Gases Chapter 10 Elements that exist as gases







































- Slides: 64

Gases Chapter 10

Elements that exist as gases 250 C and 1 atmosphere 10. 1

10. 1

Physical Characteristics of Gases • Gases assume the volume and shape of their containers. • Gases are the most compressible state of matter. • Gases will mix evenly and completely when confined to the same container. • Gases have much lower densities than liquids and solids. 10. 1

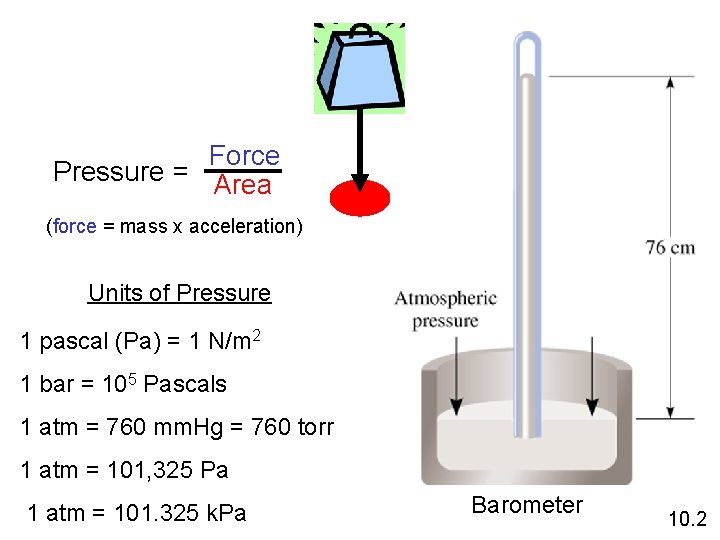
Force Pressure = Area (force = mass x acceleration) Units of Pressure 1 pascal (Pa) = 1 N/m 2 1 bar = 105 Pascals 1 atm = 760 mm. Hg = 760 torr 1 atm = 101, 325 Pa 1 atm = 101. 325 k. Pa Barometer 10. 2

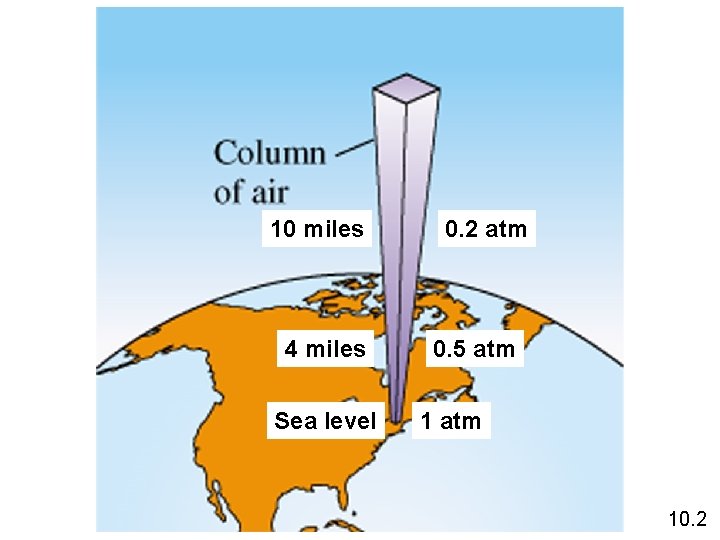
10 miles 4 miles Sea level 0. 2 atm 0. 5 atm 10. 2

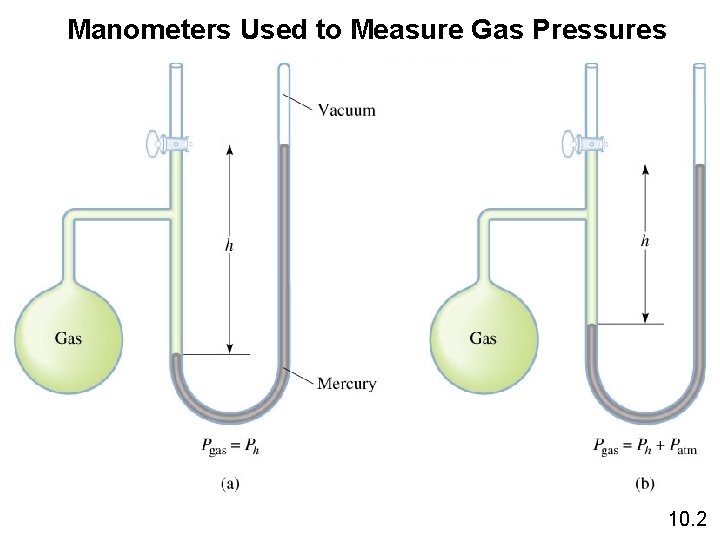
Manometers Used to Measure Gas Pressures 10. 2

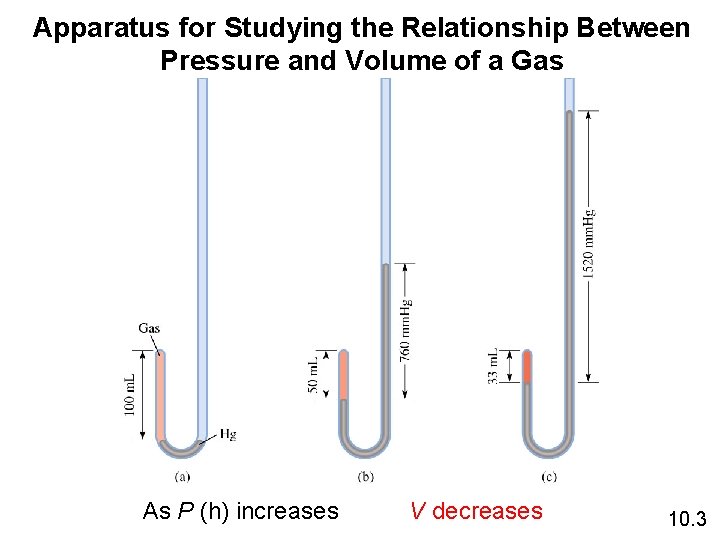
Apparatus for Studying the Relationship Between Pressure and Volume of a Gas As P (h) increases V decreases 10. 3

