Gases Chapter 13 Some basics Gases have properties

Gases Chapter 13

Some basics Gases have properties that are very different from solids and liquids. l They are very sensitive to changes in pressure and temperature and have low density. l Gases do have mass( although small). l Think of properties of gases when your ears pop while driving to Tahoe , deep-sea divers or your kid sister’s birthday balloons. l

Pressure and Temperature l l l Pressure of gases is usually denoted in atm or mm. Hg. 1 atm =760 mm. Hg 1 atm =760 torr Other units include Pascal and psi. Measured by a barometer l l Temperature of gases is measured in Kelvin = °C +273 Other unit for temperature is Fahrenheit. Measured by a thermometer

Kinetic Molecular Theory l l Volume of the molecules is negligible (zero) compared to the volume of the container. The molecules are colliding with the walls of the container and this causes the pressure of the gas. The molecules do not attract or repel each other. The average kinetic energy of the molecules is directly proportional to the Kelvin temperature.

Boyle’s Law l l When pressure of a gas is increased the volume of a gas decreases provided the temperature remains the same. PV = k (constant temperature) Pressure and volume are inversely proportional l Or as P goes up, V must go down to keep the above equation correct. l P 1 V 1 =P 2 V 2 is a relationship that helps us solve problems when conditions are changed for a gas.

Sample problem l What is the new pressure if 0. 500 L of oxygen at pressure 0. 87 atm is changed to 0. 750 L at constant temperature? Solution l Since this is at constant temperature, Boyle’s law applies, P 1 V 1 =P 2 V 2 P 1= 0. 87 atm, V 1 = 0. 500 L, P 2 = ? , V 2 = 0. 750 L l P 2 = P 1 V 1 = 0. 87 atm X 0. 500 L = 0. 58 atm V 2 0. 750 L

Practice problems l The volume of a sample of hydrogen is 250. m. L at 3. 5 atm pressure. What will be the volume when the pressure is reduced to 0. 75 atm, assuming that temperature remains constant? 1200 m. L or 1. 2 L l The pressure of a 2. 34 L sample of helium is 785 torr. Calculate the pressure in atm if volume is decreased to 2. 04 L and temperature kept constant. 1. 18 atm

Charles’s Law l l According to Charles’s Law, the volume of a gas increases when temperature increases, provided the pressure is kept constant. V = k or when temperature increases, volume must T increase as well V 1 = V 2 all temperatures have to be in KELVIN scale!!!! T 1 T 2 This law can now be used to solve problems

Sample problem The volume of a gas at 25ºC is 234 m. L. What will its volume be at 50ºC if pressure is kept constant? l Solution 25ºC + 273 = 298 K = T 1 , V 1 = 234 m. L 50ºC + 273 = 323 K = T 2 , V 2 = ? l V 1 = V 2 , V 2 = V 1 X T 2 = 234 m. L X 323 K = 254 m. L T 1 T 2 T 1 298 K

Practice problems A gas occupies 670 m. L at 45ºC. At what temperature (in ºC) will it occupy 750 m. L if pressure is kept unchanged? l 83ºC l If methane gas occupies 58. 0 L at 17ºC, what volume will it occupy at 27ºC if pressure is left the same? l 60. L l

Avogadro’s Law l l When the number of moles of gas increases, the volume increases too (duh!!) V = k (at constant temperature and n pressure) V 1= V 2 n 1 n 2 This above equation can be used to solve problems.

Problems If 6 moles of oxygen occupies 23 L how many L will 3. 67 moles occupy at constant temperature and pressure? l V 1 = V 2 n 1 n 2 V 2 = V 1 X n 2 = 23 L X 3. 67 moles = 14 L n 1 6 moles l

Ideal Gas An ideal gas is one whose molecules are not attracted to each other and the volume occupied by each molecule is too small to matter. l Most gases behave ideally at high temperatures and low pressures. l Gases are not ideal when compressed or cooled down. l

Ideal Gas Law If we combine all the three gas laws (Boyle’s, Charles’s and Avogadro's) we get the ideal gas law: l PV =n. RT where R =0. 08206 L. atm K. mol If any three of the properties are known the fourth can be calculated. l This equation has some limits, can be used only in low pressures and high temperatures. If the pressure goes up or temperature decreases, corrections need to be applied.

