Gases Chapter 5 Email benzene 4 presidentgmail com

Gases Chapter 5 E-mail: benzene 4 president@gmail. com Web-site: http: //clas. sa. ucsb. edu/staff/terri/

Gases – Ch. 5 1. Determine if the following are directly or inversely proportional – assume all other variables are constant a. Pressure and volume b. Pressure and temperature

Gases – Ch. 5 2. The valve between two tanks is opened. See below. Calculate the ratio of partial pressures (O 2: Ne) in the container after the valve is opened. a. b. c. d. e. 1. 31 1. 60 2. 10 0. 477 0. 615 3. 25 -L 8. 64 atm O 2 2. 48 -L 5. 40 atm Ne

Gases – Ch. 5 o

Gases – Ch. 5 3. In an experiment 300 m 3 of methane is collected over water at 785 torr and 65 °C. What is the volume of the dry gas (in m 3) at STP? The vapor pressure of water at 65 °C is 188 torr.

Gases – Ch. 5 4. A gaseous mixture containing 1. 5 mol Ar, 6 mol He and 3. 5 mol Ne has a total pressure of 7. 0 atm. What is the partial pressure of Ne? a. b. c. d. e. 1. 4 atm 2. 2 atm 3. 8 atm 4. 6 atm 2. 7 atm

Gases – Ch. 5 5. A mixture of oxygen and helium is 92. 3% by mass oxygen. What is the partial pressure of oxygen if the total pressure is 745 Torr?

Gases – Ch. 5 6. A sample of oxygen gas has a volume of 4. 50 L at 27°C and 800. 0 torr. How many oxygen molecules are in the sample? a. b. c. d. e. 1. 16 × 1023 5. 8 × 1022 2. 32 × 1024 1. 16 × 1022 none of these

Gases – Ch. 5 Ideal gas law Considering one set of variables for a gas under “ideal” conditions PV = n. RT R ⇒ Universal gas constant R = 0. 08206 atm. L/mol. K R = 62. 37 torr. L/mol. K R = 8. 314 KPa. L/mol. K or J/mol. K

Gases – Ch. 5 7. Consider the combustion of liquid hexane: 2 C 6 H 14 (l) + 19 O 2 (g) → 12 CO 2 (g) + 14 H 2 O (l) 1. 52 -g of hexane is combined with 2. 95 L of oxygen at 312 K and 890 torr. The carbon dioxide gas is collected and isolated at 297 K and 0. 930 atm. What volume of carbon dioxide gas will be collected, assuming 100% yield? a. b. c. d. e. 0. 504 L 1. 93 L 2. 23 L 0. 607 L 4. 04 L

Gases – Ch. 5 8. A 3. 54 -g sample of lead(II) nitrate (molar mass = 331 g/mol) is heated in an evacuated cylinder with a volume of 1. 60 L. The salt decomposes when heated, according to the following equation: 2 Pb(NO 3)2 (s) 2 Pb. O (s) + 4 NO 2 (g) + O 2 (g) Assuming complete decomposition, what is the pressure (in atm) in the cylinder after decomposition and cooling to a temperature of 300. K? Assume the Pb. O(s) takes up negligible volume.

Gases – Ch. 5 9. 2. 5 mol of O 2 gas and 3. 0 mol of solid carbon, C (s) are put into a 3. 50 -liter container at 23°C. If the carbon and oxygen react completely to form CO (g), what will be the final pressure (in atm) in the container at 23°C?

Gases – Ch. 5 10. The density of an unknown gas at STP is 0. 715 g/L. Identify the gas. a. NO b. Ne c. CH 4 d. O 2

