GAS LAWS Unit 3 A CHARACTERISTICS OF GASES



































- Slides: 48

GAS LAWS Unit 3 A

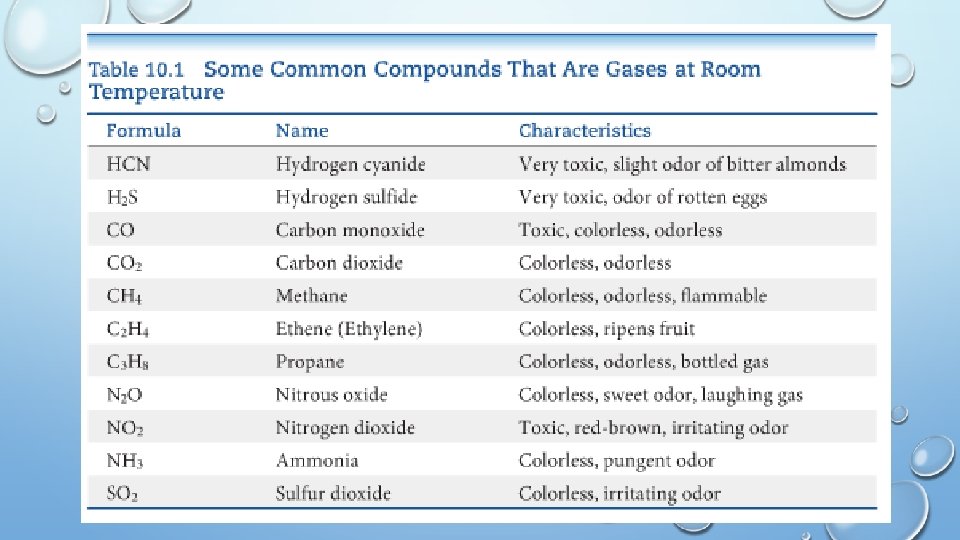
CHARACTERISTICS OF GASES • Physical properties of gases are all similar. • Composed mainly of nonmetallic elements with simple formulas and low molar masses. • Unlike liquids and solids, gases �Expand to fill their containers. �Are highly compressible. �Have extremely low densities. • Two or more gases form a homogeneous mixture.

PROPERTIES WHICH DEFINE THE STATE OF A GAS SAMPLE 1) TEMPERATURE 2) PRESSURE 3) VOLUME 4) AMOUNT OF GAS, USUALLY EXPRESSED AS NUMBER OF MOLES

PRESSURE • PRESSURE is the amount of force applied to an area: F P= A • Atmospheric pressure is the weight of air per unit of area.

UNITS OF PRESSURE • Pascal: 1 Pa = 1 N/m 2 (SI unit of pressure) • mm. Hg or Torr: these units are literally the difference in the heights measured in mm of two connected columns of mercury, as in the barometer in the figure. • Atmosphere: 1. 00 atm = 760 torr = 760 mm. Hg = 101. 325 k. Pa

Practice Pressure 1. A gas exerts a pressure of 585 torr. What is that in atm? 2. Rank the following pressures in increasing order of magnitude: 75 k. Pa, 300 torr, 0. 60 atm, and 350 mm. Hg 1. 0. 770 atm 2. 300 torr < 350 mm. Hg < 0. 60 atm < 75 k. Pa

MANOMETER The manometer is used to measure the difference in pressure between atmospheric pressure and that of a gas in a vessel. (The barometer seen on the last slide is used to measure the pressure in the atmosphere at any given time. )

STANDARD PRESSURE • Normal atmospheric pressure at sea level is referred to as standard atmospheric pressure. • It is equal to � 1. 00 atm � 760 torr (760 mm. Hg) � 101. 325 k. Pa

KINETIC-MOLECULAR THEORY �Laws tell us what happens in nature. Each of the gas laws tells us what is observed under certain conditions. �Why are these laws observed? We will discuss a theory to explain our observations.

MAIN TENETS OF KINETIC MOLECULAR THEORY 1) Gases consist of large numbers of particles that are in constant, random motion. 2) The volume of all the gas particles is negligible. 3) Attractive and repulsive forces between gas particles are negligible. 4) All collisions between the particles are perfectly elastic. 5) The average kinetic energy of the particles is proportional to the absolute temperature.

