Gases Chapter 13 Solids liquids and Gases Compare





















































- Slides: 62

Gases Chapter 13

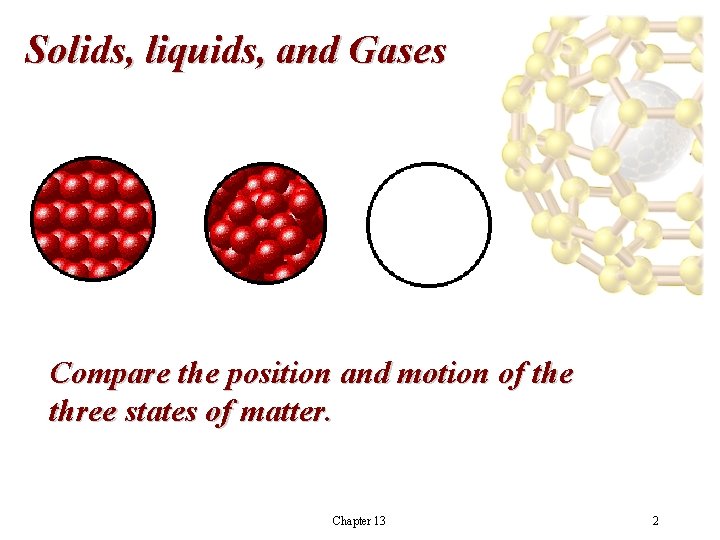
Solids, liquids, and Gases Compare the position and motion of the three states of matter. Chapter 13 2

Energy • Potential Energy – Stored energy – due to position – Particles are attracted to one another. More energy is required to keep particles farther apart. – Which of the 3 states has the highest potential energy? • Kinetic Energy – Motion energy – related to temperature – The faster the particles are moving, the higher the kinetic energy, the higher the temperature (average kinetic energy) – Which of the 3 states has the highest kinetic energy? Chapter 13 3

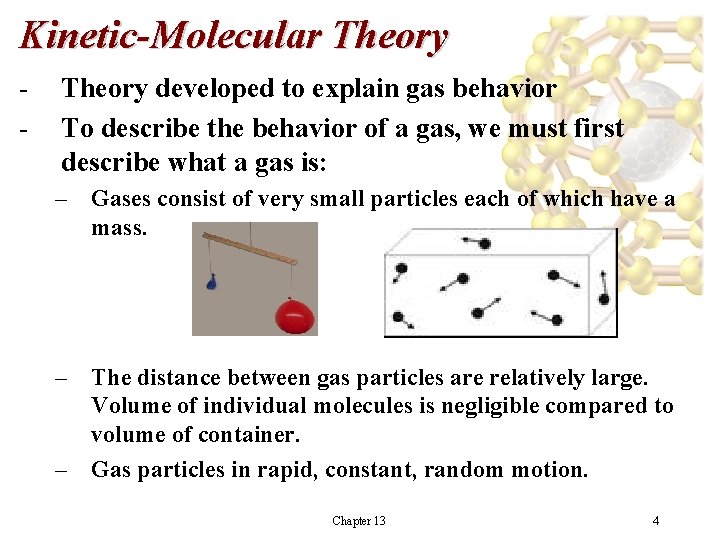
Kinetic-Molecular Theory - Theory developed to explain gas behavior To describe the behavior of a gas, we must first describe what a gas is: – Gases consist of very small particles each of which have a mass. – The distance between gas particles are relatively large. Volume of individual molecules is negligible compared to volume of container. – Gas particles in rapid, constant, random motion. Chapter 13 4