Gases – Ch. 5 o

Gases – Ch. 5 11. Air is 79% N 2 and 21% O 2 by volume. Calculate the density of air at 1. 0 atm, 25°C. a. b. c. d. e. 0. 590 g/L 1. 18 g/L 2. 46 g/L 14. 1 g/L none of these
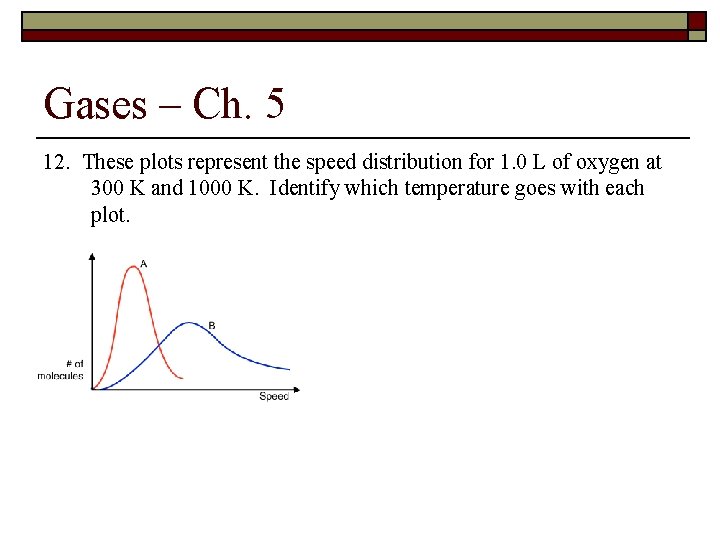
Gases – Ch. 5 12. These plots represent the speed distribution for 1. 0 L of oxygen at 300 K and 1000 K. Identify which temperature goes with each plot.

Gases – Ch. 5 Average Speed
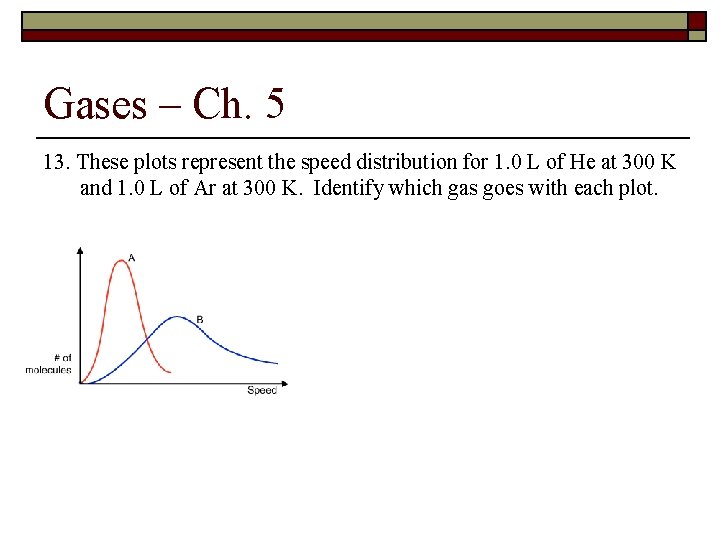
Gases – Ch. 5 13. These plots represent the speed distribution for 1. 0 L of He at 300 K and 1. 0 L of Ar at 300 K. Identify which gas goes with each plot.

Gases – Ch. 5 14. Calculate the temperature at which the average velocity of Ar (g) equals the average velocity of Ne (g) at 25°C. a. b. c. d. e. 317°C 151°C 49. 5°C 25°C none of these

Gases – Ch. 5 15. It takes 12 seconds for 8 m. L of hydrogen gas to effuse through a porous barrier at STP. How long will it take for the same volume of carbon dioxide to effuse at STP?

Gases – Ch. 5 Graham’s Law If two or more gases are effusing under the same conditions

Gases – Ch. 5 16. The diffusion rate of H 2 gas is 6. 45 times faster than that of a certain noble gas (both gases are at the same temperature). What is the noble gas? a. b. c. d. e. Ne He Ar Kr Xe

Gases – Ch. 5 17. Consider two 5 L flasks filled with different gases. Flask A has carbon monoxide at 250 torr and 0 °C while flask B has nitrogen at 500 torr and 0 °C. a. Which flask has the molecules with the greatest average kinetic energy? b. Which flask has the greatest collisions per second?

