2 = P 2 V 2 T 1 (473 cm 3)(367 K)=V 2(309 K) V 2 = 562 cm 3

E. Gas Law Problems b. A gas occupies 100. m. L at 150. k. Pa. Find its volume at 200. k. Pa. BOYLE’S LAW GIVEN: P V V 1 = 100. m. L P 1 = 150. k. Pa V 2 = ? P 2 = 200. k. Pa WORK: P 1 V 1 T 2 = P 2 V 2 T 1 (150. k. Pa)(100. m. L)=(200. k. Pa)V 2 = 75. 0 m. L

E. Gas Law Problems b. A gas occupies 7. 84 cm 3 at 71. 8 k. Pa & 25°C. Find its volume at STP. COMBINED GAS LAW GIVEN: P T V WORK: V 1 = 7. 84 cm 3 P 1 V 1 T 2 = P 2 V 2 T 1 P 1 = 71. 8 k. Pa (71. 8 k. Pa)(7. 84 cm 3)(273 K) T 1 = 25°C = 298 K =(101. 325 k. Pa) V 2 (298 K) V 2 = ? P 2 = 101. 325 k. Pa V 2 = 5. 09 cm 3 T 2 = 273 K

E. Gas Law Problems b. A gas’ pressure is 765 torr at 23°C. At what temperature will the pressure be 560. torr? GAY-LUSSAC’S LAW GIVEN: P T WORK: P 1 = 765 torr P 1 V 1 T 2 = P 2 V 2 T 1 = 23°C = 296 K (765 torr)T 2 = (560. torr)(309 K) P 2 = 560. torr T 2 = 226 K = -47°C T 2 = ?