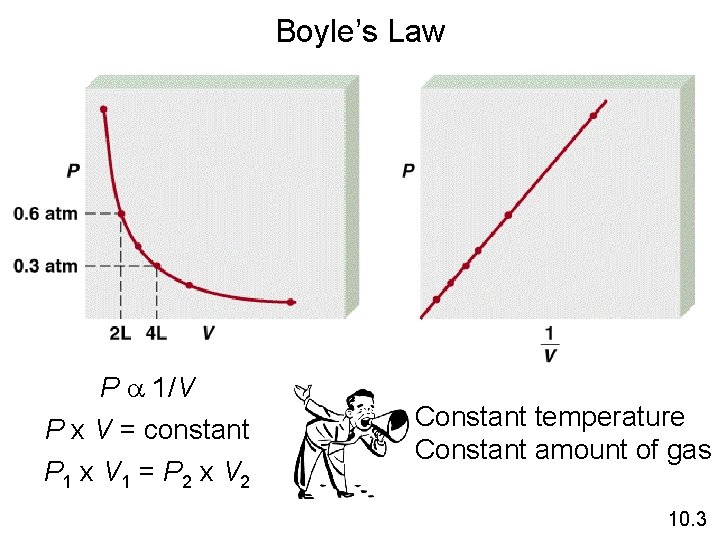
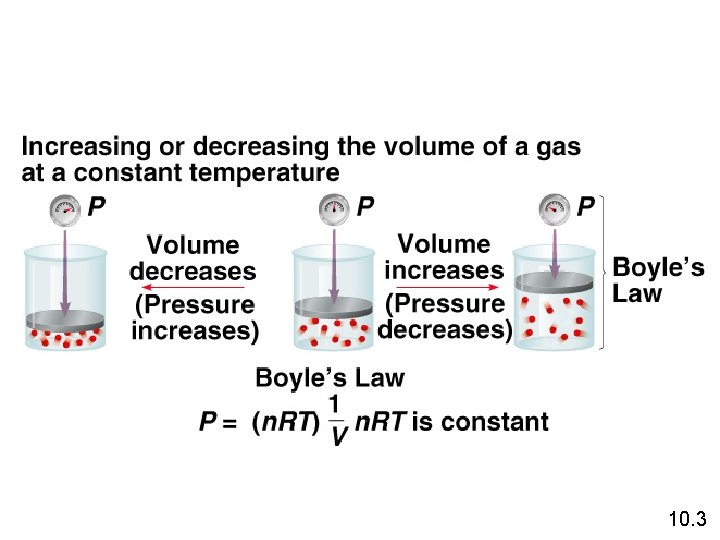
Boyle’s Law The volume of a fixed quantity of gas at constant temperature is inversely proportional to the pressure. 10. 3

Boyle’s Law P a 1/V P x V = constant P 1 x V 1 = P 2 x V 2 Constant temperature Constant amount of gas 10. 3

A sample of chlorine gas occupies a volume of 946 m. L at a pressure of 726 mm. Hg. What is the pressure of the gas (in mm. Hg) if the volume is reduced at constant temperature to 154 m. L? P x V = constant P 1 x V 1 = P 2 x V 2 P 1 = 726 mm. Hg P 2 = ? V 1 = 946 m. L V 2 = 154 m. L P 1 x V 1 726 mm. Hg x 946 m. L = P 2 = = 4460 mm. Hg 154 m. L V 2 10. 3

