Gas Stoichiometry Using the Ideal Gas Law In











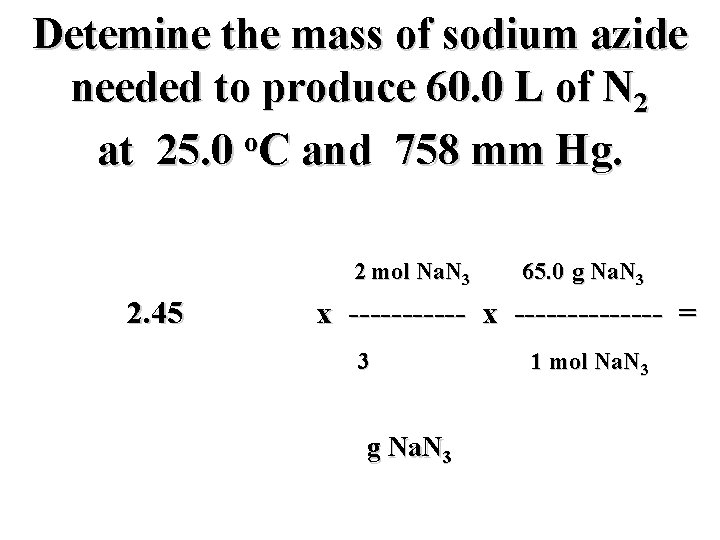

- Slides: 61

Gas Stoichiometry Using the Ideal Gas Law In Chemical Reactions.

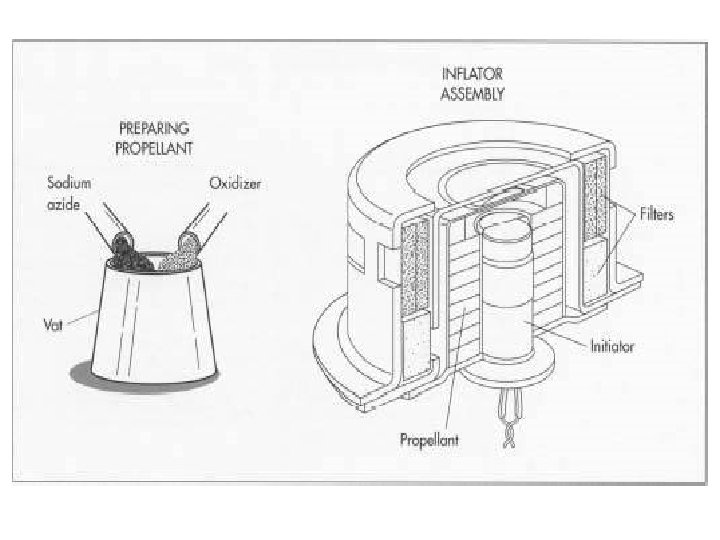
The Automotive Airbag The airbag is responsible for saving many thousands of lives and represents an important application of Gas Stoichiometry.

The Automotive Airbag

The Automotive Airbag The idea of providing protection for occupants of vehicles using some type of cushioning device is not new.

The Automotive Airbag Today’s modern airbag systems inflate in milliseconds and have proven so effective that they are now required on all cars sold in the USA and most other countries.

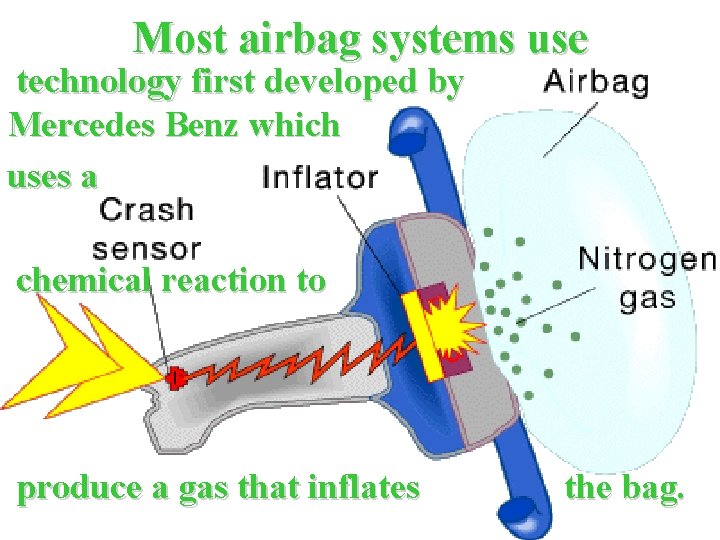
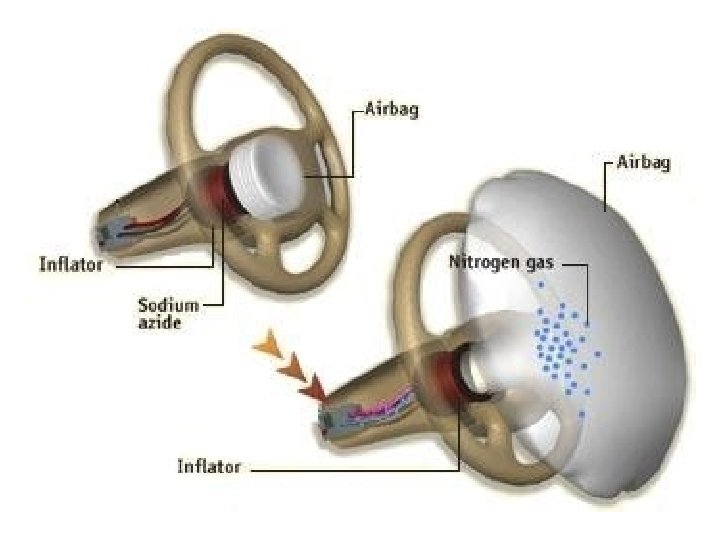
Most airbag systems use technology first developed by Mercedes Benz which uses a chemical reaction to produce a gas that inflates the bag.


The Automotive Airbag

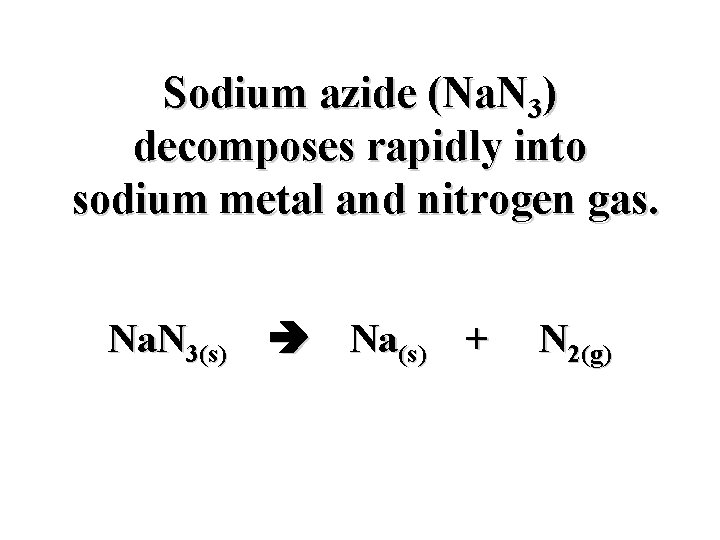
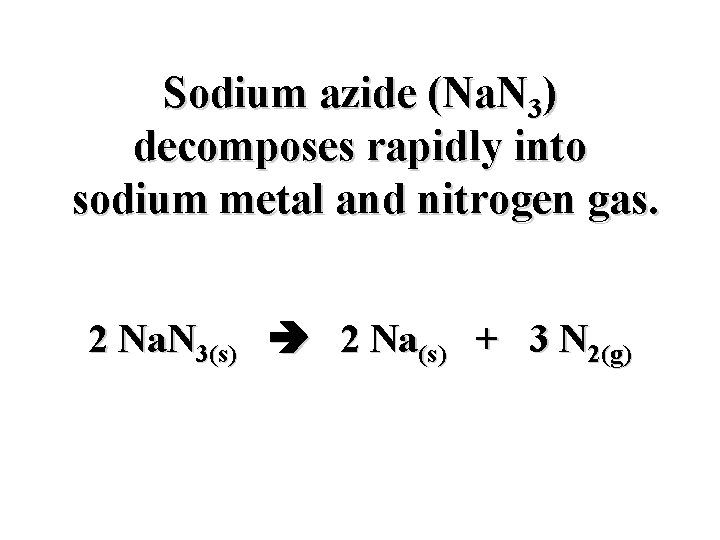
Sodium azide (Na. N 3) decomposes rapidly into sodium metal and nitrogen gas. Na. N 3(s) Na(s) + N 2(g)

Sodium azide (Na. N 3) decomposes rapidly into sodium metal and nitrogen gas. 2 Na. N 3(s) 2 Na(s) + 3 N 2(g)




