GAS LIQUID CHROMATOGRAPHY Principles Partition of molecules between

GAS LIQUID CHROMATOGRAPHY Principles Partition of molecules between gas (mobile phase) and liquid (stationary phase).

Most Common Stationary Phases 1. Separation of mixture of polar compounds Carbowax 20 M (polyethylene glycol) 2. Separation of mixtures of non-polar compounds OV 101 or SE-30 (polymer of methylsilicone) 3. Methylester of fatty acids DEGS (diethylene glycol succinate)
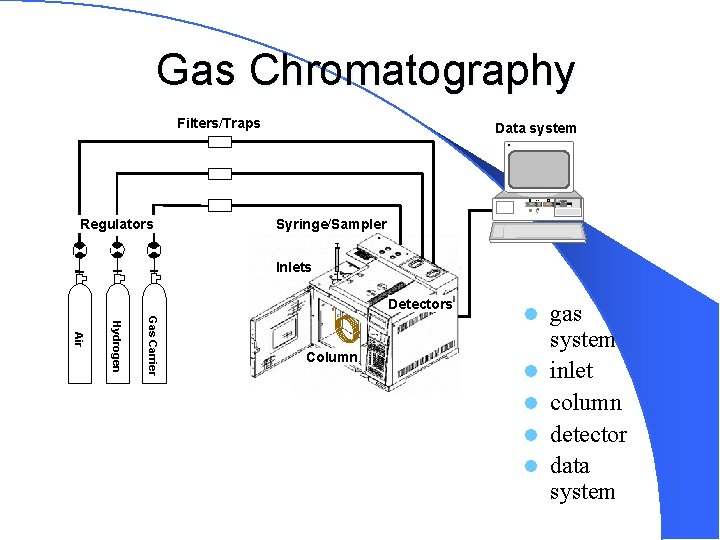
Gas Chromatography Filters/Traps Data system H RESET Regulators Syringe/Sampler Inlets Detectors Gas Carrier Hydrogen Air Column l l l gas system inlet column detector data system
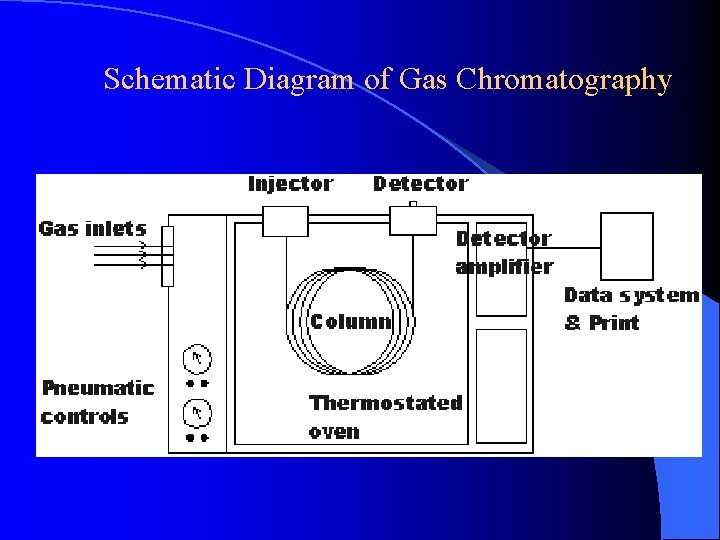
Schematic Diagram of Gas Chromatography
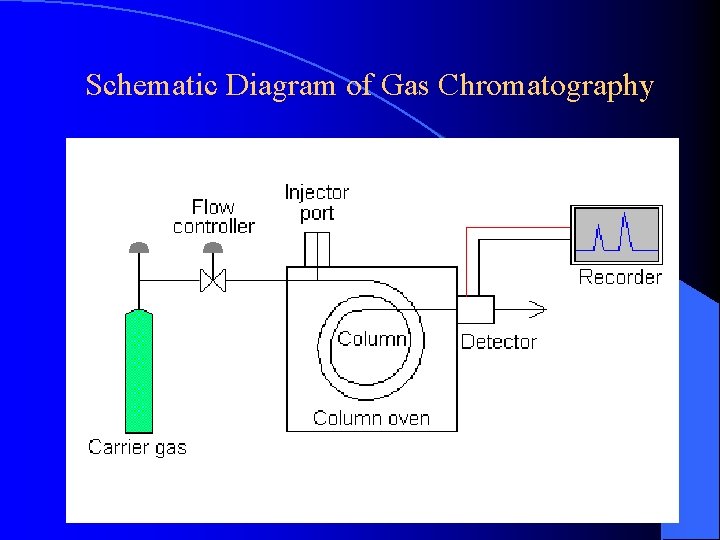
Schematic Diagram of Gas Chromatography