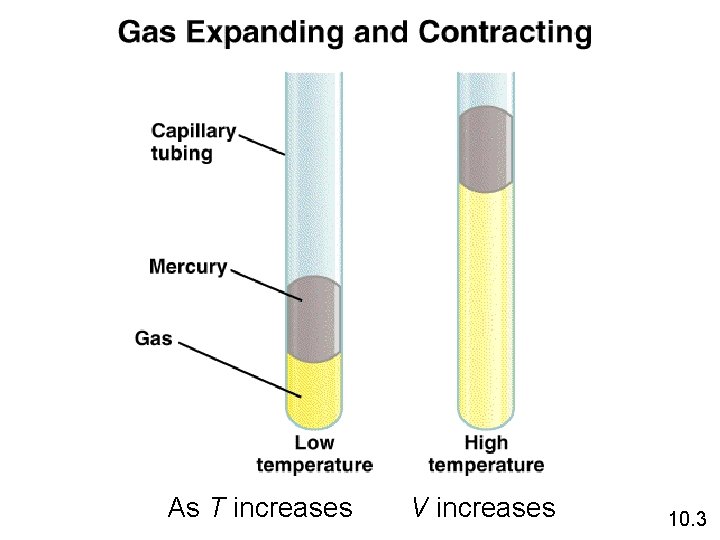
As T increases V increases 10. 3

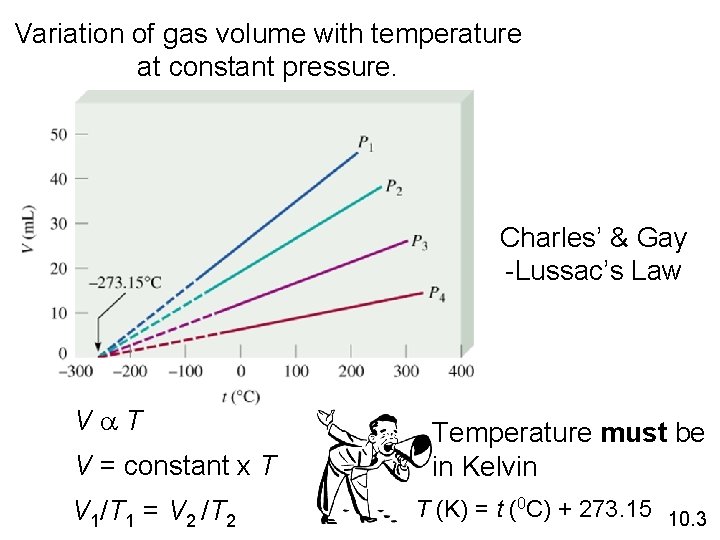
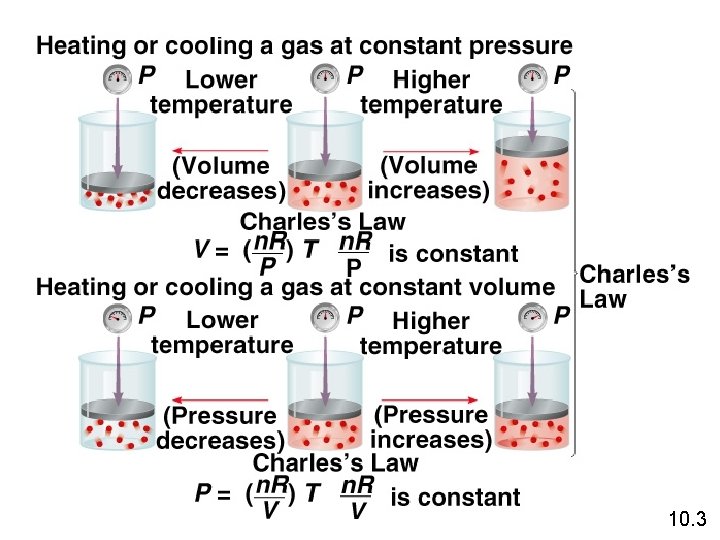
Variation of gas volume with temperature at constant pressure. Charles’ & Gay -Lussac’s Law Va. T V = constant x T V 1/T 1 = V 2 /T 2 Temperature must be in Kelvin T (K) = t (0 C) + 273. 15 10. 3

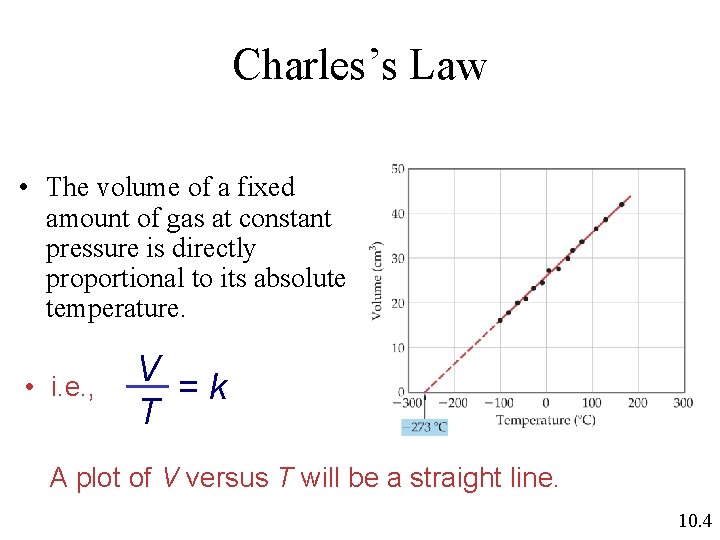
Charles’s Law • The volume of a fixed amount of gas at constant pressure is directly proportional to its absolute temperature. • i. e. , V =k T A plot of V versus T will be a straight line. 10. 4

A sample of carbon monoxide gas occupies 3. 20 L at 125 0 C. At what temperature will the gas occupy a volume of 1. 54 L if the pressure remains constant? V 1 /T 1 = V 2 /T 2 V 1 = 3. 20 L V 2 = 1. 54 L T 1 = 398. 15 K T 2 = ? T 1 = 125 (0 C) + 273. 15 (K) = 398. 15 K V 2 x T 1 = T 2 = V 1 1. 54 L x 398. 15 K 3. 20 L = 192 K 10. 3