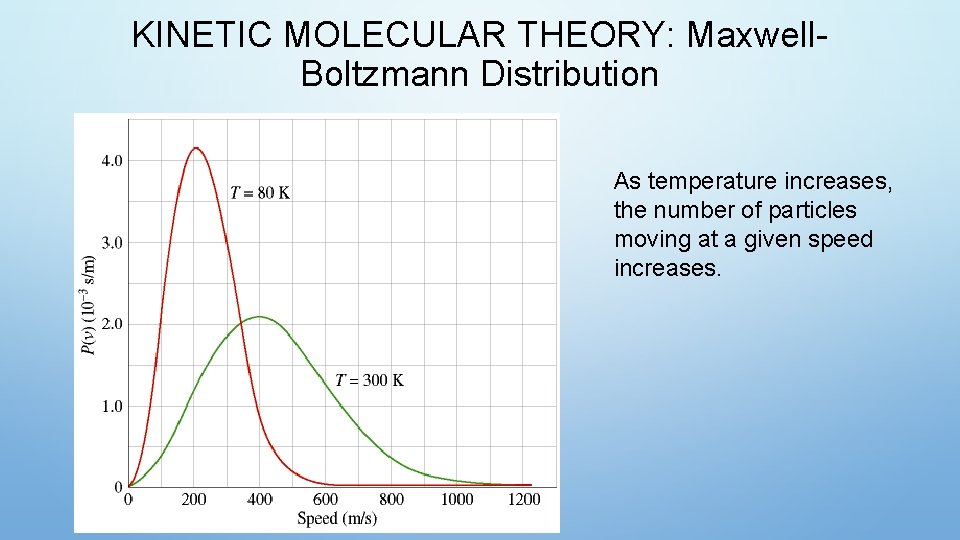
KINETIC MOLECULAR THEORY: Maxwell. Boltzmann Distribution As temperature increases, the number of particles moving at a given speed increases.


DALTON’S LAW OF PARTIAL PRESSURES • If two gases that do not react are combined in a container, they act as if they are alone in the container. • The total pressure of a mixture of gases equals the sum of the pressures that each gas would exert if it were present alone. PTOTAL = P 1 + P 2 + P 3 + …

MOLE FRACTION • That ratio of moles of a substance to total moles is called the mole fraction, χ. • Because each gas in a mixture acts as if it is alone, we can relate amount in a mixture to partial pressures: P 1 = χ1 Ptotal

Dalton’s Law Practice 1 PHe = 9. 2 atm PO 2 = 2. 4 atm PT = 11. 6 atm

Dalton’s Law Practice 2 0. 210

Dalton’s Law Practice 3 593 torr

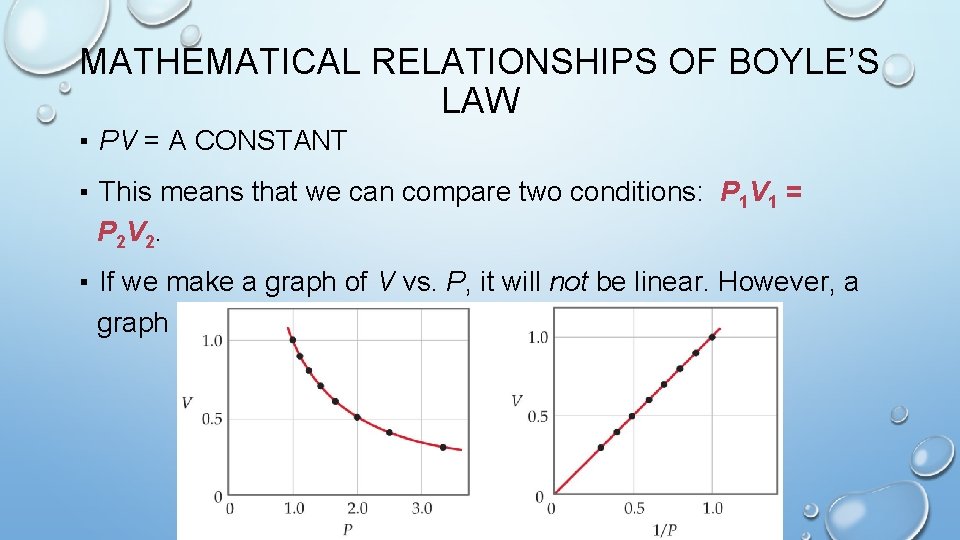
BOYLE’S LAW The volume of a fixed quantity of gas at constant temperature is inversely proportional to the pressure.

