GASES Chapter 12 in your text book KINETICMOLECULAR

GASES Chapter 12 in your text book

KINETIC-MOLECULAR THEORY OF GASES • Gas particles are in constant random and rapid motion. • The space between gas molecules is very large compared to the actual size of a molecule/atom of gas. • Gas molecules collide with each other and the sides of the container that holds them. • Collisions with other gas particles are completely elastic and result in no net change in kinetic energy. • Collisions with the sides of the container result in the creation of force per unit area of a surface =pressure. Pressure exerted by a gas is the result of the collisions of gas molecules against the walls of its container.

Properties of Gases • • are fluids = any substance that can flow have low density are highly compressible completely fill a container have mass exert pressure move thru each other rapidly pressure depends on the temperature and volume of container

Atmospheric Pressure • Pressure exerted on objects on surface of earth by the atmospheric gases in response to gravitational pull • STP (Standard Temperature & Atmospheric Pressure) – Amount of force applied on objects at sea level

UNITS OF PRESSURE atm = atmospheric pressure mm. Hg = millimeters of mercury Pa = pascals k. Pa = kilopascals torr = Torr

Pressure Conversions 1 atm = 760 mm. Hg 1 atm = 760 torr 1 atm = 101. 3 k. Pa 1 atm = 101, 325 Pa 1 atm = 14. 70 lb/in 2 (psi) 1 bar = 100, 000 pascals 1 N/m 2 = 1 pascal So, 1 atm = 760 mm. Hg = 760 torr = 101. 3 k. Pa = 101, 325 Pa