Problems What volume is occupied by 0. 250 mol of CO 2 at 25°C and 371 torr? l PV =n. RT P = 371/760 = 0. 488 atm T = 298 K R =0. 08206 L. atm K. mol V= n. RT =. 250 mol X 298 K X. 08206 L. atm P 0. 488 atm K. mol = 12. 5 L l

Practice A 1. 5 mol of radon gas has a volume of 21. 0 L at 33°C. What is the pressure of the gas? l 1. 8 atm l What is the mass of oxygen needed to fill a tank of volume 22. 7 L at temperature 34°C and pressure 1. 5 atm? l 43. 3 g l

Combined Gas Law We can use the combined gas law if the conditions of pressure, volume or temperature for the same amount of gas are altered. l P 1 V 1 = P 2 V 2 T 1 T 2 Temperature must be in Kelvin !!!! l

Sample problem A sample of neon gas has a volume of 27. 5 m. L at 22. 0°C and 740. torr pressure. What will its volume be at temperature 15. 0°C and pressure 755 torr? l P 1 V 1 = P 2 V 2 T 1 T 2 l 740. torr X 27. 5 m. L = 755 torr X V 2 295 K 288 K V 2=26. 3 m. L l

Dalton’s Law of partial pressures According to Dalton’s law the pressure of a mixture of gases is related to the number of moles of the gas in the mixture. l Ptotal = p 1 +p 2 + p 3 +…. . l ntotal = n 1 + n 2 + n 3 + …. . l Ptotal V =ntotal. RT l

Problem l A 6. 00 L tank contains 32. 0 g of oxygen gas and 18. 02 g of water vapor. At 20°C what is the total pressure of the tank? l 32. 0 g = 1. 00 mol O 2; 18. 02 g = 1. 00 mol H 2 O ntotal = 1. 00 + 1. 00 = 2. 00 mol l l Ptotal = ntotal. RT = 2. 00* 0. 0821* 293 K = 8. 01 atm V 6. 00 L

Practice If a 23. 0 L tank contains 3. 00 mol of H 2, 2. 00 mol of He and 1. 00 mol of Ne gases, calculate the total pressure of the tank at 40°C? Calculate the partial pressure of each gas as well. l Ptotal = 6. 7 atm l p. H 2 = 3. 35 atm l p. He = 2. 23 atm l p. Ne = 1. 12 atm l
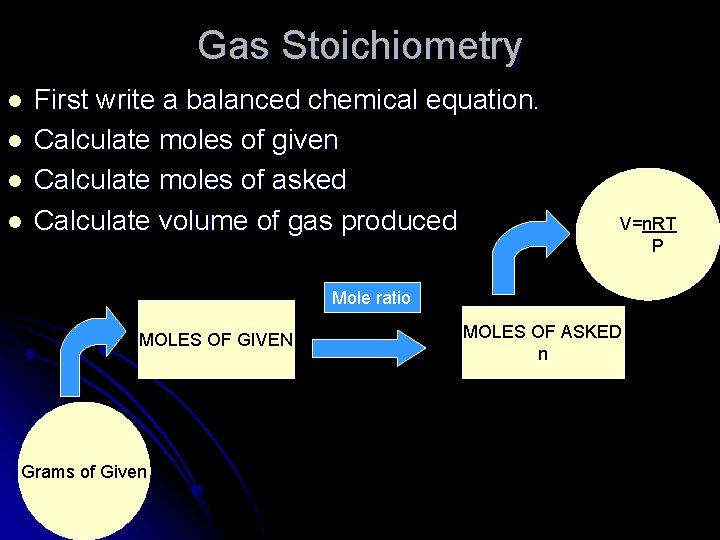
Gas Stoichiometry l l First write a balanced chemical equation. Calculate moles of given Calculate moles of asked Calculate volume of gas produced V=n. RT P Mole ratio MOLES OF GIVEN Grams of Given MOLES OF ASKED n

Sample problem l Calculate the volume of NH 3 produced when 43 g of N 2 reacts with excess H 2 at 29°C and 0. 997 atm pressure. l N 2 + 3 H 2 2 NH 3 43 g X 1 mol. N 2 X 2 mol NH 3 = 3. 07 mol NH 3 28. 02 g 1 mol. N 2 V = n. RT = 3. 07 mol* 0. 0821* 302 K = 76. L P 0. 997 atm l

Practice problem Calculate the volume of oxygen produced from the decomposition of 6. 00 g of KCl. O 3 to KCl and O 2 at 30. 0°C and 100. k. Pa pressure. l 1. 85 L l

Graham’s Law of Diffusion l l According to Graham’s law, the rates of diffusion of two gases is inversely proportional to the square root of their molar masses. In other words, lighter gases travel faster under similar conditions of temperature. Rate. A = √molar mass. B Rate. B √ molar mass. A

Practice Find the molar mass of a gas that diffuses 0. 31 times as fast as oxygen gas. l If oxygen travels at a rate of 1, then the other gas travels at 0. 31, l Rate O = √molar mass gas 2 Rate gas √molar mass O 2 1 = √molar mass gas 0. 31 √ 32. 0 g/mol molar mass gas = 330 g/mol l

Suppose a gas diffuses 1. 41 times faster than sulfur dioxide. What is the molar mass of this gas? l Rategas =√ 64. 0 g/mol Rate. SO 2 √molar massgas l 1. 41 = √ 64. 0 g/mol √ molar massgas molar mass gas = 32. 2 g/mol
- Slides: 27