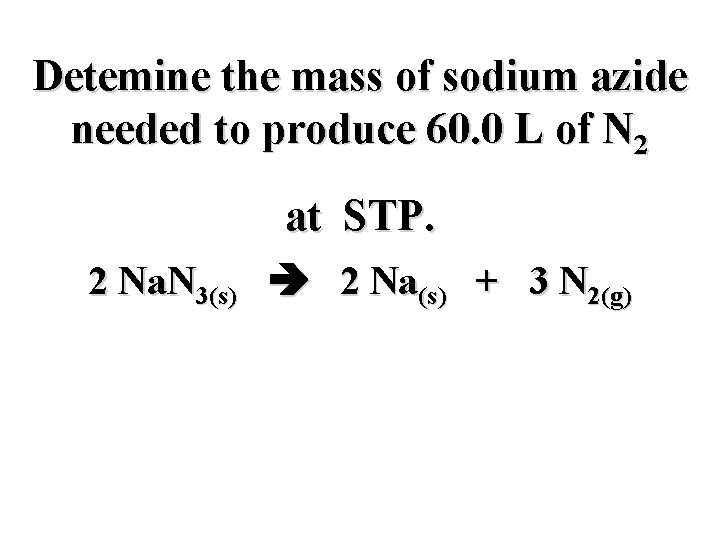
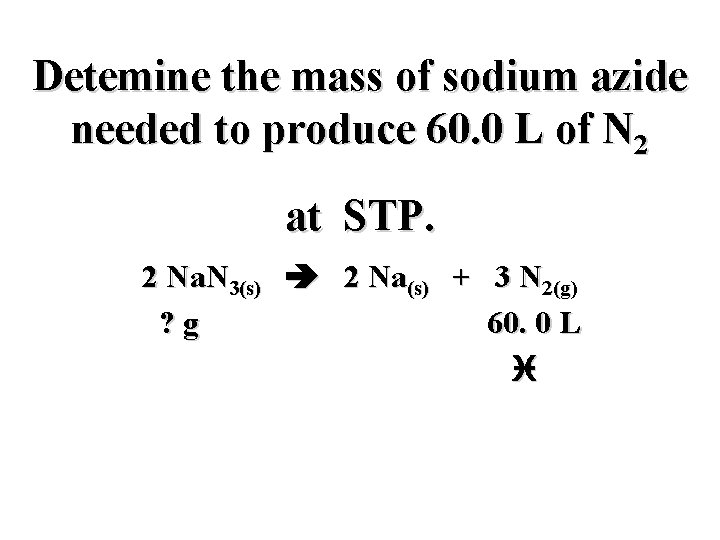
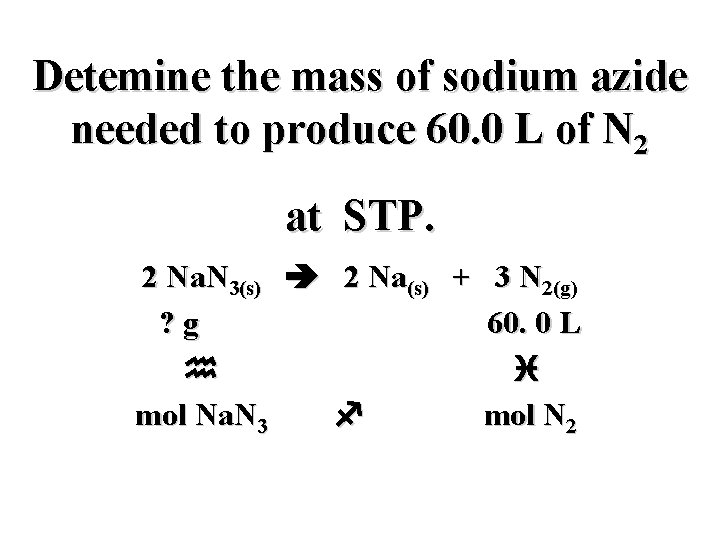
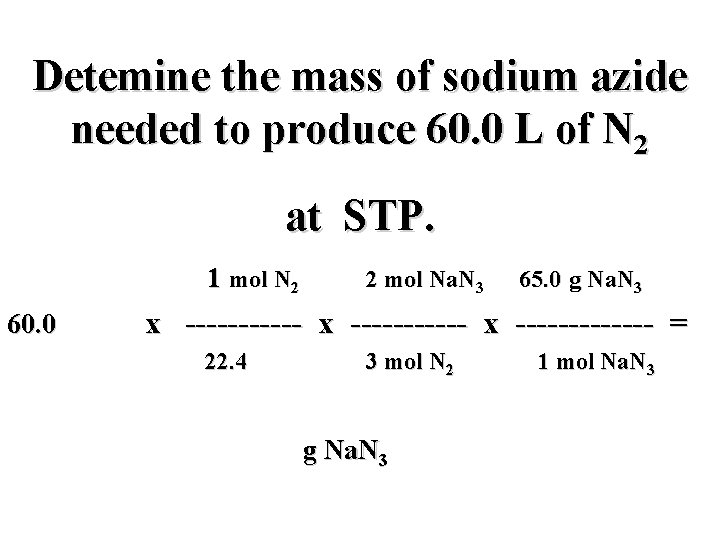
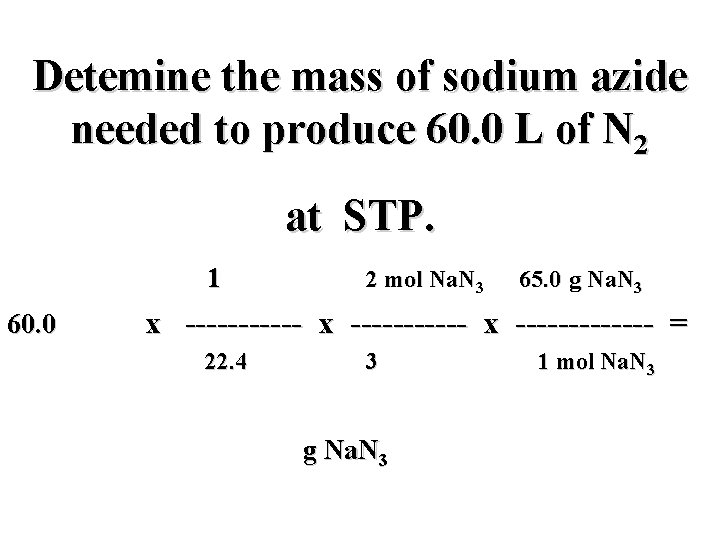
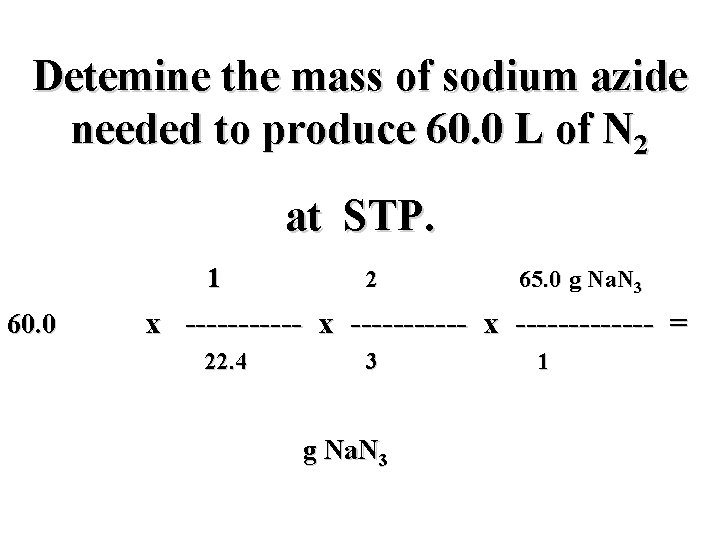
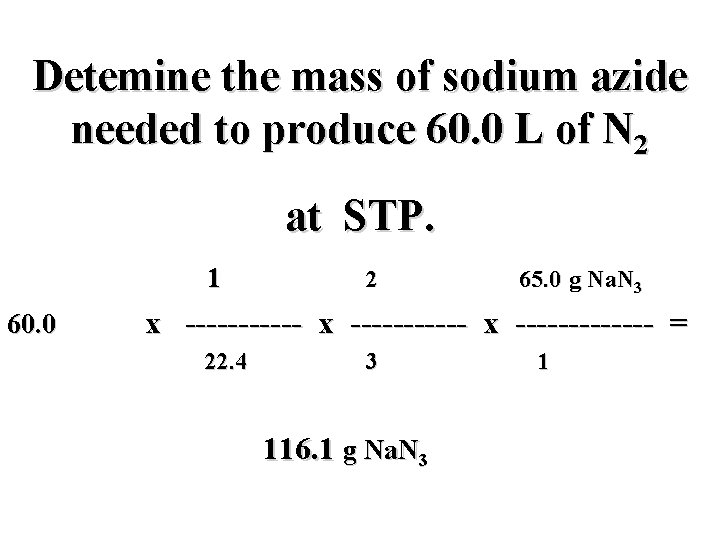
Detemine the mass of sodium azide needed to produce 60. 0 L of N 2 at STP. 2 Na. N 3(s) 2 Na(s) + 3 N 2(g)

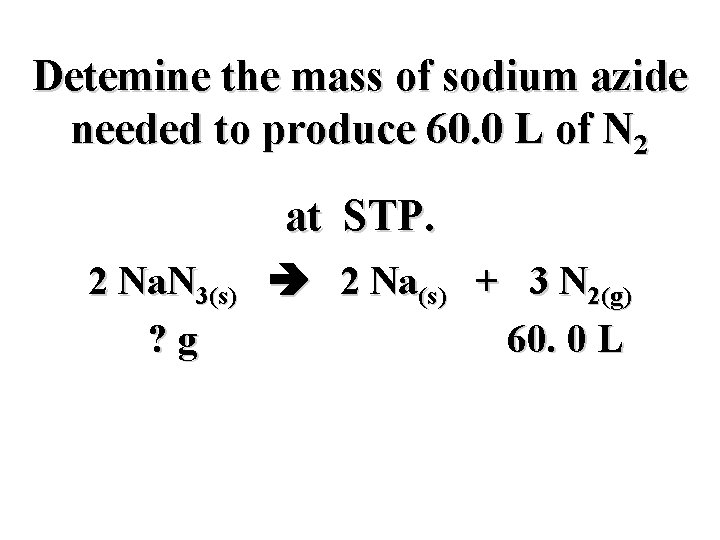
Detemine the mass of sodium azide needed to produce 60. 0 L of N 2 at STP. 2 Na. N 3(s) 2 Na(s) + 3 N 2(g) ? g 60. 0 L