DETECTORS Flame Ionization Detector (Nanogram - ng) High temperature of hydrogen flame (H 2 +O 2 + N 2) ionizes compounds eluted from column into flame. The ions collected on collector or electrode and were recorded on recorder due to electric current.
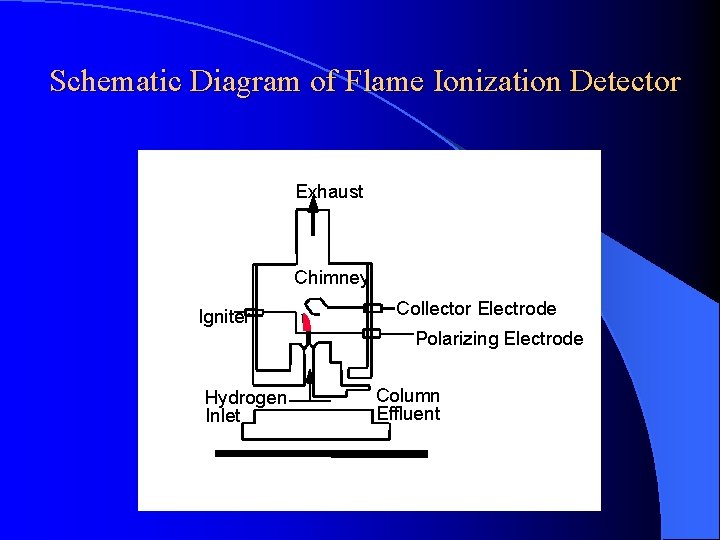
Schematic Diagram of Flame Ionization Detector Exhaust Chimney Igniter Collector Electrode Polarizing Electrode Hydrogen Inlet Column Effluent
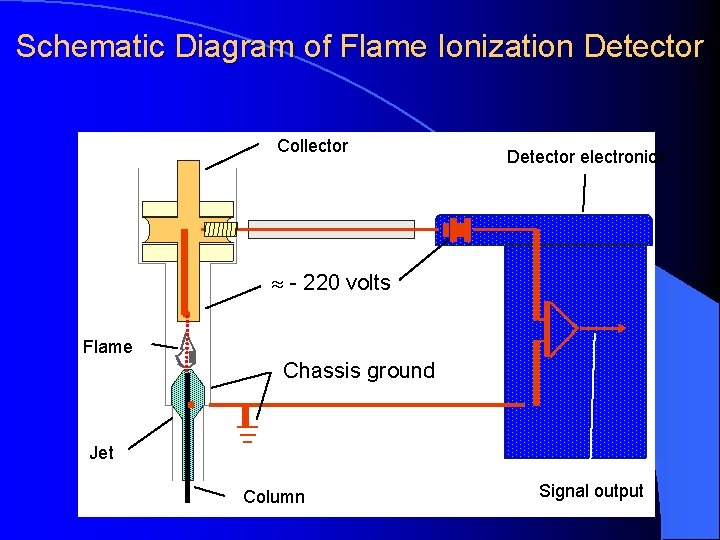
Schematic Diagram of Flame Ionization Detector Collector Detector electronics - 220 volts Flame Chassis ground Jet Column Signal output