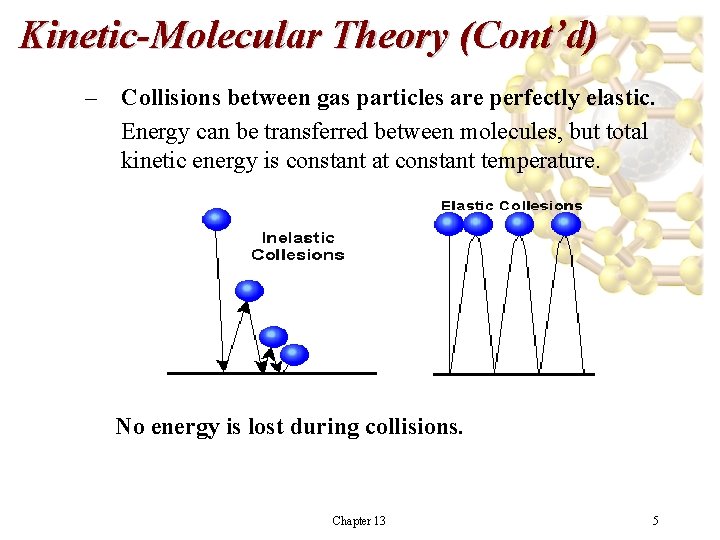
Kinetic-Molecular Theory (Cont’d) – Collisions between gas particles are perfectly elastic. Energy can be transferred between molecules, but total kinetic energy is constant at constant temperature. No energy is lost during collisions. Chapter 13 5

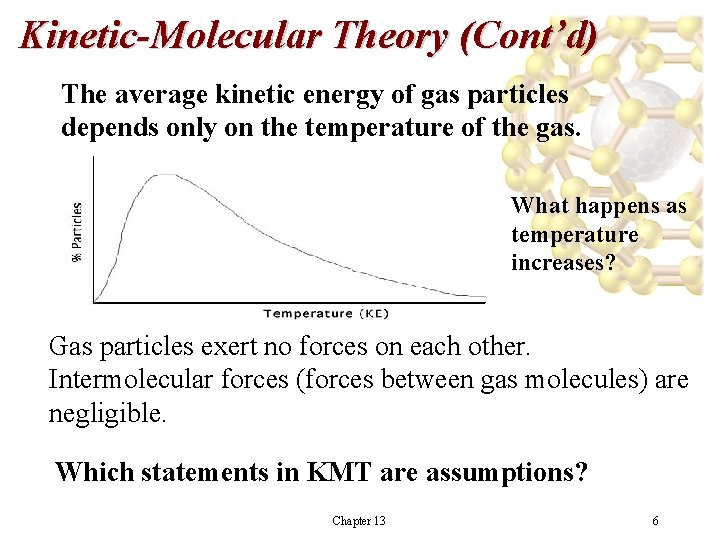
Kinetic-Molecular Theory (Cont’d) The average kinetic energy of gas particles depends only on the temperature of the gas. What happens as temperature increases? Gas particles exert no forces on each other. Intermolecular forces (forces between gas molecules) are negligible. Which statements in KMT are assumptions? Chapter 13 6

Let’s generate some gas Properties of a gas - Expand to fill a volume (expandability) - Compressible - Takes shape of container - Diffuses and flows Chapter 13 7

Variables that can be measured for gases – Temperature – Volume – Amount – Pressure Chapter 13 8

Temperature (T) • Measured in Fahrenheit, Celsius, or Kelvin. • For this chapter, we have to use Kelvin. ◦C = K – 273 K = ◦C + 273 Volume (V) • Measured in Liters, cubic meters, gallons, etc… Amount (n) • Measured in moles Chapter 13 9

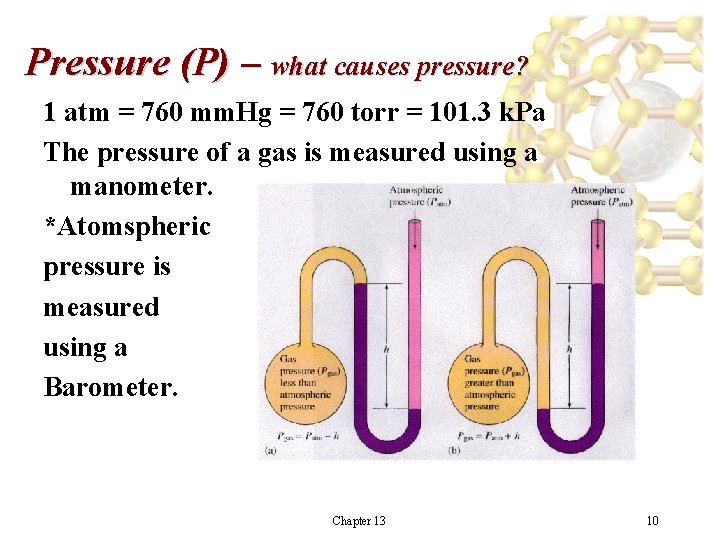
Pressure (P) – what causes pressure? 1 atm = 760 mm. Hg = 760 torr = 101. 3 k. Pa The pressure of a gas is measured using a manometer. *Atomspheric pressure is measured using a Barometer. Chapter 13 10