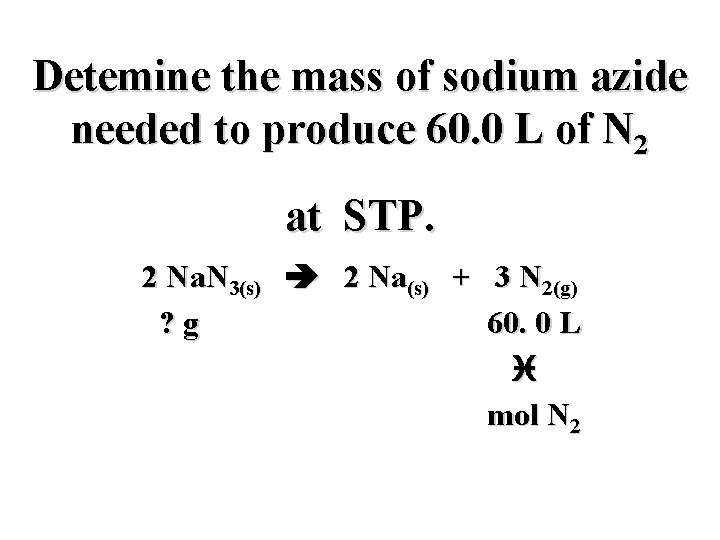
Detemine the mass of sodium azide needed to produce 60. 0 L of N 2 at STP. 2 Na. N 3(s) 2 Na(s) + 3 N 2(g) ? g 60. 0 L i

Detemine the mass of sodium azide needed to produce 60. 0 L of N 2 at STP. 2 Na. N 3(s) 2 Na(s) + 3 N 2(g) ? g 60. 0 L i mol N 2

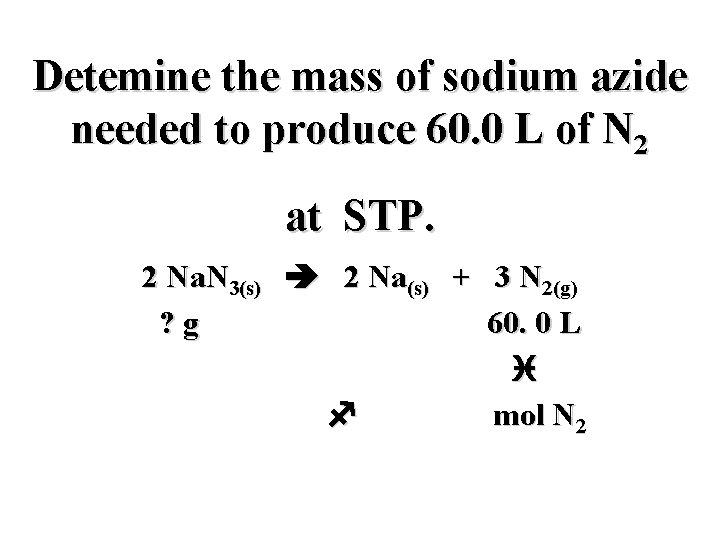
Detemine the mass of sodium azide needed to produce 60. 0 L of N 2 at STP. 2 Na. N 3(s) 2 Na(s) + 3 N 2(g) ? g 60. 0 L i f mol N 2

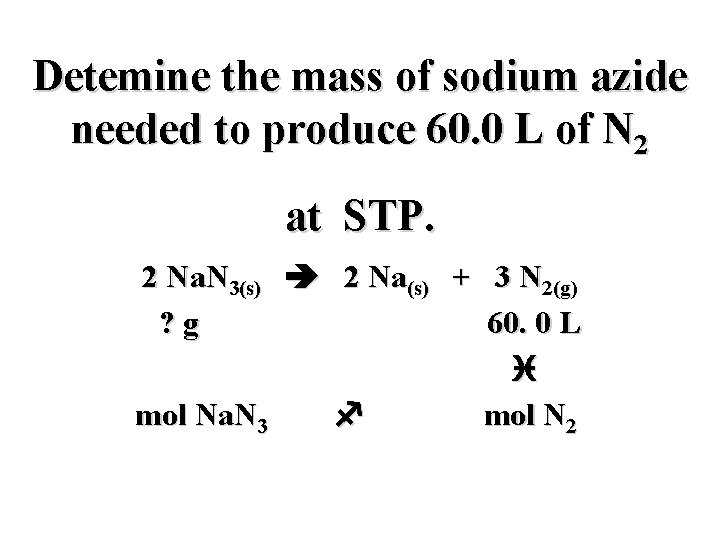
Detemine the mass of sodium azide needed to produce 60. 0 L of N 2 at STP. 2 Na. N 3(s) 2 Na(s) + 3 N 2(g) ? g 60. 0 L i mol Na. N 3 f mol N 2

Detemine the mass of sodium azide needed to produce 60. 0 L of N 2 at STP. 2 Na. N 3(s) 2 Na(s) + 3 N 2(g) ? g 60. 0 L h i mol Na. N 3 f mol N 2

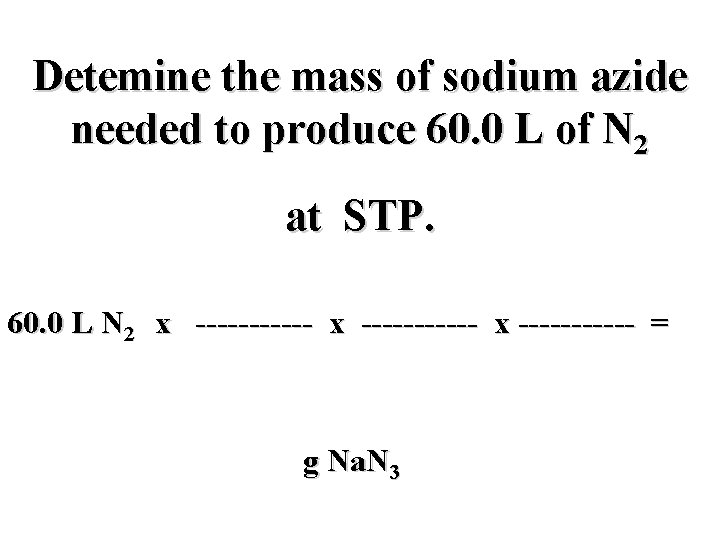
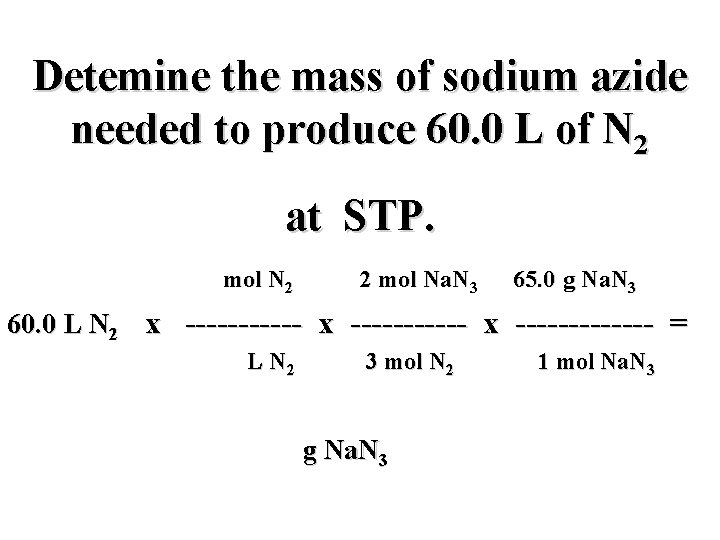
Detemine the mass of sodium azide needed to produce 60. 0 L of N 2 at STP. 60. 0 L N 2 x ----------- x ------ = g Na. N 3

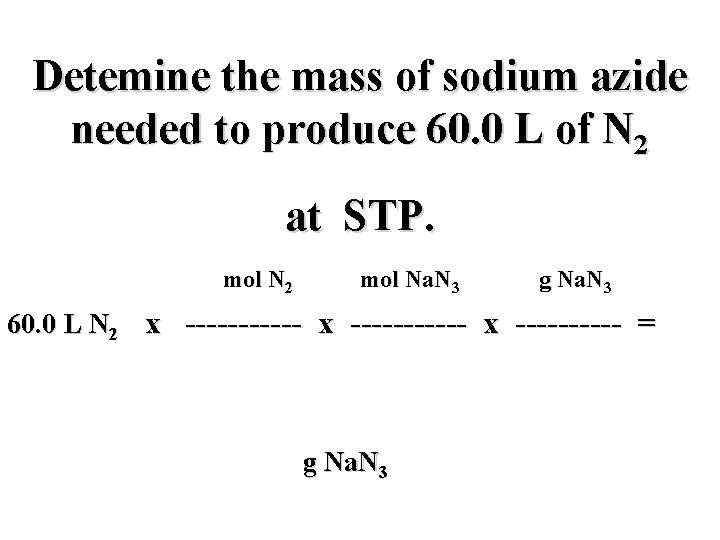
Detemine the mass of sodium azide needed to produce 60. 0 L of N 2 at STP. mol N 2 mol Na. N 3 g Na. N 3 60. 0 L N 2 x ----------- x ----- = g Na. N 3