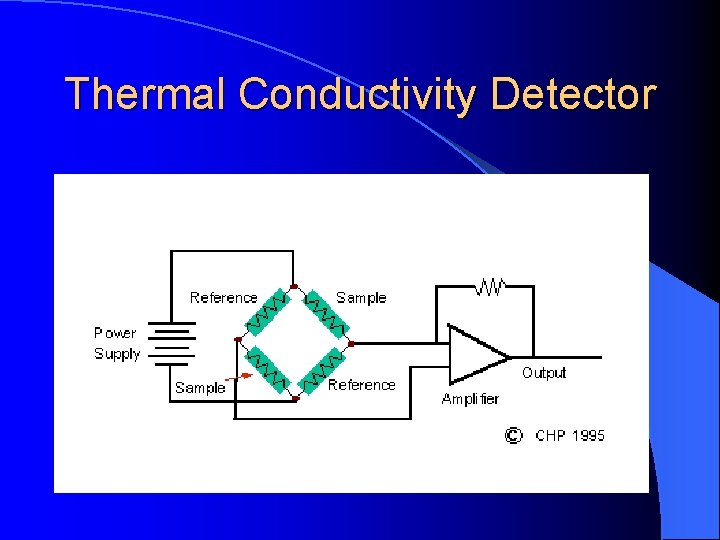
Thermal Conductivity Detector Measures the changes of thermal conductivity due to the sample (mg). Sample can be recovered.

Thermal Conductivity Detector Principal: The thermal balance of a heated filament Electrical power is converted to heat in a resistant filament and the temperature will climb until heat power loss form the filament equals the electrical power input. The filament may loose heat by radiation to a cooler surface and by conduction to the molecules coming into contact with it.
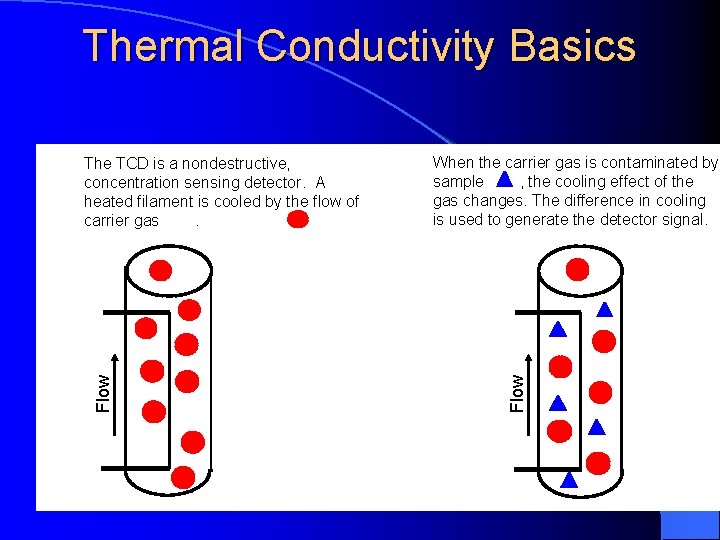
Thermal Conductivity Basics When the carrier gas is contaminated by sample , the cooling effect of the gas changes. The difference in cooling is used to generate the detector signal. Flow The TCD is a nondestructive, concentration sensing detector. A heated filament is cooled by the flow of carrier gas.