Unit Conversions 22◦C = ______ K 37 K = ______ ◦C 18 m. L = ______ L 4. 3 L = ______ cm 3 500 cm 3= ______ m. L 2. 2 dm 3 = _______ L Chapter 13 11

Unit Conversions 2. 8 g N 2 = ______ mol 612 mol SO 3 = _______ g 22 k. Pa = ______ atm 289 mm. Hg = _______ k. Pa 4. 3 atm = ______ torr 518 k. Pa = _______ mm. Hg Chapter 13 12

The Gas Laws - There are four variables required to describe a gas: - Amount of substance: moles (n) - Volume of substance: volume (V) - Pressures of substance: pressure (P) - Temperature of substance: temperature (T) - The gas laws will hold two of the variables constant and see how the other two vary (n, V, P, T) Chapter 13 13

Today’s Lab – Boyle’s Law • We will maintain a constant temperature and number of moles of gas. • So we will vary the Pressure and the Volume and see how they relate. PV = k inverse or P/V = k direct Chapter 13 14

Bolyle’s Law Lab – 16 pts total • • Heading – 1 pt Purpose – 1 pt Procedure – 1 pt Data – 4 pts (make sure they have units for the 3 rd and 4 th columns. • Graph – 3 pts (should have a title and labeled axis) • Questions – 6 pts (in complete sentences) 1) 1/2 2) doubled 3) inverse 4) PV=k 5) The pressure and volume of a gas at a constant temperature are inversely proportional to each other. 6) Source of error. Chapter 13 15

Variables for Gases Discussed Before • • T – Temperature V – Volume P – Pressure n – Amount of a substance (moles) Chapter 13 16

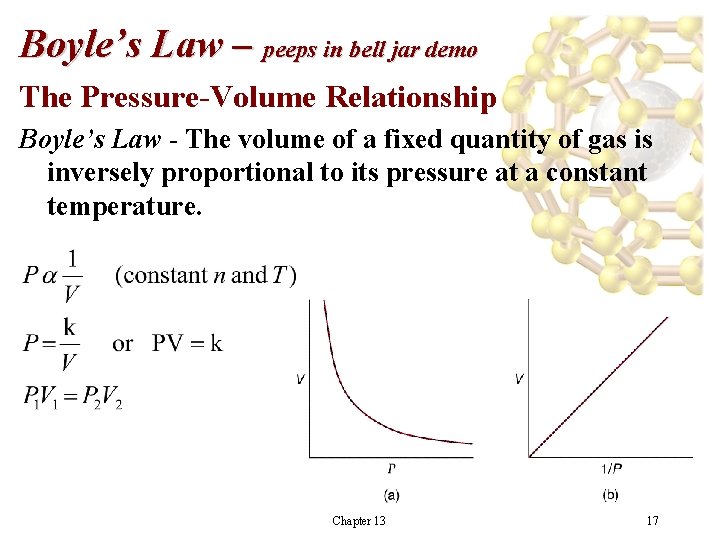
Boyle’s Law – peeps in bell jar demo The Pressure-Volume Relationship Boyle’s Law - The volume of a fixed quantity of gas is inversely proportional to its pressure at a constant temperature. Chapter 13 17

A gas occupies 22 L at 2. 43 atm. What is the new volume if the pressure changed to 5. 11 atm? Chapter 13 18

Gay-Lussac’s Law – can crush demo The Pressure-Temperature Relationship: Gay-Lussac’s Law – As the temperature of an enclosed gas increases, the pressure increases if the volume is constant. Chapter 13 19

A gas has a pressure of 1. 47 atm at 303 K. What is the new temperature if the pressure changed to 680 mm. Hg? Chapter 13 20