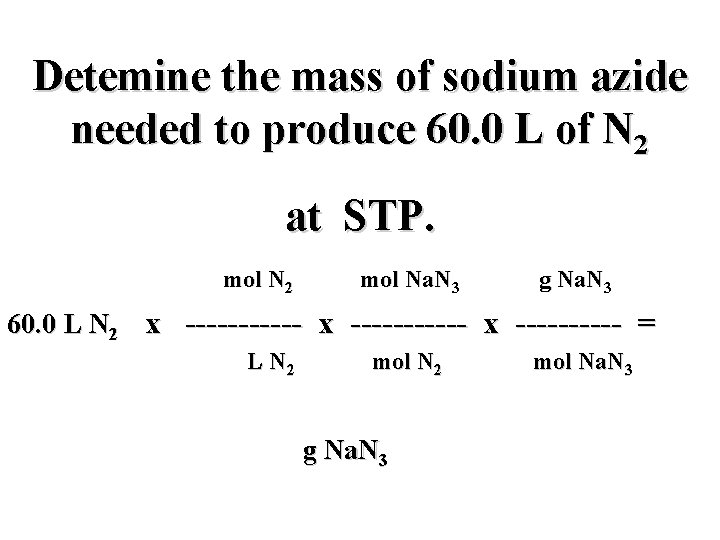
Detemine the mass of sodium azide needed to produce 60. 0 L of N 2 at STP. mol N 2 mol Na. N 3 g Na. N 3 60. 0 L N 2 x ----------- x ----- = L N 2 mol N 2 g Na. N 3 mol Na. N 3

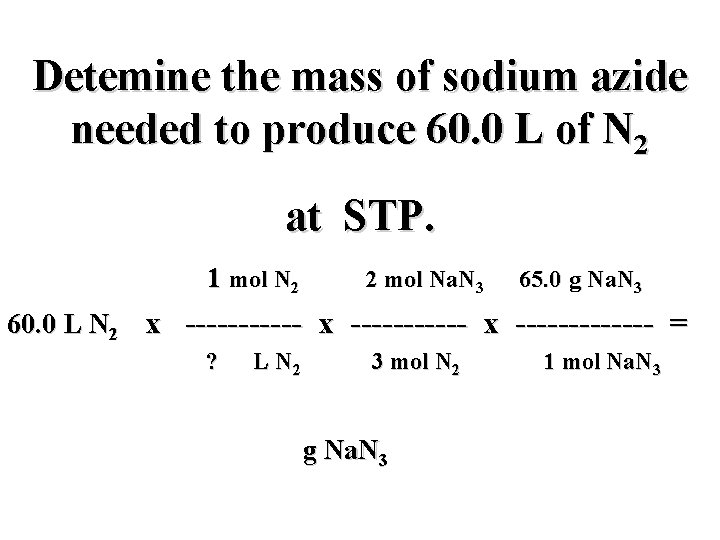
Detemine the mass of sodium azide needed to produce 60. 0 L of N 2 at STP. mol N 2 2 mol Na. N 3 65. 0 g Na. N 3 60. 0 L N 2 x ----------- x ------- = L N 2 3 mol N 2 g Na. N 3 1 mol Na. N 3

Detemine the mass of sodium azide needed to produce 60. 0 L of N 2 at STP. 1 mol N 2 2 mol Na. N 3 65. 0 g Na. N 3 60. 0 L N 2 x ----------- x ------- = ? L N 2 3 mol N 2 g Na. N 3 1 mol Na. N 3

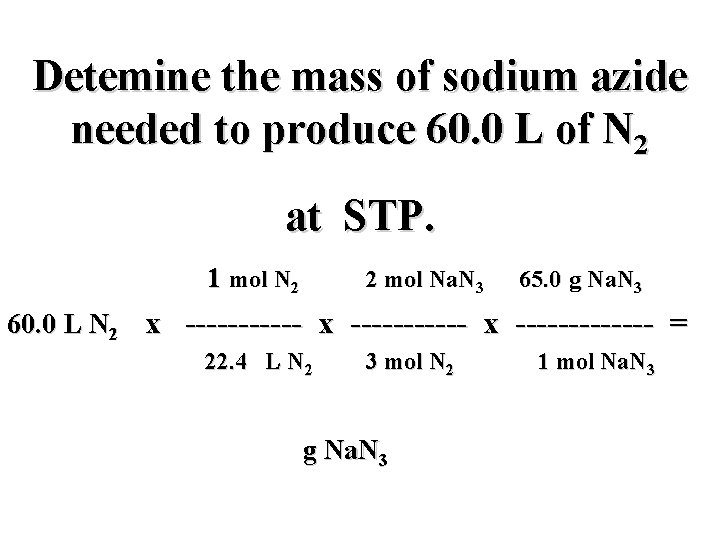
Detemine the mass of sodium azide needed to produce 60. 0 L of N 2 at STP. 1 mol N 2 2 mol Na. N 3 65. 0 g Na. N 3 60. 0 L N 2 x ----------- x ------- = 22. 4 L N 2 3 mol N 2 g Na. N 3 1 mol Na. N 3

Detemine the mass of sodium azide needed to produce 60. 0 L of N 2 at STP. 1 mol N 2 2 mol Na. N 3 65. 0 g Na. N 3 60. 0 L N 2 x ----------- x ------- = 22. 4 L N 2 3 mol N 2 g Na. N 3 1 mol Na. N 3

Detemine the mass of sodium azide needed to produce 60. 0 L of N 2 at STP. 1 mol N 2 2 mol Na. N 3 65. 0 g Na. N 3 60. 0 L N 2 x ----------- x ------- = 22. 4 L N 2 3 mol N 2 g Na. N 3 1 mol Na. N 3

Detemine the mass of sodium azide needed to produce 60. 0 L of N 2 at STP. 1 mol N 2 2 mol Na. N 3 65. 0 g Na. N 3 60. 0 L N 2 x ----------- x ------- = 22. 4 L N 2 3 mol N 2 g Na. N 3 1 mol Na. N 3

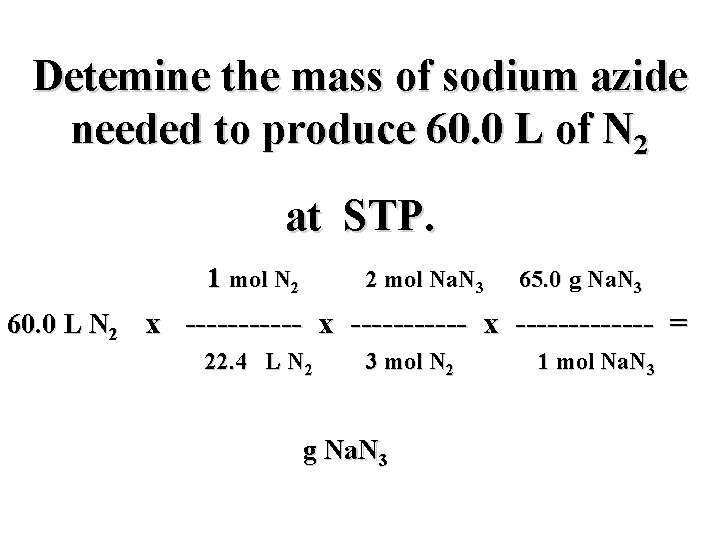
Detemine the mass of sodium azide needed to produce 60. 0 L of N 2 at STP. 1 mol N 2 2 mol Na. N 3 65. 0 g Na. N 3 60. 0 L N 2 x ----------- x ------- = 22. 4 L N 2 3 mol N 2 g Na. N 3 1 mol Na. N 3

Detemine the mass of sodium azide needed to produce 60. 0 L of N 2 at STP. 1 mol N 2 2 mol Na. N 3 65. 0 g Na. N 3 60. 0 L N 2 x ----------- x ------- = 22. 4 L N 2 3 mol N 2 116. 1 g Na. N 3 1 mol Na. N 3

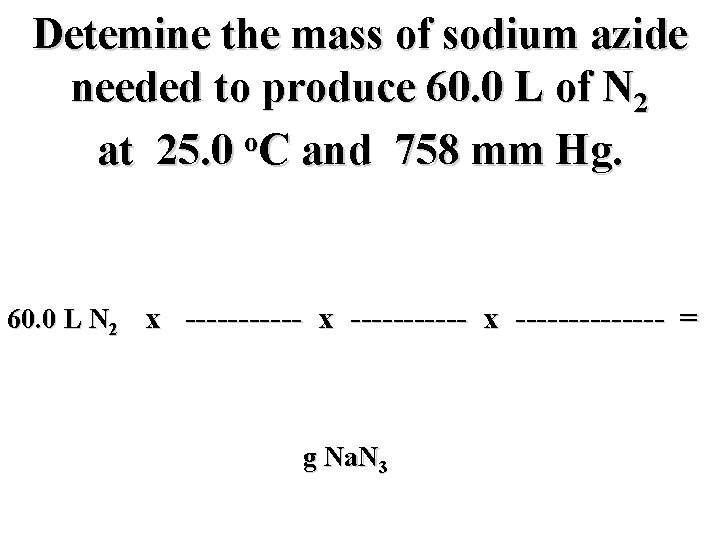
Detemine the mass of sodium azide needed to produce 60. 0 L of N 2 o at 25. 0 C and 758 mm Hg.