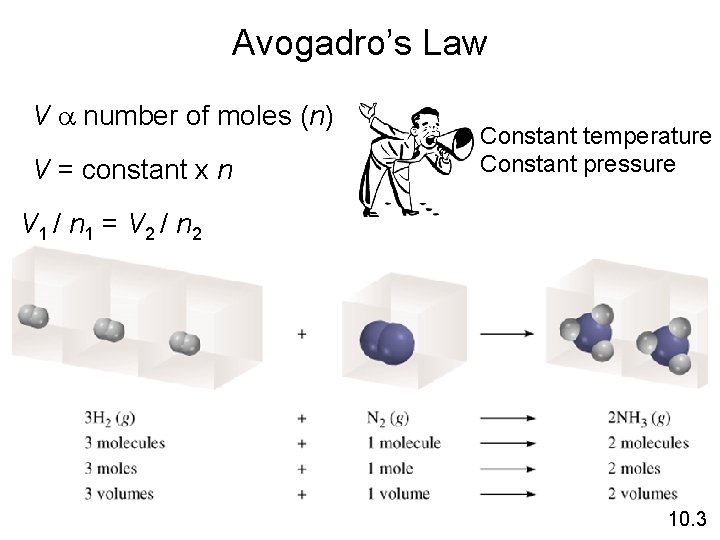
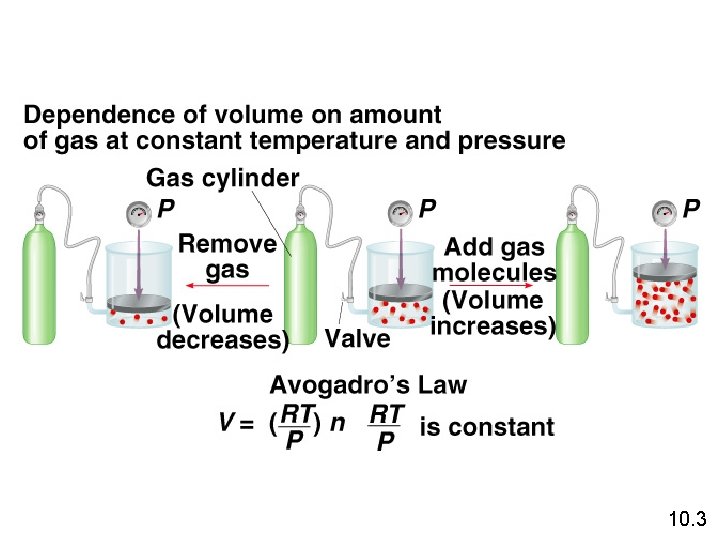
Avogadro’s Law • The volume of a gas at constant temperature and pressure is directly proportional to the number of moles of the gas. • Mathematically, this means V = kn 10. 3

Avogadro’s Law V a number of moles (n) V = constant x n Constant temperature Constant pressure V 1 / n 1 = V 2 / n 2 10. 3

Ammonia burns in oxygen to form nitric oxide (NO) and water vapor. How many volumes of NO are obtained from one volume of ammonia at the same temperature and pressure? 4 NH 3 + 5 O 2 1 mole NH 3 4 NO + 6 H 2 O 1 mole NO At constant T and P 1 volume NH 3 1 volume NO 10. 3

Final and Initial State Problems • Use Combined Gas Law • List all variables (P, V, n, T) in two columns… one for initial state, one for final state. • Substitute and solve ( make sure units cancel)

Final and Initial State Problems A sample of gas occupies 355 ml and 15 o. C and 755 torr of pressure. What temperature will the gas have at the same pressure if its volume increases to 453 ml?

Final and Initial State Problems A sample of gas at 30. 0 o has a pressure of 1. 23 bar and a volume of 0. 0247 m 3. If the volume of the gas is compressed to 0. 00839 m 3 at the same temperature, what is its pressure at this volume?

Final and Initial State Problems • A Sample of gas at 27. 0 o. C has a volume of 2. 08 L and a pressure of 750. 0 mm Hg. If the gas is in a sealed container, what is its pressure in bar when the temperature (in o. C) doubles?

Ideal-Gas Equation • So far we’ve seen that V 1/P (Boyle’s law) V T (Charles’s law) V n (Avogadro’s law) • Combining these, we get n. T V P 10. 3

10. 3

10. 3

10. 3

Ideal-Gas Equation The relationship then becomes n. T V P n. T V=R P or PV = n. RT 10. 4

Ideal-Gas Equation The constant of proportionality is known as R, the ideal gas constant. 10. 4

Ideal Gas Equation Boyle’s law: V a 1 (at constant n and T) P Charles’ law: V a T (at constant n and P) Avogadro’s law: V a n (at constant P and T) Va n. T P n. T V = constant x =R P P R is the gas constant PV = n. RT 10. 4

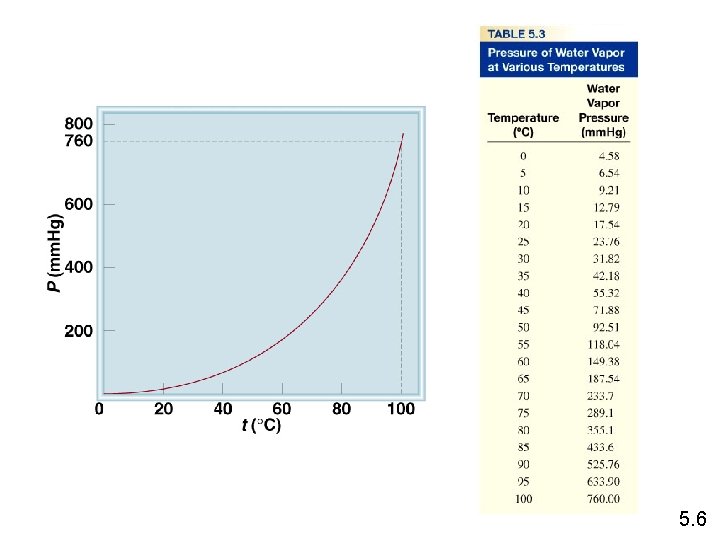
The conditions 0 0 C and 1 atm are called standard temperature and pressure (STP). Experiments show that at STP, 1 mole of an ideal gas occupies 22. 414 L. PV = n. RT (1 atm)(22. 414 L) PV R= = n. T (1 mol)(273. 15 K) R = 0. 082057 L • atm / (mol • K) 10. 4