How does elevation affect atmospheric pressure? High elevations = less atmospheric pressure (less dense air exerts less pressure) EX. Denver Colorado Low elevations = greater atmospheric pressure (atmosphere is denser as you move closer to the earth’s surface) EX. Death Valley
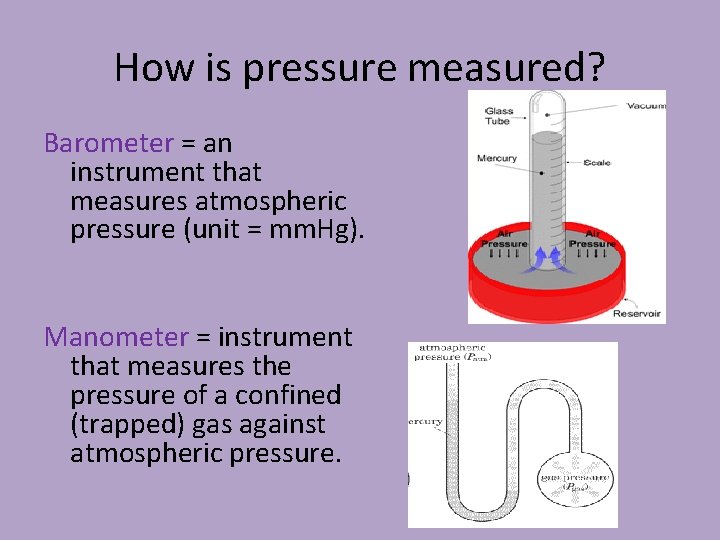
How is pressure measured? Barometer = an instrument that measures atmospheric pressure (unit = mm. Hg). Manometer = instrument that measures the pressure of a confined (trapped) gas against atmospheric pressure.
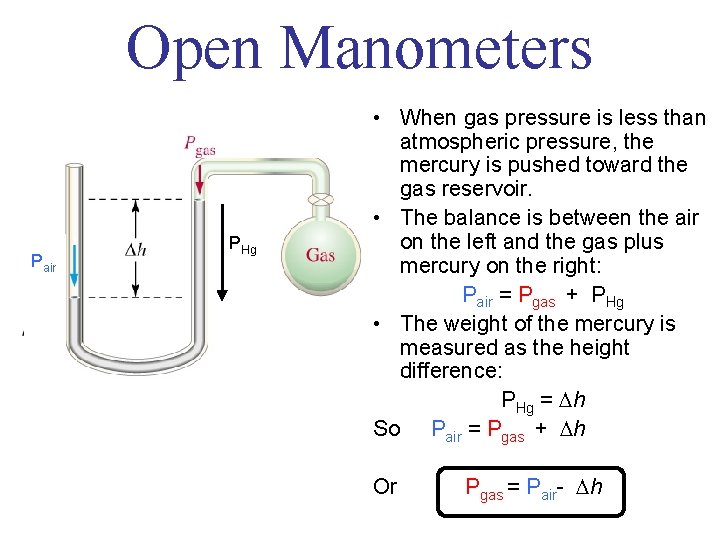
Open Manometers Pair PHg • When gas pressure is less than atmospheric pressure, the mercury is pushed toward the gas reservoir. • The balance is between the air on the left and the gas plus mercury on the right: Pair = Pgas + PHg • The weight of the mercury is measured as the height difference: PHg = h So Pair = Pgas + h Or Pgas = Pair- h
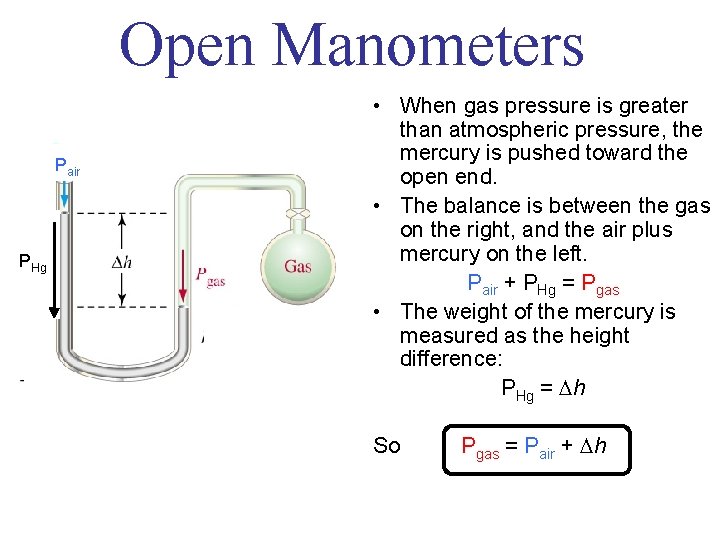
Open Manometers Pair PHg • When gas pressure is greater than atmospheric pressure, the mercury is pushed toward the open end. • The balance is between the gas on the right, and the air plus mercury on the left. Pair + PHg = Pgas • The weight of the mercury is measured as the height difference: PHg = h So Pgas = Pair + h
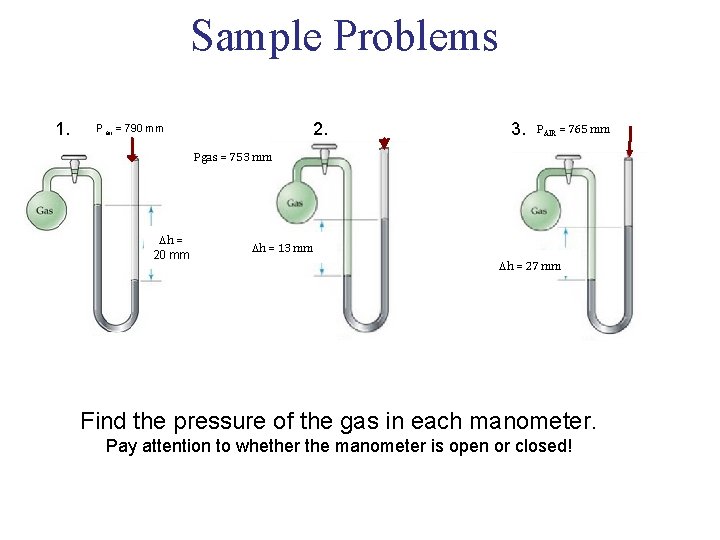
Sample Problems 1. 2. Pair = 790 mm 3. PAIR = 765 mm Pgas = 753 mm h = 20 mm h = 13 mm h = 27 mm Find the pressure of the gas in each manometer. Pay attention to whether the manometer is open or closed!

Sample Problem Answers 1. Pgas+ h = Pair Pgas= 790 mm - 20 mm = 770 mm Hg 2. Pgas = vacuum + 13 mm = 13 mm Hg 3. Pgas = Pair + h Pgas = 765 + 27 = 792 mm Hg

Measureable Properties of Gases • • P = pressure exerted by gas V = volume (L) occupied by the gas T = temperature (K) of the gas n = number of moles of the gas

Boyle’s Law P 1 x V 1 = P 2 x V 2 When the temperature and the amount of gas is constant, the pressure & volume have an indirect relationship. if V increases, the P decreases if V decreases, the P increases


Example • A syringe holds 2. 0 ml of oxygen gas at a pressure of 672 mm. Hg. The volume of the syringe is reduced to 1. 5 ml by pushing in the plunger. If temperature and moles remain constant what is the new pressure in atmospheres?

Charles’s Law V 1 = V 2 T 1 T 2 When the pressure and amount of gas are constant, temperature and volume have a direct relationship: if V increases, T increases if V decreases, T decreases


EXAMPLE If 1, 235 m. L of nitrogen gas is placed in a sealed container at a temperature of 57°C, what will its volume (in liters) be when the temperature of the gas reaches 150°C?

62. 7 m. L of helium is at a pressure of 842 mm. Hg. The volume of the gas increased to 96. 5 m. L, what is the new pressure?