Detemine the mass of sodium azide needed to produce 60. 0 L of N 2 o at 25. 0 C and 758 mm Hg. 60. 0 L N 2 x ----------- x ------- = g Na. N 3

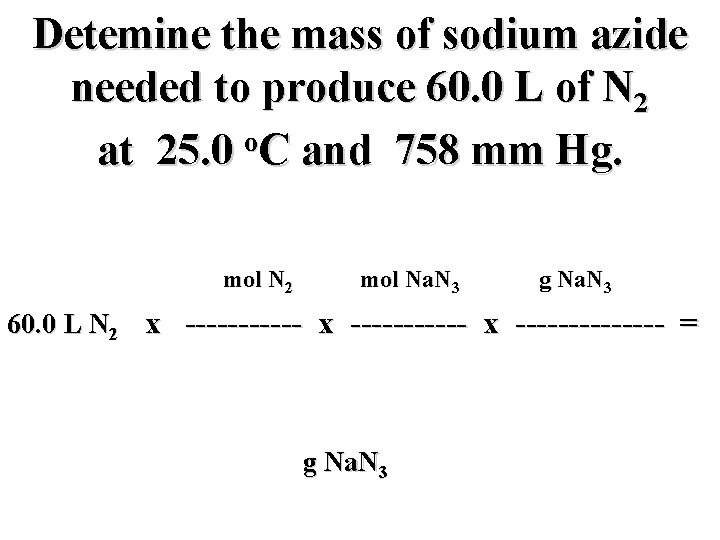
Detemine the mass of sodium azide needed to produce 60. 0 L of N 2 o at 25. 0 C and 758 mm Hg. mol N 2 mol Na. N 3 g Na. N 3 60. 0 L N 2 x ----------- x ------- = g Na. N 3

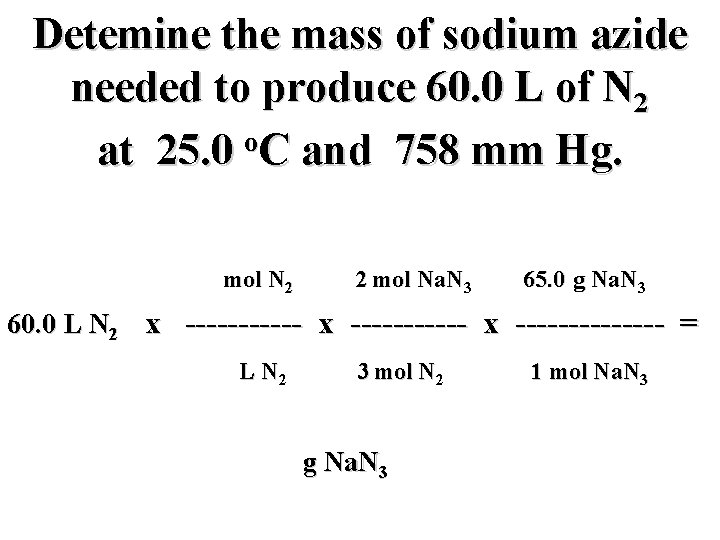
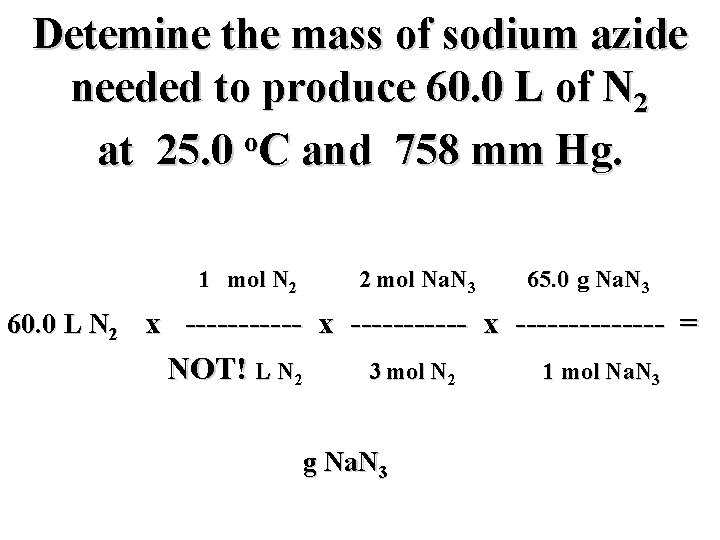
Detemine the mass of sodium azide needed to produce 60. 0 L of N 2 o at 25. 0 C and 758 mm Hg. mol N 2 2 mol Na. N 3 65. 0 g Na. N 3 60. 0 L N 2 x ----------- x ------- = L N 2 3 mol N 2 g Na. N 3 1 mol Na. N 3

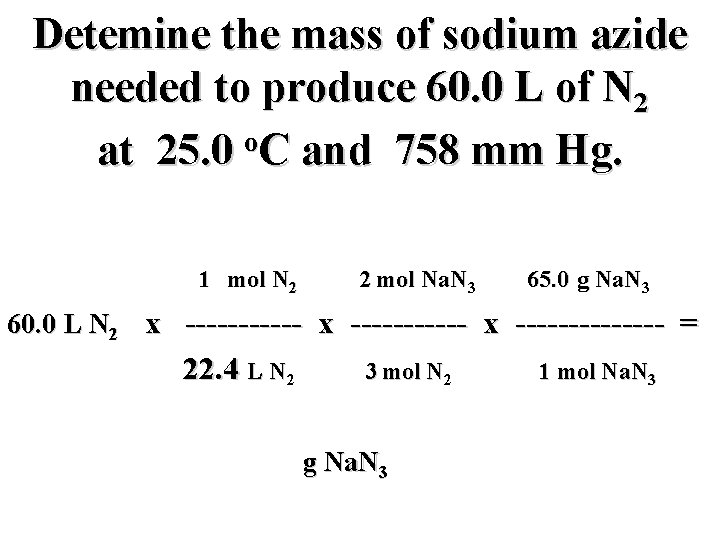
Detemine the mass of sodium azide needed to produce 60. 0 L of N 2 o at 25. 0 C and 758 mm Hg. 1 mol N 2 2 mol Na. N 3 65. 0 g Na. N 3 60. 0 L N 2 x ----------- x ------- = 22. 4 L N 2 3 mol N 2 g Na. N 3 1 mol Na. N 3

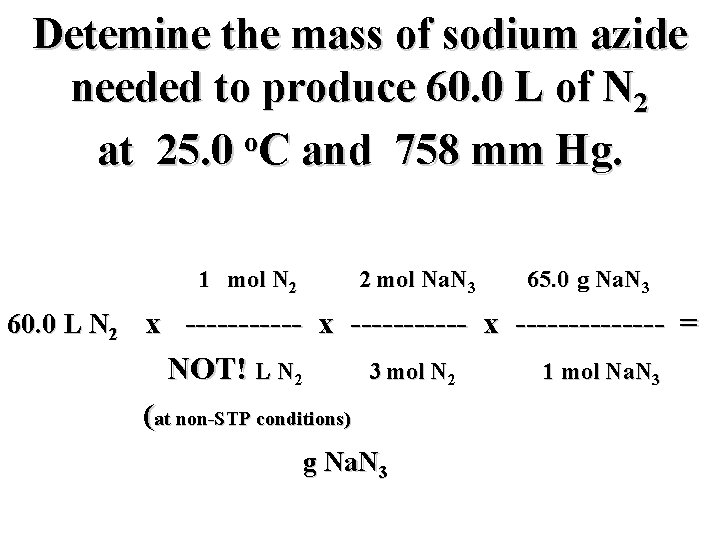
Detemine the mass of sodium azide needed to produce 60. 0 L of N 2 o at 25. 0 C and 758 mm Hg. 1 mol N 2 2 mol Na. N 3 65. 0 g Na. N 3 60. 0 L N 2 x ----------- x ------- = NOT! L N 2 3 mol N 2 g Na. N 3 1 mol Na. N 3

Detemine the mass of sodium azide needed to produce 60. 0 L of N 2 o at 25. 0 C and 758 mm Hg. 1 mol N 2 2 mol Na. N 3 65. 0 g Na. N 3 60. 0 L N 2 x ----------- x ------- = NOT! L N 2 3 mol N 2 (at non-STP conditions) g Na. N 3 1 mol Na. N 3

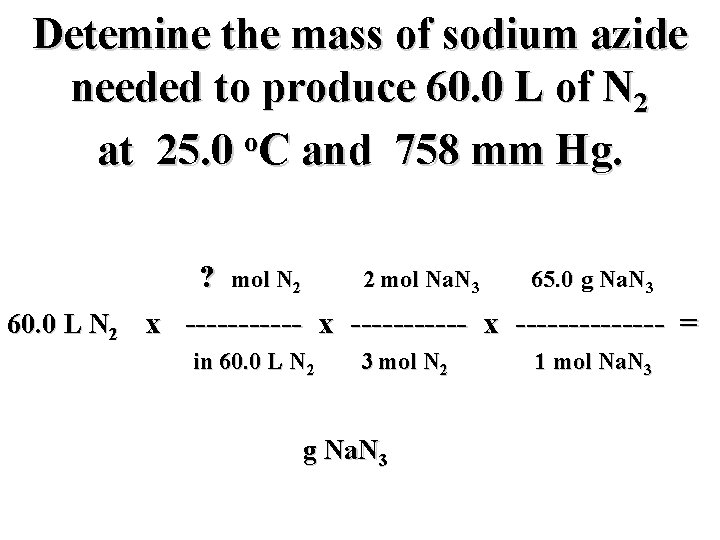
Detemine the mass of sodium azide needed to produce 60. 0 L of N 2 o at 25. 0 C and 758 mm Hg. 60. 0 L N 2 ? mol N 2 2 mol Na. N 3 65. 0 g Na. N 3 x ----------- x ------- = in 60. 0 L N 2 3 mol N 2 g Na. N 3 1 mol Na. N 3

Detemine the mass of sodium azide needed to produce 60. 0 L of N 2 o at 25. 0 C and 758 mm Hg.