What is the volume (in liters) occupied by 49. 8 g of HCl at STP? T = 0 0 C = 273. 15 K PV = n. RT V= P P = 1 atm n = 49. 8 g x 1. 37 mol x 0. 0821 V= L • atm mol • K 1 mol HCl = 1. 37 mol 36. 45 g HCl x 273. 15 K 1 atm V = 30. 6 L 10. 4

Argon is an inert gas used in lightbulbs to retard the vaporization of the filament. A certain lightbulb containing argon at 1. 20 atm and 18 0 C is heated to 85 0 C at constant volume. What is the final pressure of argon in the lightbulb (in atm)? PV = n. RT n, V and R are constant n. R = P = constant T V P 1 P 2 = T 1 T 2 P 1 = 1. 20 atm T 1 = 291 K P 2 = ? T 2 = 358 K T 2 = 1. 20 atm x 358 K = 1. 48 atm P 2 = P 1 x 291 K T 1 10. 4

Single State Problems • Use Ideal gas Law – you’re given three for the four variables and R - solve for the unknown. • PV = n. RT

Single State Problems • How many grams of oxygen gas in a 10. 0 L container will exert a pressure of 712 torr at a temperature of 25. 0 o C?

Single State Problems • At what temperature will 26. 42 grams of sulfur dioxide in a 250. 0 ml container exert a pressure of 0. 842 atm?

Other calculations involving the Ideal Gas Law • We can often determine the molar mass of a gas by measuring its mass at a given volume, temperature and pressure. • Ideal Gas Law can also be used to determine the density of a gas at given conditions of temperature and pressure.

Densities of Gases If we divide both sides of the ideal-gas equation by V and by RT, we get n P = V RT 10. 4

Densities of Gases • We know that – moles molecular mass = mass n =m • So multiplying both sides by the molecular mass ( ) gives m P = V RT 10. 4

Densities of Gases • Mass volume = density • So, m P d= = V RT Note: One only needs to know the molecular mass, the pressure, and the temperature to calculate the density of a gas. 10. 4

Molecular Mass We can manipulate the density equation to enable us to find the molecular mass of a gas: P d= RT Becomes d. RT = P 10. 4

Density (d) Calculations PM m d= = V RT m is the mass of the gas in g M is the molar mass of the gas Molar Mass (M ) of a Gaseous Substance d. RT M= P d is the density of the gas in g/L 10. 4

A 2. 10 -L vessel contains 4. 65 g of a gas at 1. 00 atm and 27. 0 0 C. What is the molar mass of the gas? d. RT M= P M= 2. 21 g L 4. 65 g m = = 2. 21 d= V 2. 10 L x 0. 0821 L • atm mol • K g L x 300. 15 K 1 atm M = 54. 6 g/mol 10. 4

• When 4. 93 grams of carbon tetrachloride gas are in a 1. oo liter container at 400. 0 K, the gas exerts a pressure of 800. 0 torr. What is the molar mass of carbon tetrachloride?

• A gaseous compound with a mass of 3. 216 grams has a volume of 2236 m. L at 27. 0 o. C and 735 mm Hg. Calculate the molar mass of the compound.

• Calculate the density of ammonia at 0 o. C and 1 atm of pressure.

• Calculate the molar mass of a compound with a density of 0. 630 g/L at 25. 0 o. C and 730. 0 torr pressure.

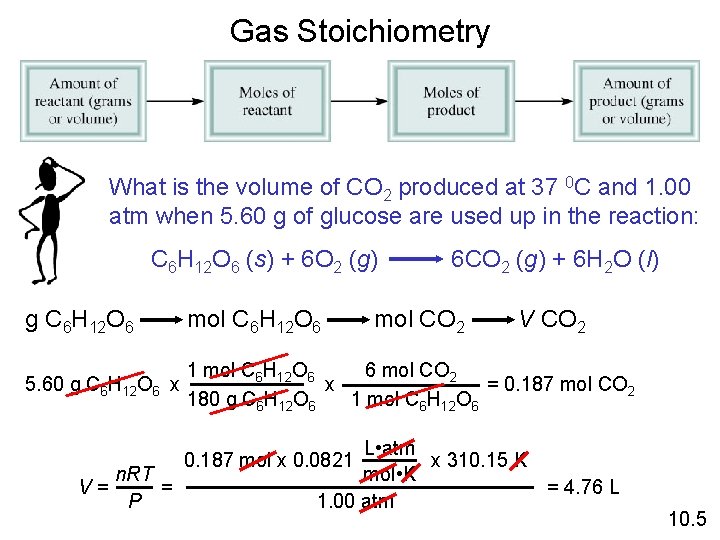
Gas Stoichiometry What is the volume of CO 2 produced at 37 0 C and 1. 00 atm when 5. 60 g of glucose are used up in the reaction: C 6 H 12 O 6 (s) + 6 O 2 (g) 6 CO 2 (g) + 6 H 2 O (l) g C 6 H 12 O 6 mol C 6 H 12 O 6 5. 60 g C 6 H 12 O 6 x 6 mol CO 2 1 mol C 6 H 12 O 6 x = 0. 187 mol CO 2 180 g C 6 H 12 O 6 1 mol C 6 H 12 O 6 V= n. RT = P mol CO 2 V CO 2 L • atm x 310. 15 K mol • K 1. 00 atm 0. 187 mol x 0. 0821 = 4. 76 L 10. 5