Gases – Ch. 5 Useful equations KEavg = 3/2 RT KE = 1/2 mu 2

Gases – Ch. 5 18. Under what conditions will a gas behave the most like an ideal gas?
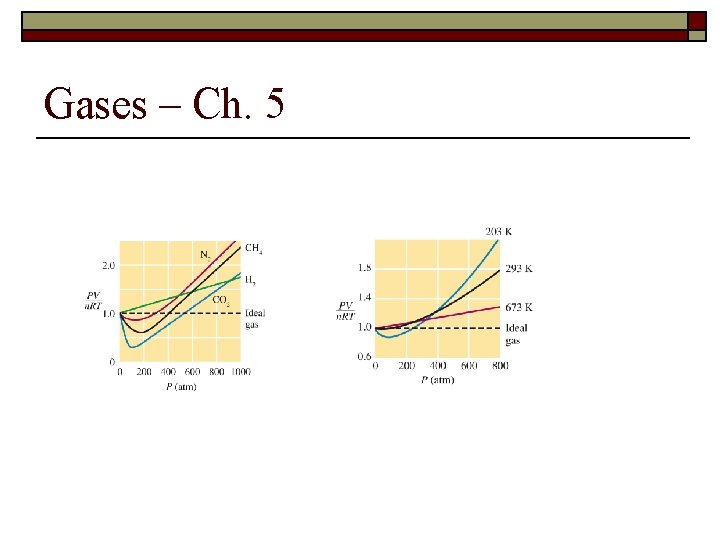
Gases – Ch. 5

Gases – Ch. 5 19. Which of the following gases will have the lowest molar volume at STP? a. He b. CH 2 Cl 2 c. CO 2

Gases – Ch. 5 o

Gases – Ch. 5
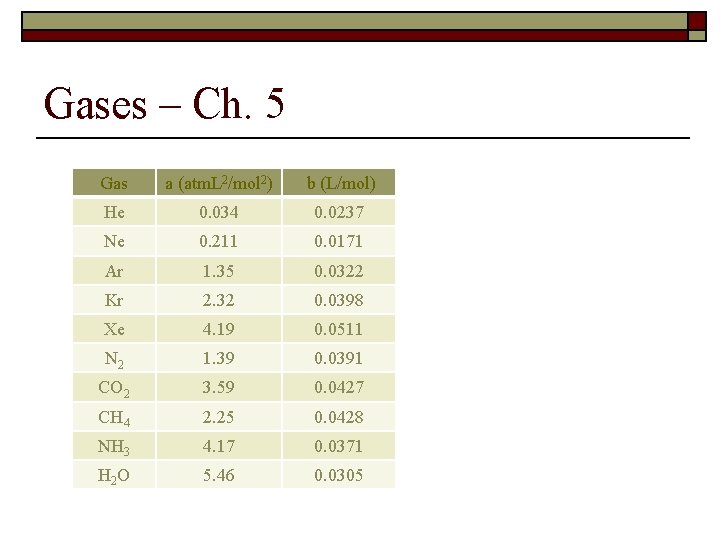
Gases – Ch. 5 Gas a (atm. L 2/mol 2) b (L/mol) He 0. 034 0. 0237 Ne 0. 211 0. 0171 Ar 1. 35 0. 0322 Kr 2. 32 0. 0398 Xe 4. 19 0. 0511 N 2 1. 39 0. 0391 CO 2 3. 59 0. 0427 CH 4 2. 25 0. 0428 NH 3 4. 17 0. 0371 H 2 O 5. 46 0. 0305
Gases – Ch. 5 You have completed ch. 5

Answer key – Ch. 5 1. Determine if the following are directly or inversely proportional – assume all other variables are constant a. Pressure and volume inversely b. Pressure and temperature directly PV = n. RT If you multiply variables ⇒ inversely proportional If you divide variables ⇒ directly proportional

Answer key – Ch. 5 2. The valve between two tanks is opened. See below. Calculate the ratio of partial pressures (O 2: Ne) in the container after the valve is opened. a. 1. 31 b. 1. 60 c. 2. 10 d. 0. 477 e. 0. 615

Answer key – Ch. 5 o

Answer key – Ch. 5 4. A gaseous mixture containing 1. 5 mol Ar, 6 mol He and 3. 5 mol Ne has a total pressure of 7. 0 atm. What is the partial pressure of Ne? a. 0. 95 atm b. 2. 2 atm c. 3. 8 atm d. 2. 7 atm e. 4. 8 atm

Answer key – Ch. 5 o 4. 80 total moles

Answer key – Ch. 5 6. A sample of oxygen gas has a volume of 4. 50 L at 27°C and 800. 0 torr. How many oxygen molecules does it contain? a. 1. 16 × 1023 b. 5. 8 × 1022 c. 2. 32 × 1024 d. 1. 16 × 1022 e. none of these

Answer key – Ch. 5 7. Consider the combustion of liquid hexane: 2 C 6 H 14 (l) + 19 O 2 (g) → 12 CO 2 (g) + 14 H 2 O (l) 1. 52 -g of hexane is combined with 2. 95 L of oxygen at 312 K and 890 torr. The carbon dioxide gas is collected and isolated at 297 K and 0. 930 atm. What volume of carbon dioxide gas will be collected, assuming 100% yield? a. 0. 504 L b. 1. 93 L c. 2. 23 L d. 0. 607 L e. 4. 04 L