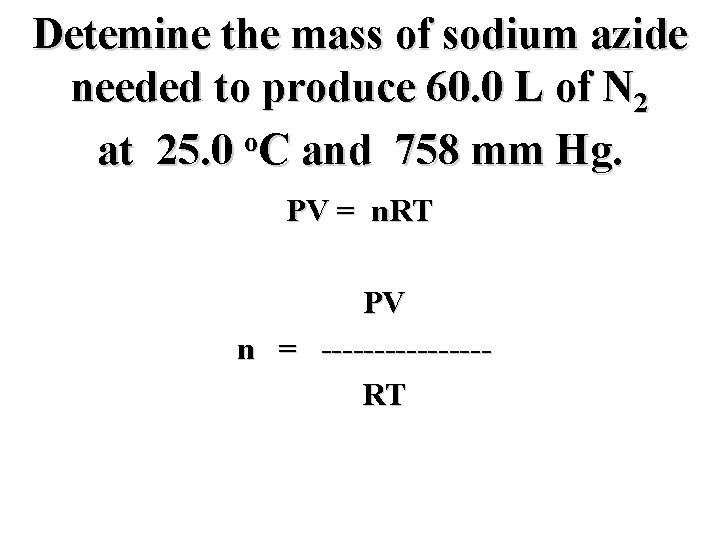
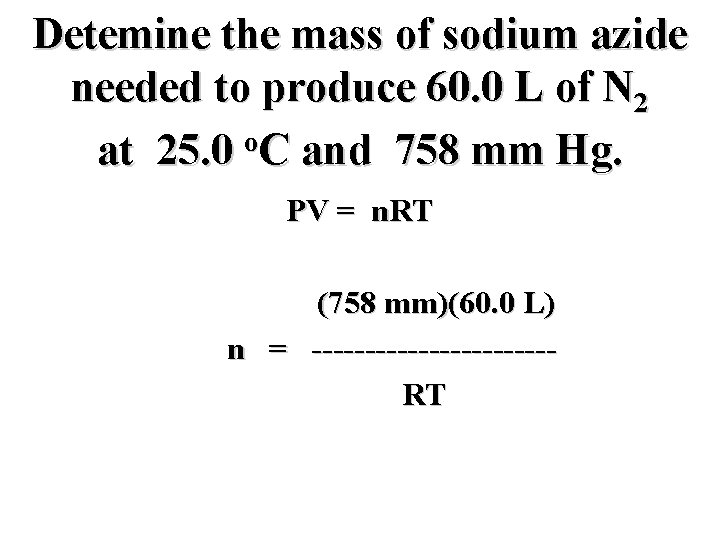
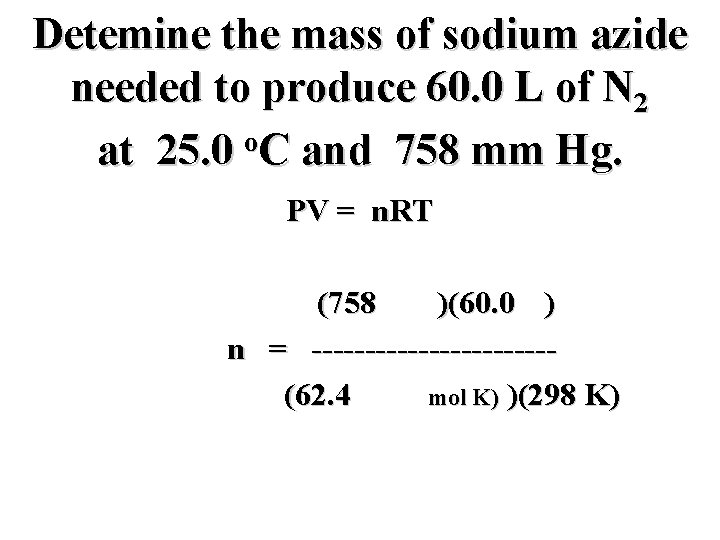
Detemine the mass of sodium azide needed to produce 60. 0 L of N 2 o at 25. 0 C and 758 mm Hg. PV = n. RT

Detemine the mass of sodium azide needed to produce 60. 0 L of N 2 o at 25. 0 C and 758 mm Hg. PV = n. RT n = --------

Detemine the mass of sodium azide needed to produce 60. 0 L of N 2 o at 25. 0 C and 758 mm Hg. PV = n. RT PV n = --------RT

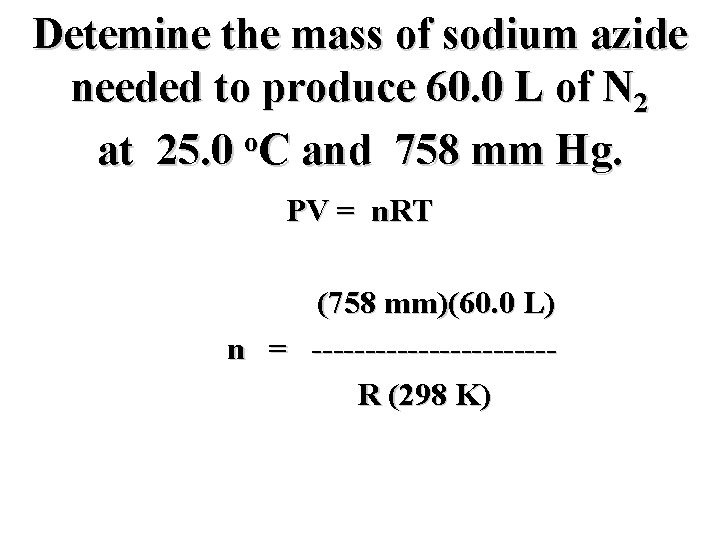
Detemine the mass of sodium azide needed to produce 60. 0 L of N 2 o at 25. 0 C and 758 mm Hg. PV = n. RT (758 mm)(60. 0 L) n = -----------RT

Detemine the mass of sodium azide needed to produce 60. 0 L of N 2 o at 25. 0 C and 758 mm Hg. PV = n. RT (758 mm)(60. 0 L) n = -----------R (298 K)

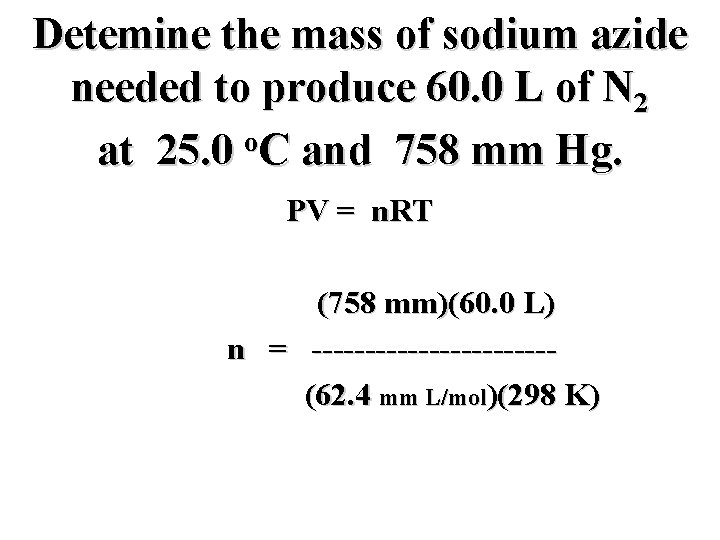
Detemine the mass of sodium azide needed to produce 60. 0 L of N 2 o at 25. 0 C and 758 mm Hg. PV = n. RT (758 mm)(60. 0 L) n = -----------(62. 4 mm L/mol)(298 K)

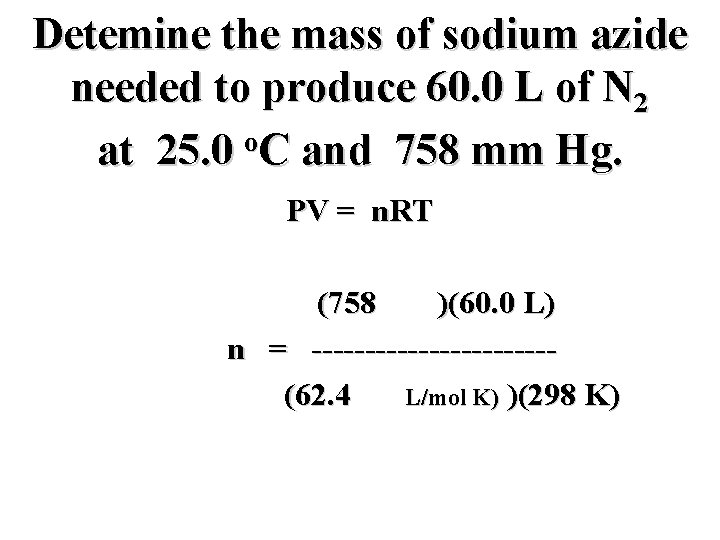
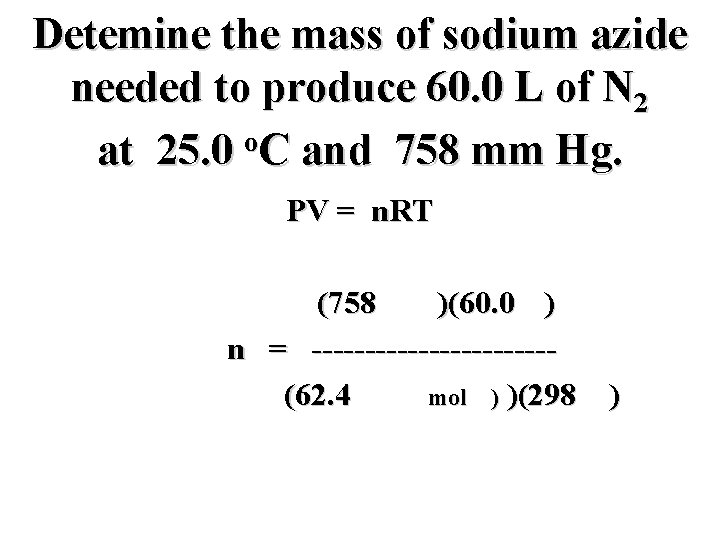
Detemine the mass of sodium azide needed to produce 60. 0 L of N 2 o at 25. 0 C and 758 mm Hg. PV = n. RT (758 mm)(60. 0 L) n = -----------(62. 4 mm L/mol K) )(298 K)