Thermal Conductivity Detector When a compound elutes, thermal conductivity of the gaseous mixture of carrier gas and compound gas is lowered, and the filament in the sample column becomes hotter than the other control column. Its resistance increased, and this imbalance between control and sample filament resistances is measured by a simple gadget and a signal is recorded

Thermal Conductivity Detector

Relative Thermal Conductivity Compound Carbon Tetrachloride Benzene Hexane Argon Methanol Nitrogen Helium Hydrogen Relative Thermal Conductivity 0. 05 0. 11 0. 12 0. 13 0. 17 1. 00 1. 28

Thermal Conductivity Detector • Responds to all compounds • Adequate sensitivity for many compounds • Good linear range of signal • Simple construction • Signal quite stable provided carrier gas glow rate, block temperature, and filament power are controlled • Nondestructive detection

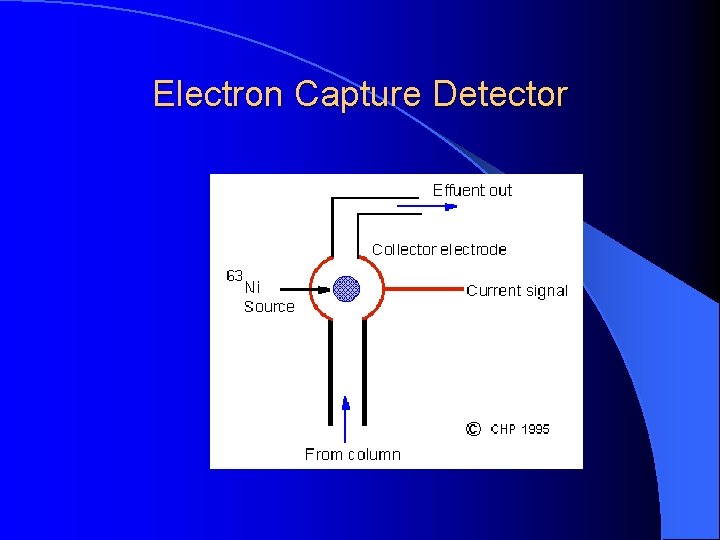
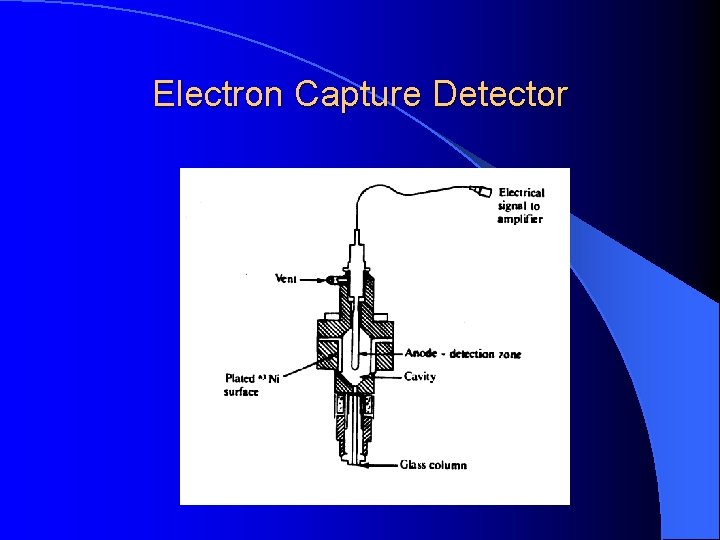
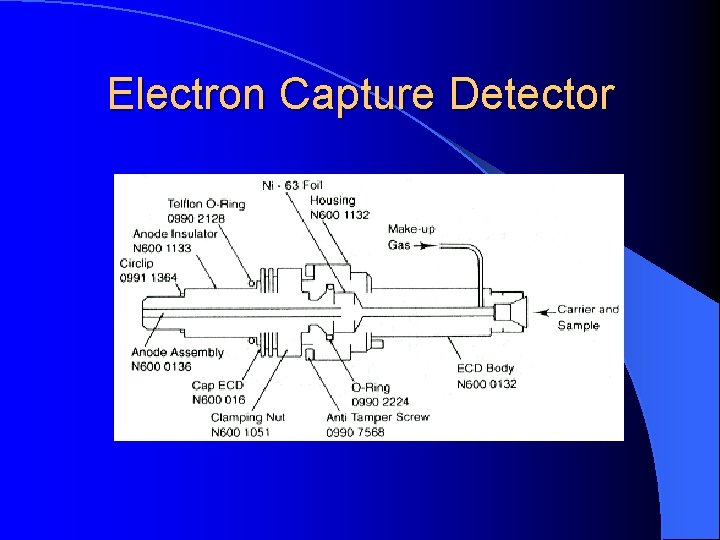
Electron Capture Detector For pesticide analysis (picogram). Accept electrons of carrier gas.