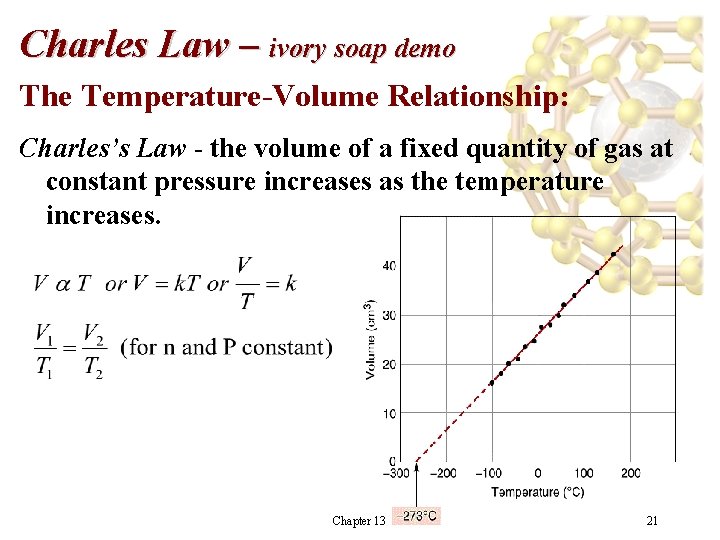
Charles Law – ivory soap demo The Temperature-Volume Relationship: Charles’s Law - the volume of a fixed quantity of gas at constant pressure increases as the temperature increases. Chapter 13 21

A gas occupies 14 L at 275 K. What is the new volume if the temperature changed to 297 K? Chapter 13 22

Avogadro’s Law The Quantity-Volume Relationship: Avogadro’s Law - The volume of gas at a given temperature and pressure is directly proportional to the number of moles of gas. Chapter 13 23

A balloon contains 1. 98 mol of a gas and has a volume of 4. 2 L. Some of the gas was let out to give a volume of 3. 1 L. What is the amount of gas left in the balloon? Chapter 13 24

The Gas Laws Summary Boyle’s Law Charles’ Law Avogadro’s Law Guy-Lussac’s Law Chapter 13 25

The Ideal Gas Equation Combine the gas laws (Boyle, Charles, Guy. Lussac, Avogadro) yields a new law or equation. Ideal gas equation: PV = n. RT P = pressure (atm or mm. Hg or k. Pa) V = volume (L) n = amount (mol) R = gas constant T = temperature (K) Chapter 13 26

Finding R with Dry Ice Lab PV = n. RT What units are used for R? Chapter 13 27

Calculating R with Dry Ice Lab – 20 pts total • • • Heading – 1 pt Purpose – 1 pt Procedure – 1 pt Data – 10 pts (make sure they have units) Calculations – 2 pts Conclusion – 5 pts – – – Their value for R Literature value for R % error Sources of error How they affected their results (higher or lower and must be consistent with their result for R) Chapter 13 28

Calculating R We define STP (standard temperature and pressure) as 0 C = 273 K, and 1 atm = 760 mm. Hg = 101. 3 k. Pa Volume of 1 mol of gas at STP is 22. 4 L. PV = n. RT Chapter 13 29

The Ideal Gas Equation-Finding R Chapter 13 30

Combined Gas Law • So in cases with changing conditions and Chapter 13 31

Ideal vs. Combined Gas Law-When Use • Ideal Gas Law: Conditions not changing • Combined Gas Law: Conditions Changing Chapter 13 32

Example 1 • How many moles of a gas at 100 degrees C does it take to fill a 1. 0 L flask to a pressure of 1. 50 atm? Not a changing situation so use Ideal Gas Law PV=n. RT Chapter 13 33

Example 2 At 60 Celsius a 0. 10 L sample of a gas has a pressure of 75. 6 k. Pa. What would its volume be at STP? Changing situation so use combined gas law. Chapter 13 34

Example 3 • What pressure would 3. 55 grams of argon gas be under in a 2. 40 L cylinder at -35 Celsius? Not a changing situation so use Ideal Gas Law PV=n. RT Chapter 13 35

Example 4 • What is the density of bromine gas the gas fills a 52. 5 L cylinder at 145 K and 583 mm. Hg? Not a changing situation so use Ideal Gas Law PV=n. RT Chapter 13 36

Example 5 A gas occupies 4430 m. L at 30 Celsius. It was transferred in a 3. 5 L cylinder. What is the new temperature. Changing situation so use combined gas law. Chapter 13 37

Demos Egg in E-flask Fountain Chapter 13 38

Tank Car • This tank car was cleaned with steam then all the valves were shut and tank car was sealed. The workers went home and when they came back the next morning this is what they saw. Chapter 13 39