Question of the Day If 254 m. L of nitrogen gas is placed in a sealed container at a temperature of 52°C, what will its volume (in liters) be when the temperature of the gas reaches 140°C?
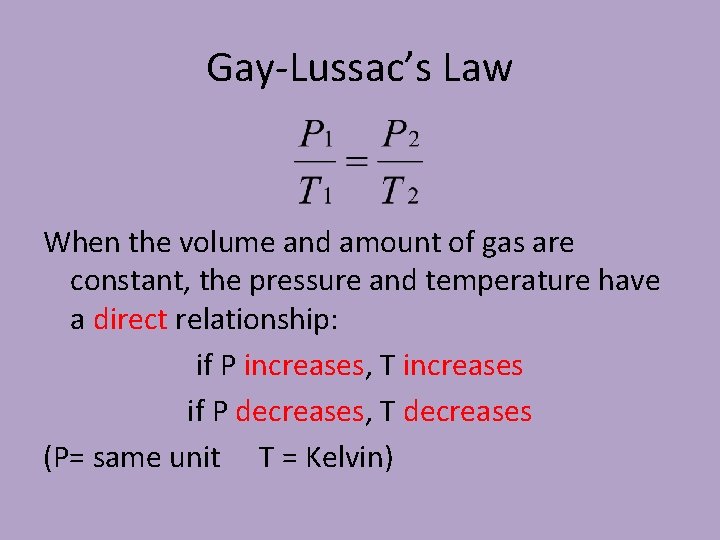
Gay-Lussac’s Law When the volume and amount of gas are constant, the pressure and temperature have a direct relationship: if P increases, T increases if P decreases, T decreases (P= same unit T = Kelvin)

Avogadro’s Law When pressure and temperature are constant, volume & the number of moles have a direct relationship if V increases, moles increases if V decreases, moles decreases (V = liters n = moles)

Gay-Lussac’s Law A can of gas is at 30. kpa and 20°C. What is its pressure at 48°C

Avogadro’s Law If 0. 105 mol of helium gas occupies a volume of 2. 35 L at a certain temperature & pressure, what volume would 0. 432 mol of helium occupy under the same conditions?
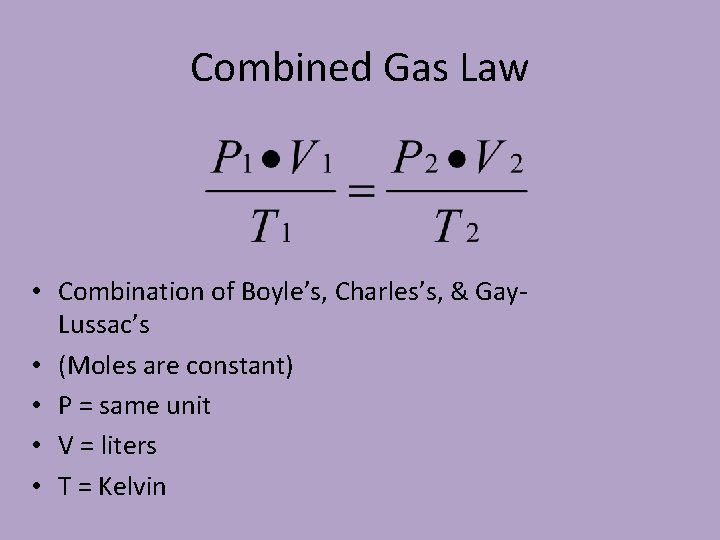
Combined Gas Law • Combination of Boyle’s, Charles’s, & Gay. Lussac’s • (Moles are constant) • P = same unit • V = liters • T = Kelvin

Combined Gas Law A sample of methane gas that has a volume of 3. 8 L at 8°C and 362 k. Pa is heated to 84°C and the pressure is raised to 573 k. Pa. Calculate its new volume

Dalton’s Law of Partial Pressures • Each gas in a mixture creates pressure as if the other gases were not present. • The total pressure is the sum of the pressures created by the gases in the mixture • The pressure each gas exerts in the mixture is called its partial pressure Ptotal = P 1 + P 2 + P 3 + ……. P 4

Dalton’s Law A mixture of neon and argon gases exert a total pressure of 2. 39 atm. The partial pressure of neon alone is 1. 84 atm. What is the partial pressure of argon gas?

Ideal Gas Law PV = n. RT P must be in atm V must be in liters T must be in Kelvin n must be in moles R = 0. 08206

Ideal Gas Law A weather balloon contains 1. 10 x 105 mol of helium and has a volume of 2. 37 x 106 L at 1. 00 atm. Calculate the temperature of the helium in the balloon in Kelvin and °C.
- Slides: 31