Electron Capture Detector ECD detects ions in the exiting from the gas chromatographic column by the anode electrode. 3 H or 63 Ni which emits particles. Ionization : N 2 (Nitrogen carrier gas) + (e) = N 2+ + 2 e These N 2+ establish a “base line” X (F, Cl and Br) containing sample + (e) XIon recombination : X- + N 2+ = X + N 2 The “base line” will decrease and this decrease constitutes the signal. Insecticides, pesticides, vinyl chloride, and fluorocarbons
Electron Capture Detector
Electron Capture Detector

Gas Chromatography Application
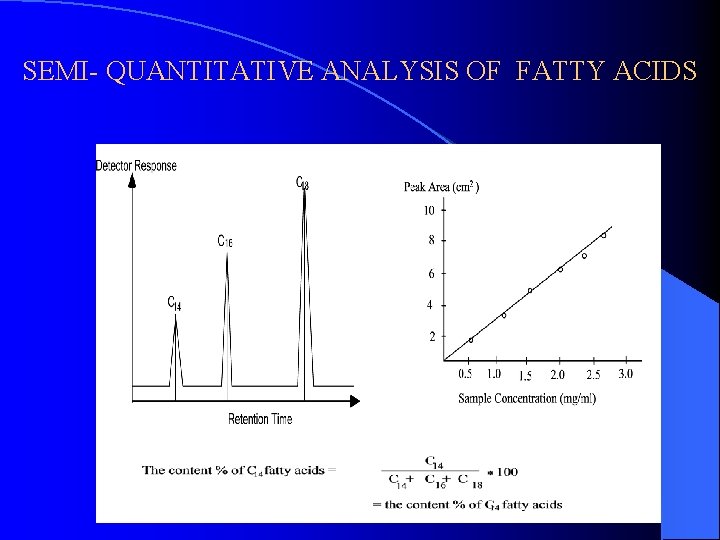
SEMI- QUANTITATIVE ANALYSIS OF FATTY ACIDS
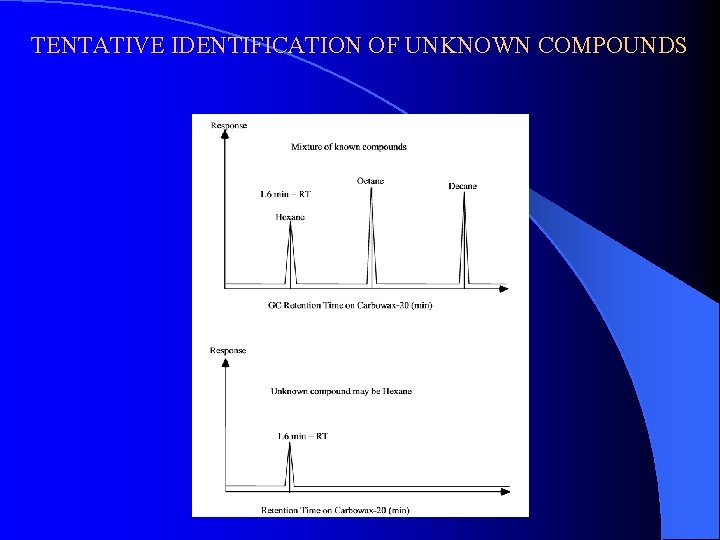
TENTATIVE IDENTIFICATION OF UNKNOWN COMPOUNDS
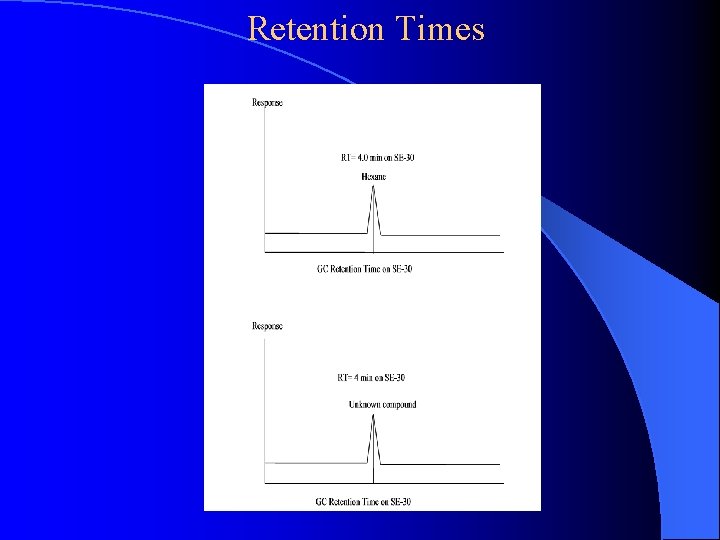
Retention Times