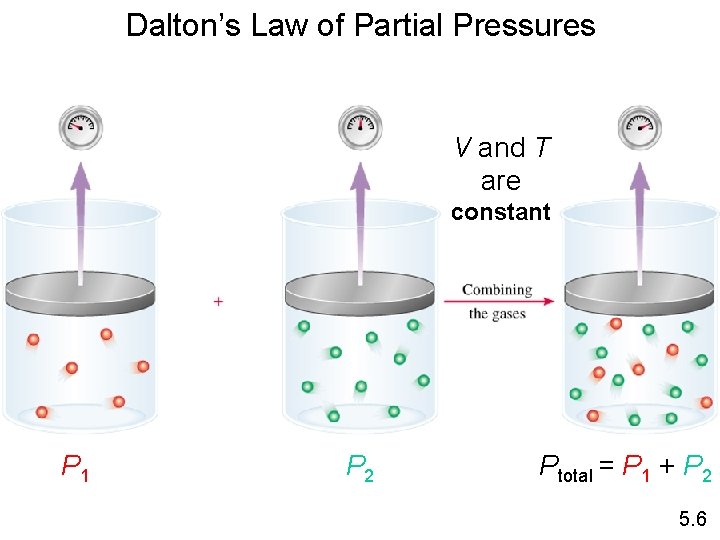
Dalton’s Law of Partial Pressures V and T are constant P 1 P 2 Ptotal = P 1 + P 2 5. 6

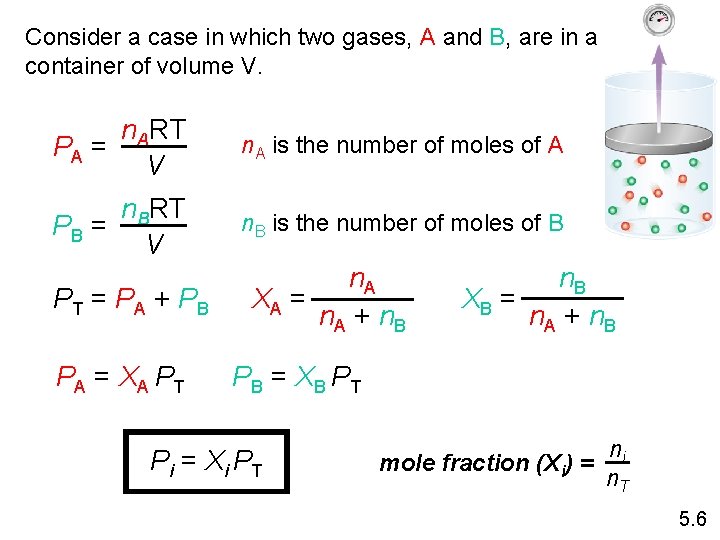
Consider a case in which two gases, A and B, are in a container of volume V. n. ART PA = V n. A is the number of moles of A n. BRT PB = V n. B is the number of moles of B PT = PA + PB PA = XA PT n. A XA = n. A + n. B XB = n. A + n. B PB = XB PT Pi = Xi PT mole fraction (Xi) = ni n. T 5. 6

A sample of natural gas contains 8. 24 moles of CH 4, 0. 421 moles of C 2 H 6, and 0. 116 moles of C 3 H 8. If the total pressure of the gases is 1. 37 atm, what is the partial pressure of propane (C 3 H 8)? Pi = Xi PT PT = 1. 37 atm 0. 116 Xpropane = 8. 24 + 0. 421 + 0. 116 = 0. 0132 Ppropane = 0. 0132 x 1. 37 atm = 0. 0181 atm 5. 6

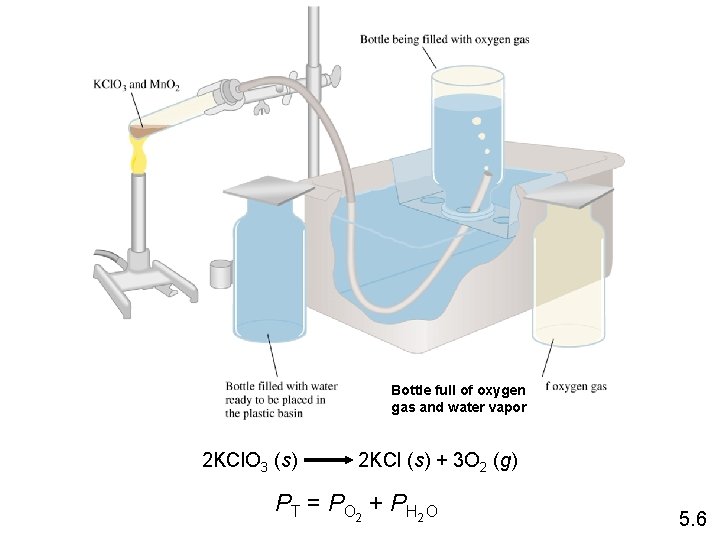
Bottle full of oxygen gas and water vapor 2 KCl. O 3 (s) 2 KCl (s) + 3 O 2 (g) PT = PO 2 + PH 2 O 5. 6

5. 6

Chemistry in Action: Scuba Diving and the Gas Laws P Depth (ft) Pressure (atm) 0 1 33 2 66 3 V 5. 6

Kinetic Molecular Theory of Gases 1. A gas is composed of molecules that are separated from each other by distances far greater than their own dimensions. The molecules can be considered to be points; that is, they possess mass but have negligible volume. 2. Gas molecules are in constant motion in random directions, and they frequently collide with one another. Collisions among molecules are perfectly elastic. 3. Gas molecules exert neither attractive nor repulsive forces on one another. 4. The average kinetic energy of the molecules is proportional to the temperature of the gas in kelvins. Any two gases at the same temperature will have the same average kinetic energy KE = ½ mu 2 5. 7