Detemine the mass of sodium azide needed to produce 60. 0 L of N 2 o at 25. 0 C and 758 mm Hg. PV = n. RT (758 mm)(60. 0 L) n = -----------(62. 4 mm L/mol K) )(298 K)

Detemine the mass of sodium azide needed to produce 60. 0 L of N 2 o at 25. 0 C and 758 mm Hg. PV = n. RT (758 mm)(60. 0 L) n = -----------(62. 4 mm L/mol K) )(298 K)

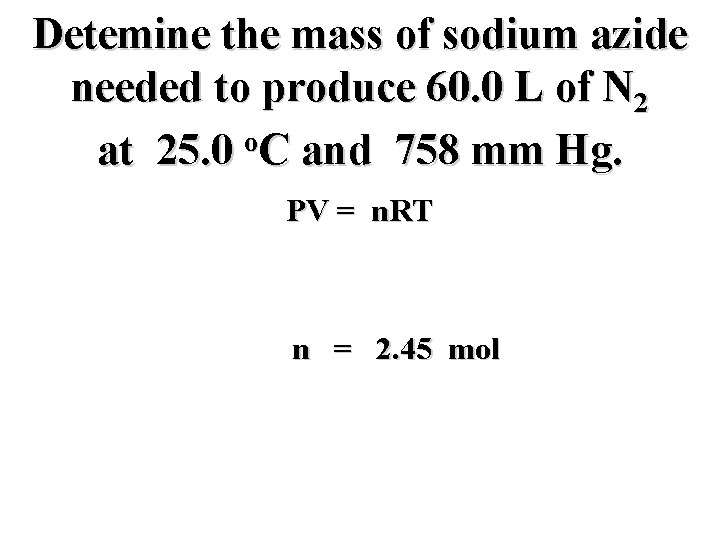
Detemine the mass of sodium azide needed to produce 60. 0 L of N 2 o at 25. 0 C and 758 mm Hg. PV = n. RT n = 2. 45 mol

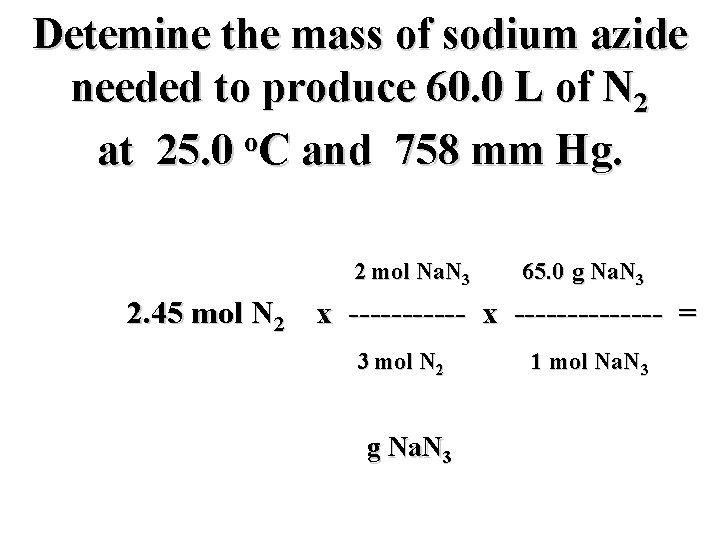
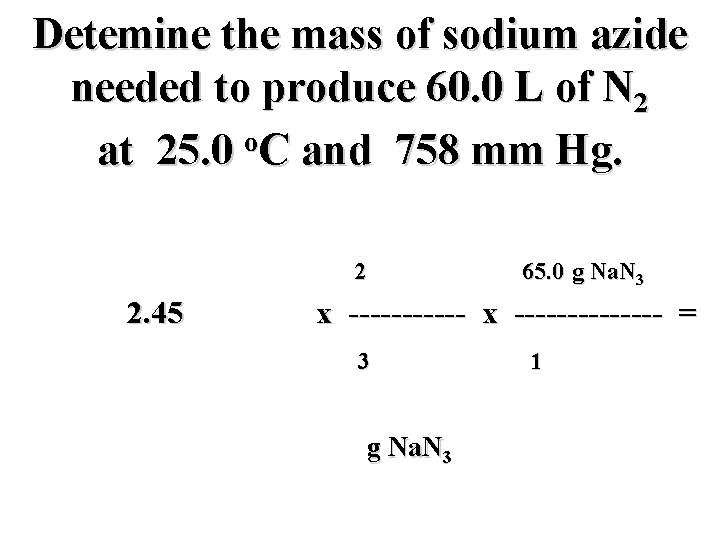
Detemine the mass of sodium azide needed to produce 60. 0 L of N 2 o at 25. 0 C and 758 mm Hg. 2 mol Na. N 3 65. 0 g Na. N 3 2. 45 mol N 2 x -------------- = 3 mol N 2 g Na. N 3 1 mol Na. N 3
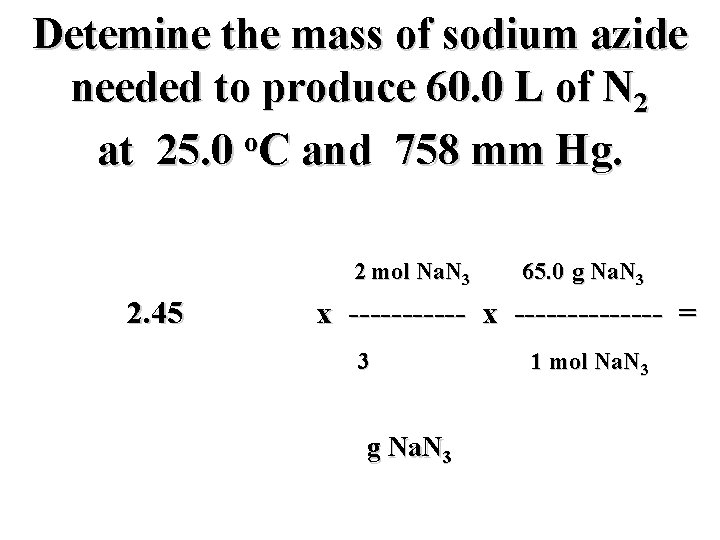
Detemine the mass of sodium azide needed to produce 60. 0 L of N 2 o at 25. 0 C and 758 mm Hg. 2 mol Na. N 3 65. 0 g Na. N 3 2. 45 mol N 2 x -------------- = 3 mol N 2 g Na. N 3 1 mol Na. N 3

Detemine the mass of sodium azide needed to produce 60. 0 L of N 2 o at 25. 0 C and 758 mm Hg. 2 mol Na. N 3 65. 0 g Na. N 3 2. 45 mol N 2 x -------------- = 3 mol N 2 g Na. N 3 1 mol Na. N 3

Detemine the mass of sodium azide needed to produce 60. 0 L of N 2 o at 25. 0 C and 758 mm Hg. 2 mol Na. N 3 65. 0 g Na. N 3 2. 45 mol N 2 x -------------- = 3 mol N 2 106. 2 g Na. N 3 1 mol Na. N 3