GLC ADVANTAGES 1. Very good separation 2. Time (analysis is short) 3. Small sample is needed - ml 4. Good detection system 5. Quantitatively analyzed

DISADVANTAGES OF GAS CHROMATOGRAPHY Material has to be volatilized at 250 C without decomposition.
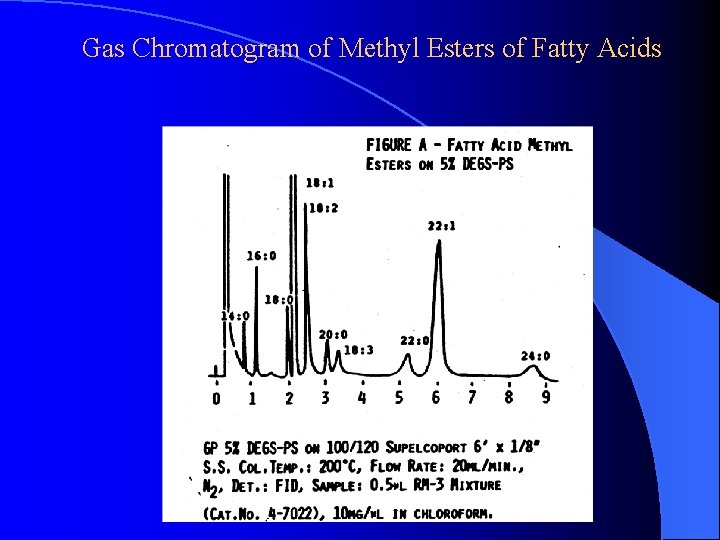
Gas Chromatogram of Methyl Esters of Fatty Acids
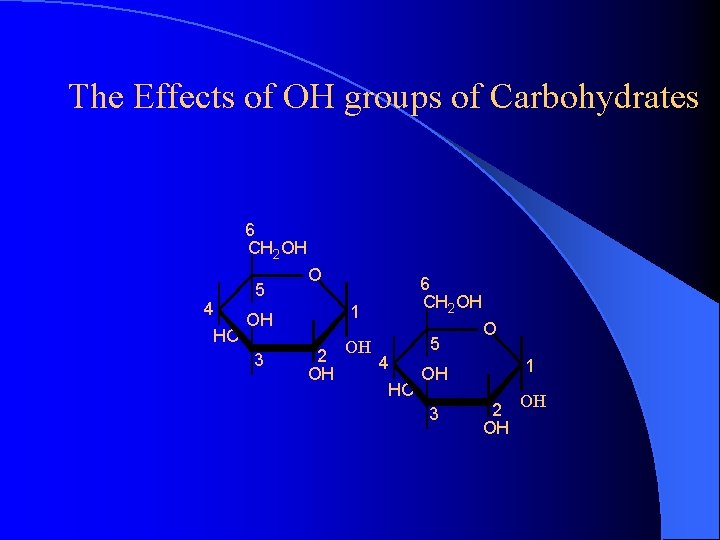
The Effects of OH groups of Carbohydrates 6 CH 2 OH 4 HO 5 O 1 OH 3 6 CH 2 OH OH 4 HO 5 O 1 OH 3 2 OH OH
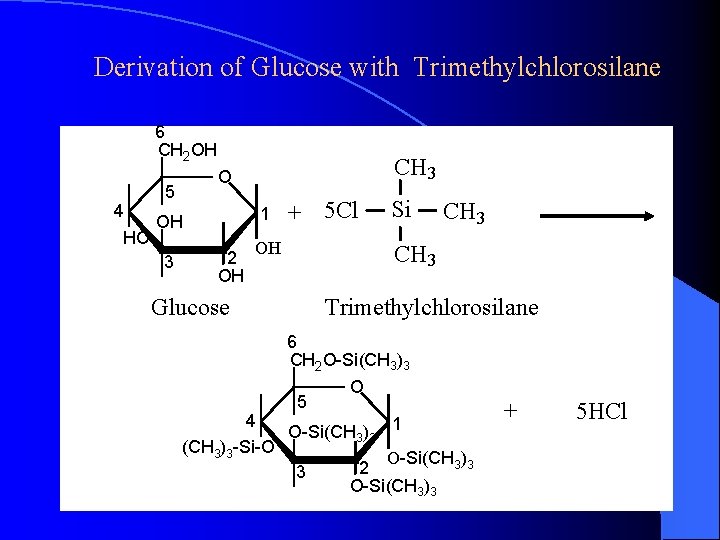
Derivation of Glucose with Trimethylchlorosilane 6 CH 2 OH 5 4 1 OH HO 3 CH 3 O 2 OH + 5 Cl OH 4 (CH 3)3 -Si-O CH 3 Glucose Si Trimethylchlorosilane 6 CH 2 O-Si(CH 3)3 O 5 O-Si(CH ) 1 3 3 3 O-Si(CH 3)3 2 O-Si(CH 3)3 + 5 HCl

Effects of Derivation 1. Time consumption 2. Side reaction 3. Loss of sample

THIN LAYER CHROMATOGRAPHY Stationary Phase -----> Silica Gel Mobile Phase -------> (developing) Solvent

THIN LAYER CHROMATOGRAPHY


Thermal Conductivity Detector The detector contains two filaments: one exposed only to carrier gas, while the other is exposed to the carrier gas for sample analysis. When the gas for the sample analysis is only carrier gas , the two filaments can be balanced. Instead of a direct measurement of filament temperature, the filament resistant, which is a function of temperature, is measured.

Thermal Conductivity Detector The ability of a colliding molecule to carry off heat depending on its thermal conductivity. Hydrogen and helium have high thermal conductivity and therefore will be more efficient at “cooling” a heated filament than other gases will

Thermal Conductivity Detector The TCD will respond to any substance different from the carrier gas as long as its concentration is sufficiently high enough.
Thermal Conductivity Detector

Thermal Conductivity Detector

Electron Capture Detector Electron capture compound, X (highly electonegative element), tends to capture free electrons and increase the amount to ion recombination X (F, Cl and Br) + e XIon recombination : X- + N 2+ = X + N 2 The current will decrease and this decrease constitutes the signal. Halogens, lead, phosphorous, nitro groups, silicone and polynuclear aromatics. Insecticides, pesticides, vinyl chloride, and fluorocarbons
Electron Capture Detector
- Slides: 39