Kinetic theory of gases and … • Compressibility of Gases • Boyle’s Law P a collision rate with wall Collision rate a number density Number density a 1/V P a 1/V • Charles’ Law P a collision rate with wall Collision rate a average kinetic energy of gas molecules Average kinetic energy a T Pa. T 5. 7

Kinetic theory of gases and … • Avogadro’s Law P a collision rate with wall Collision rate a number density Number density a n Pan • Dalton’s Law of Partial Pressures Molecules do not attract or repel one another P exerted by one type of molecule is unaffected by the presence of another gas Ptotal = SPi 5. 7

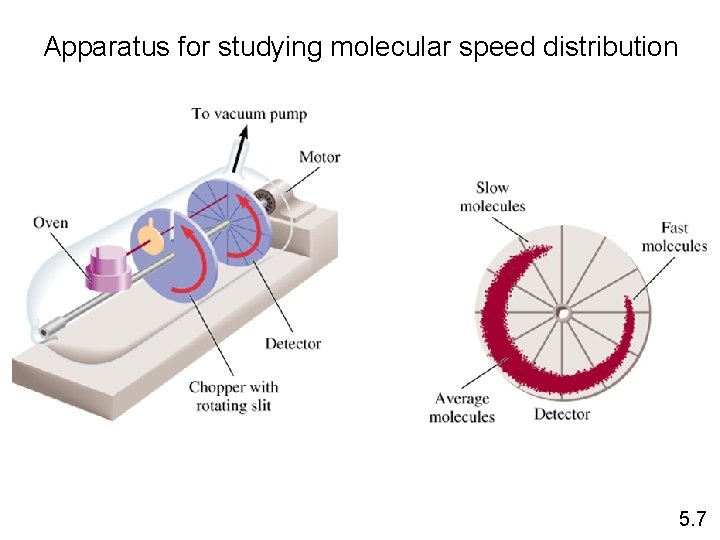
Apparatus for studying molecular speed distribution 5. 7

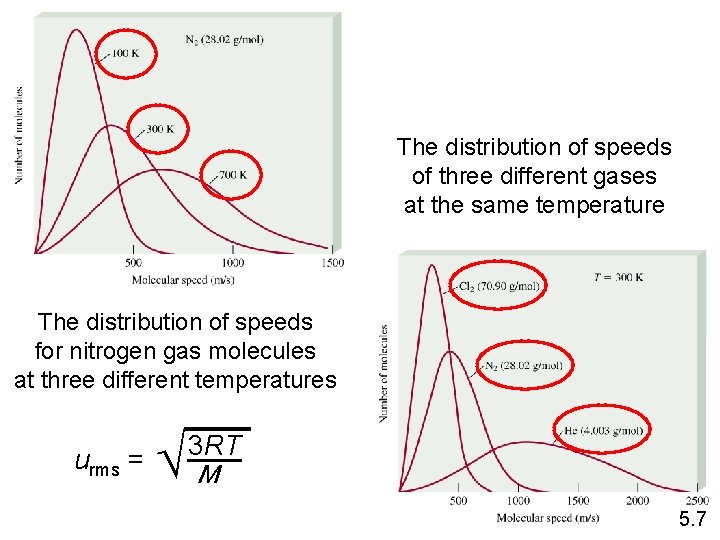
The distribution of speeds of three different gases at the same temperature The distribution of speeds for nitrogen gas molecules at three different temperatures urms = M 3 RT 5. 7

Chemistry in Action: Super Cold Atoms Gaseous Rb Atoms 1. 7 x 10 -7 K Bose-Einstein Condensate

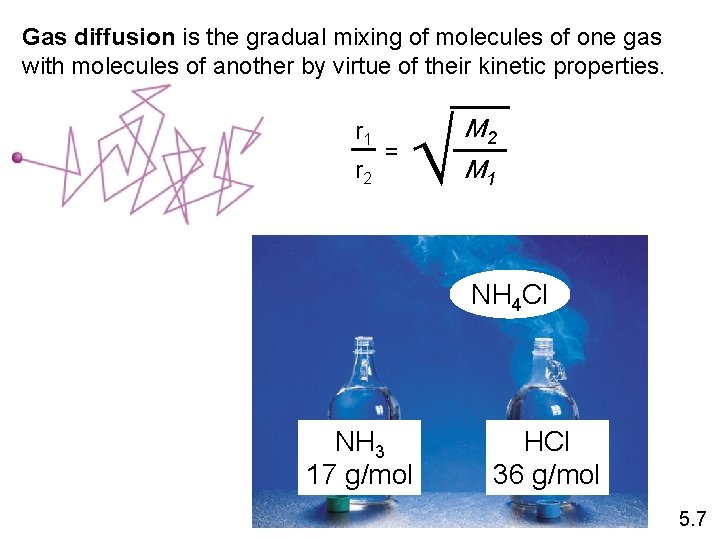
Gas diffusion is the gradual mixing of molecules of one gas with molecules of another by virtue of their kinetic properties. r 1 r 2 = M 2 M 1 NH 4 Cl NH 3 17 g/mol HCl 36 g/mol 5. 7