Chapter 13 40

Chapter 13 41

Chapter 13 42

Ideal vs. Real gases Ideal gases behave “ideally” according to the kinetic molecular theory and follow the ideal gas law PV = n. RT. But kinetic molecular theory has several assumptions that work most of the time but not always. Chapter 13 43

Kinetic Molecular Theory Assumptions 1) The volume of gas particles are so small and the spaces between particles are so large. Therefore KMT assumes that gas particles have no volume. 2) The distance between gas particles are very large. Therefore KMT assumes that there is no attraction (IMF) between gas particles. Chapter 13 44

But Real gases have volume Under most conditions the volume of gas particles is negligible. But at small volumes or if the gas particles are large, the volume of the gas particles become significant. To compensate, we plug in the following equation for V in PV = n. RT Chapter 13 45

And real gases have attractive forces (IMF) Under most conditions, the intermolecular forces between gas particles are negligible. But at high pressure and low temperatures, the attractive forces become significant. To compensate for the pressure difference caused by IMF we plug in the following equation instead of P in PV = n. RT Chapter 13 46

Ideal vs. Real gases Which gas behaves more ideally? Ne or HCl? Neon because it’s particles have a smaller volume and weaker intermolecular forces. Chapter 13 47

Ideal vs. Real gases In PV = n. RT , plugging in We get the Van der Waals Equation Chapter 13 48

Example • Use the Van der Waals equation to calculate the temperature of 3. 6 moles of nitrogen gas in a 4. 6 L cylinder at 2. 5 atm if a and b for nitrogen are 1. 390 and 0. 03910 respectively. Chapter 13 49

Gas Mixtures and Partial Pressures Dalton’s Law - In a gas mixture the total pressure is given by the sum of partial pressures of each component: Pt = P 1 + P 2 + P 3 + … - The pressure due to an individual gas is called a partial pressure. Chapter 13 50

Partial Pressure of Dry Air At top of Mt. Everest, total atmospheric pressure is 33. 73 k. Pa (about 1/3 that of sea level). So partial pressure of O 2 is 7. 06 k. Pa. To support respiration the partial pressure must be 10. 76 k. Pa or higher so oxygen tanks are needed. Chapter 13 51

Example What is the total pressure of a mixture of gases containing oxygen, hydrogen, and water vapor if their partial pressures are 120 k. Pa, 94 k. Pa, and 137 k. Pa respectively? Chapter 13 52

Molecular Effusion and Diffusion Graham’s Law of Diffusion or Effusion – The escape of gas through a small opening. Diffusion – The movement of gas particles through another gas. Chapter 13 53

Molecular Effusion and Diffusion Graham’s Law of Effusion or Diffusion Chapter 13 54

Kinetic-Molecular Theory Molecular Speed • As kinetic energy increases, the velocity of the gas molecules increases. • Average kinetic energy, KE, is related to root mean square speed: KE= ½mv 2 Chapter 13 55

Graham’s Law of Effusion or Diffusion • Gases at same temperature – tell me about them – They have the same kinetic energy (KE) • Which would move faster? CO 2 or O 2 – O 2 would move faster because KE=1/2 mv 2 and O 2 has the lower mass • Thomas Graham noticed how rate of diffusion (or effusion) related to molar mass Chapter 13 56

Molecular Effusion and Diffusion Graham’s Law of Effusion or Diffusion Graham’s Law of Effusion – The rate of effusion of a gas is inversely proportional to the square root of its molecular mass. m 1 and m 2 are molar mass and v 1 and v 2 are effusion/diffusion rates Chapter 13 57

Molecular Effusion and Diffusion Graham’s Law of Effusion/Diffusion – The rate of effusion/diffusion of a gas is inversely proportional to the square root of its molecular mass. Chapter 13 58

Example Find the relative rate of diffusion for the gases krypton and bromine. Chapter 13 59

Gas Laws & Stoichiometry PV = n. RT Link? calculations in reactions with molar ratios Moles!! Chapter 13 60

Example 1 - What mass of Na. Cl could be produced form excess sodium and 10. 0 L of chlorine gas at 23 o. C and 1. 02 atm? Chapter 13 61

Example 2 - Calcium carbonate decomposes to form carbon dioxide and calcium oxide. What volume of carbon dioxide at 25 o. C and 99. 8 k. Pa is produced if 25. 0 g of calcium carbonate is decomposed? Chapter 13 62