Answer key – Ch. 5 8. A 3. 54 -g sample of lead(II) nitrate (molar mass = 331 g/mol) is heated in an evacuated cylinder with a volume of 1. 60 L. The salt decomposes when heated, according to the following equation: 2 Pb(NO 3)2 (s) 2 Pb. O (s) + 4 NO 2 (g) + O 2 (g) Assuming complete decomposition, what is the pressure (in atm) in the cylinder after decomposition and cooling to a temperature of 300. K? Assume the Pb. O(s) takes up negligible volume.

Answer key – Ch. 5 9. 2. 5 mol of O 2 gas and 3. 0 mol of solid carbon, C (s) are put into a 3. 50 -liter container at 23°C. If the carbon and oxygen react completely to form CO (g), what will be the final pressure (in atm) in the container at 23°C? 2 C (s) + O 2 (g) 2 CO (g) Determine limiting reagent C ⇒ 3 mol C/2 = 1. 5 O 2 ⇒ 2. 5 mol/1 = 2. 5 ⇒ C is the LR Since C is the LR ⇒ in addition to the CO formed there will be excess O 2 in the container so the pressure will be the total pressure ⇒ Ptotal = ntotal. RT/V …continue to next slide

Answer key – Ch. 5 9. …continued 2 C (s) O 2 (g) → 2 CO (g) 3 mole 2. 5 mole 0 mole 1 mole 3 mole -3 0

Answer key – Ch. 5 10. The density of an unknown gas at STP is 0. 715 g/L. Identify the gas. a. NO b. Ne c. CH 4 d. O 2

Answer key – Ch. 5 11. Air is 79% N 2 and 21% O 2 by volume. Calculate the density of air at 1. 0 atm, 25°C. a. 0. 590 g/L b. 1. 18 g/L c. 2. 46 g/L d. 14. 1 g/L e. none of these D = 1. 18 g/L

Answer key – Ch. 5 12. These plots represent the speed distribution for 1. 0 L of oxygen at 300 K and 1000 K. Identify which temperature goes with each plot. Average Speed of A Average Speed of B According to the average speed equation ⇒ uavg = (8 RT/π M)1/2 we can see the relationship between average speed and temperature as T ↑ uavg ↑ since uavg B > uavg A ⇒ TB>TA Plot A ⇒ 300 K Plot B ⇒ 1000 K

Answer key – Ch. 5 13. These plots represent the speed distribution for 1. 0 L of He at 300 K and 1. 0 L of Ar at 300 K. Identify which gas goes with each plot. Average Speed of A Average Speed of B

Answer key – Ch. 5 14. Calculate the temperature at which the average velocity of Ar (g) equals the average velocity of Ne (g) at 25°C. a. 317°C b. 151°C c. 49. 5°C d. 25°C e. none of these

Answer key – Ch. 5 o

Answer key – Ch. 5 16. The diffusion rate of H 2 gas is 6. 45 times as great as that of a certain noble gas (both gases are at the same temperature). What is the noble gas? a. Ne b. He c. Ar d. Kr e. Xe

Answer key – Ch. 5 o

Answer key – Ch. 5 18. Under what conditions will a real gas behave like an ideal gas? An “ideal” gas is one that in reality adheres to the ideal gas law ⇒ meaning experimental values agree with calculated values using PV = n. RT Gases are more likely to behave “idealy” when the pressure is low and/or the temperature is high Deviations from the ideal gas law is due to the attractive forces between the gas particles and the volume of the gas particles relative to the volume of the container

Answer key – Ch. 5 19. Which of the following gases will have the lowest molar volume at STP? a. He b. CH 2 Cl 2 The molar volume of an “ideal” gas is 22. 4 L/mol c. CO 2 as the attractive forces of the gas particles ↑ the molar volume ↓ – later (in ch 16) we will learn the specifics of attractive forces however for now we can use the relationship that as molar mass ↑ attractive forces ↑ (an exception is water – although water is rather on the light side it has quite strong attractive forces called H-Bonds which we’ll see further in ch 16) Therefore since CH 2 Cl 2 has the highest molar mass it has the strongest attractive forces and the lowest molar volume
- Slides: 51