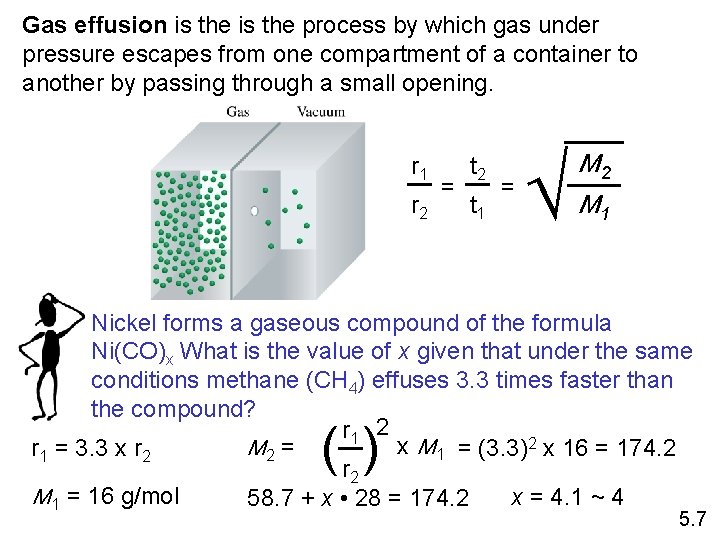
Gas effusion is the process by which gas under pressure escapes from one compartment of a container to another by passing through a small opening. r 1 r 2 = t 2 t 1 = M 2 M 1 Nickel forms a gaseous compound of the formula Ni(CO)x What is the value of x given that under the same conditions methane (CH 4) effuses 3. 3 times faster than the compound? r 1 2 x M 1 = (3. 3)2 x 16 = 174. 2 M 2 = r 1 = 3. 3 x r 2 M 1 = 16 g/mol x = 4. 1 ~ 4 58. 7 + x • 28 = 174. 2 ( ) 5. 7

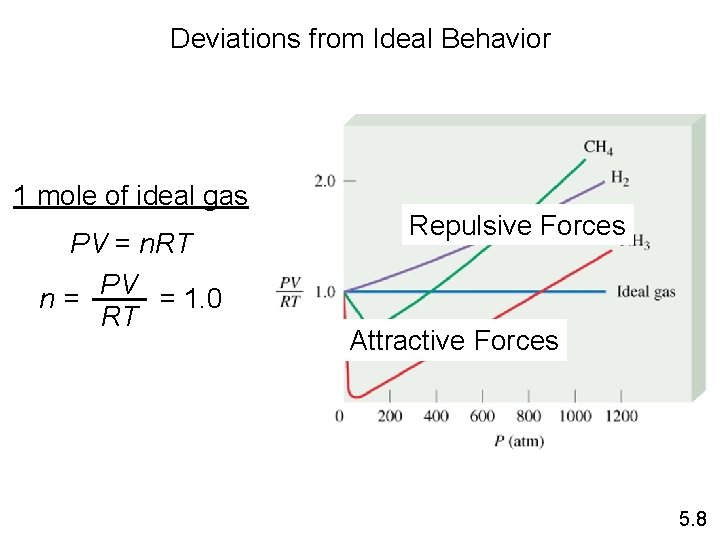
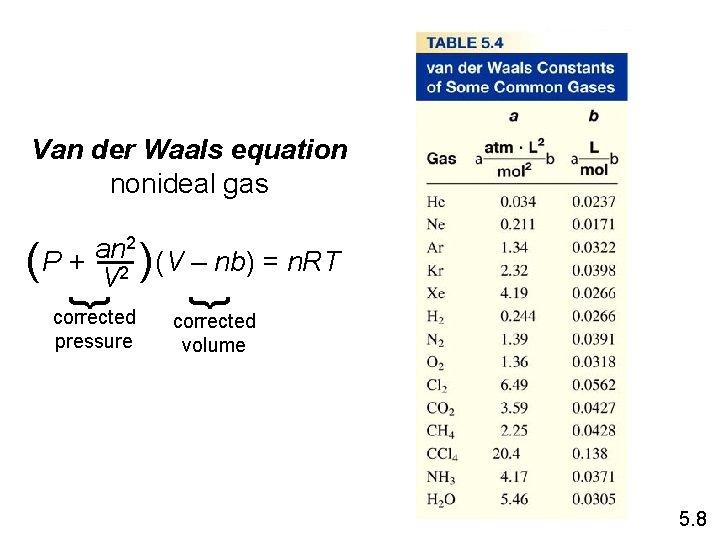
Deviations from Ideal Behavior 1 mole of ideal gas PV = n. RT PV = 1. 0 n= RT Repulsive Forces Attractive Forces 5. 8

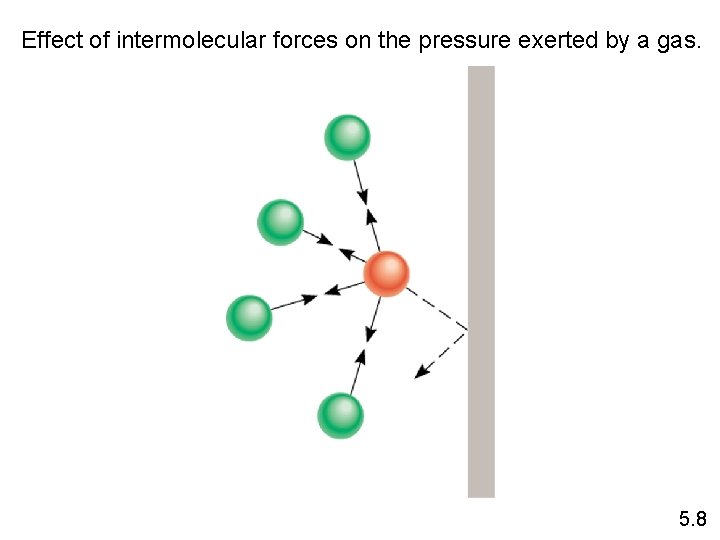
Effect of intermolecular forces on the pressure exerted by a gas. 5. 8

Van der Waals equation nonideal gas 2 an ( P + V 2 )(V – nb) = n. RT } } corrected pressure corrected volume 5. 8