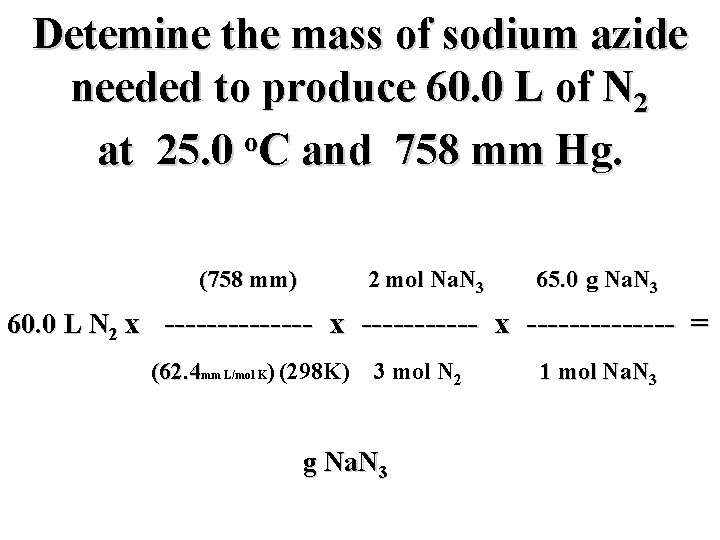
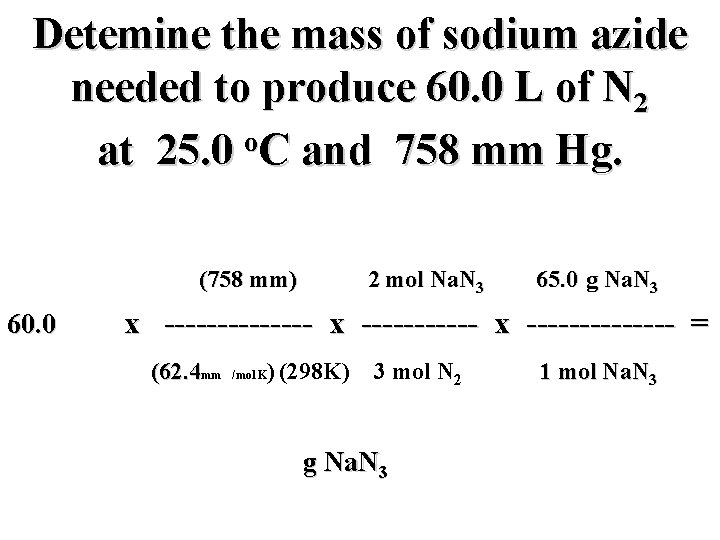
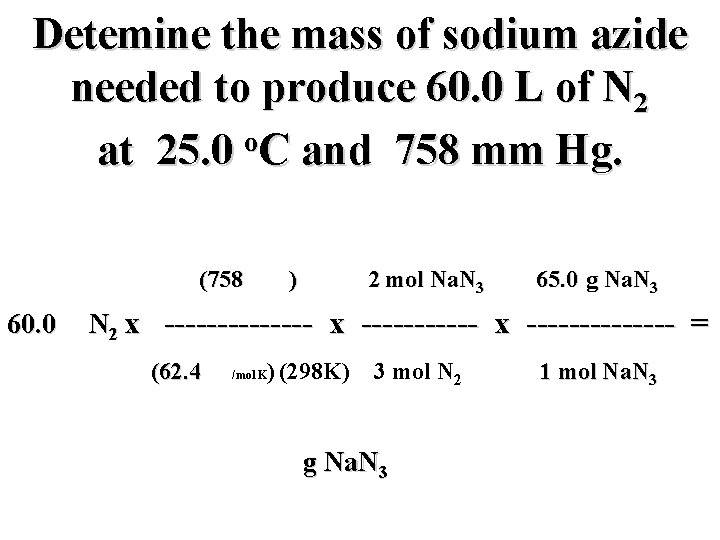
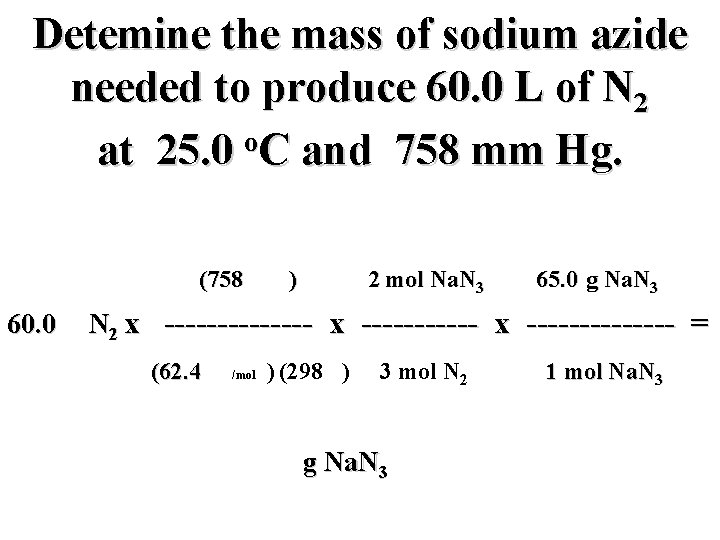
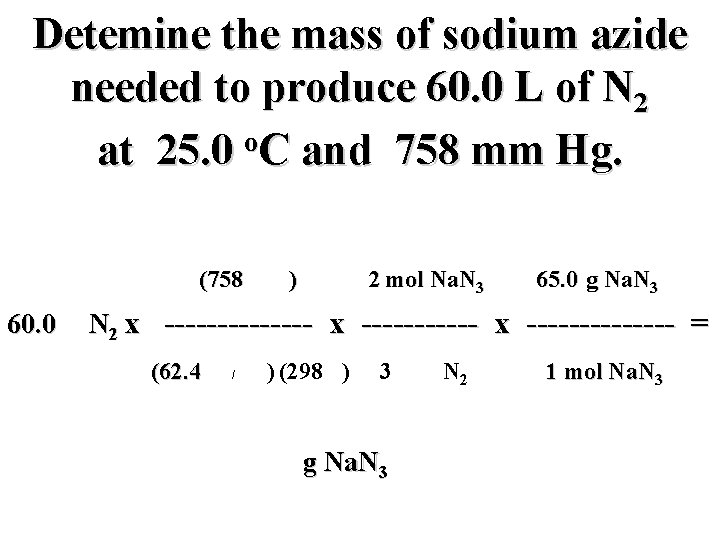
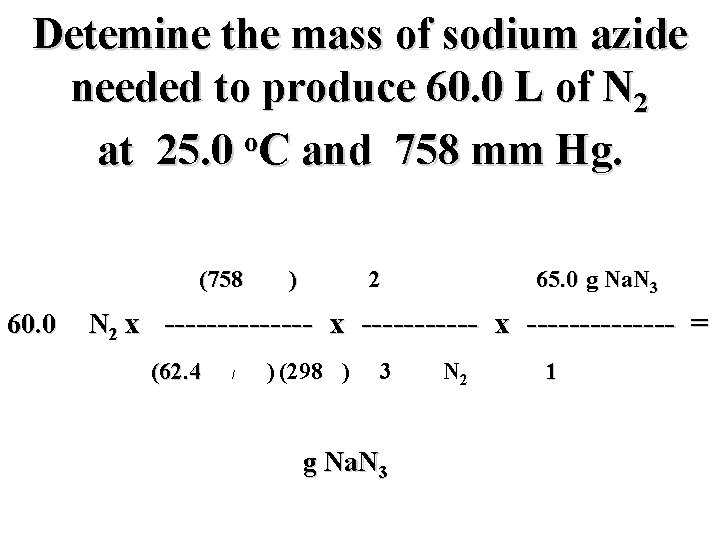
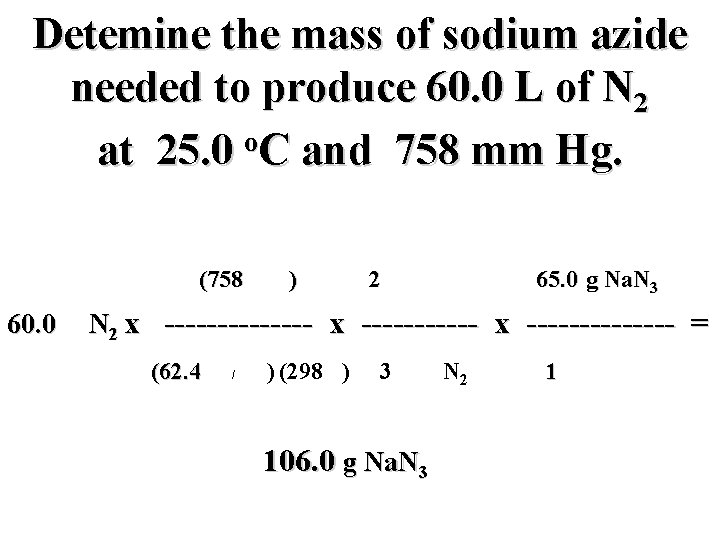
Detemine the mass of sodium azide needed to produce 60. 0 L of N 2 o at 25. 0 C and 758 mm Hg. (758 mm) 2 mol Na. N 3 65. 0 g Na. N 3 60. 0 L N 2 x -------------- = (62. 4 mm L/mol K) (298 K) 3 mol N 2 g Na. N 3 1 mol Na. N 3

Detemine the mass of sodium azide needed to produce 60. 0 L of N 2 o at 25. 0 C and 758 mm Hg. (758 mm) 2 mol Na. N 3 65. 0 g Na. N 3 60. 0 L N 2 x -------------- = (62. 4 mm L/mol K) (298 K) 3 mol N 2 g Na. N 3 1 mol Na. N 3

Detemine the mass of sodium azide needed to produce 60. 0 L of N 2 o at 25. 0 C and 758 mm Hg. (758 mm) 2 mol Na. N 3 65. 0 g Na. N 3 60. 0 L N 2 x -------------- = (62. 4 mm L/mol K) (298 K) 3 mol N 2 g Na. N 3 1 mol Na. N 3

Detemine the mass of sodium azide needed to produce 60. 0 L of N 2 o at 25. 0 C and 758 mm Hg. (758 mm) 2 mol Na. N 3 65. 0 g Na. N 3 60. 0 L N 2 x -------------- = (62. 4 mm L/mol K) (298 K) 3 mol N 2 g Na. N 3 1 mol Na. N 3

Detemine the mass of sodium azide needed to produce 60. 0 L of N 2 o at 25. 0 C and 758 mm Hg. (758 mm) 2 mol Na. N 3 65. 0 g Na. N 3 60. 0 L N 2 x -------------- = (62. 4 mm L/mol K) (298 K) 3 mol N 2 g Na. N 3 1 mol Na. N 3

Detemine the mass of sodium azide needed to produce 60. 0 L of N 2 o at 25. 0 C and 758 mm Hg. (758 mm) 2 mol Na. N 3 65. 0 g Na. N 3 60. 0 L N 2 x -------------- = (62. 4 mm L/mol K) (298 K) 3 mol N 2 g Na. N 3 1 mol Na. N 3

Detemine the mass of sodium azide needed to produce 60. 0 L of N 2 o at 25. 0 C and 758 mm Hg. (758 mm) 2 mol Na. N 3 65. 0 g Na. N 3 60. 0 L N 2 x -------------- = (62. 4 mm L/mol K) (298 K) 3 mol N 2 106. 0 g Na. N 3 1 mol Na. N 3