MATHEMATICAL RELATIONSHIPS OF BOYLE’S LAW ▪ PV = A CONSTANT ▪ This means that we can compare two conditions: P 1 V 1 = P 2 V 2. ▪ If we make a graph of V vs. P, it will not be linear. However, a graph of V vs. 1/P will result in a linear relationship.

Boyle’s Law Practice 0. 57 L

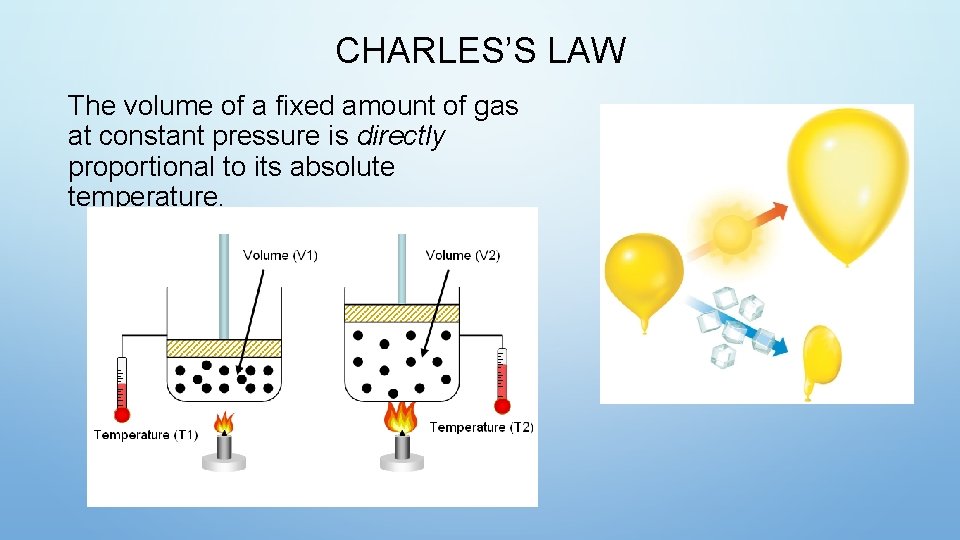
CHARLES’S LAW The volume of a fixed amount of gas at constant pressure is directly proportional to its absolute temperature.

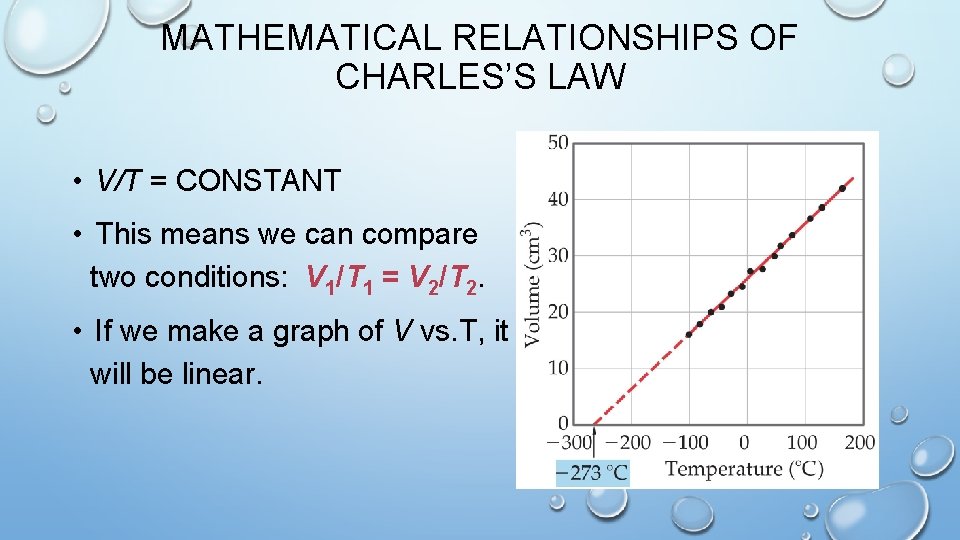
MATHEMATICAL RELATIONSHIPS OF CHARLES’S LAW • V/T = CONSTANT • This means we can compare two conditions: V 1/T 1 = V 2/T 2. • If we make a graph of V vs. T, it will be linear.

Charles’s Law Practice 2. 79 L

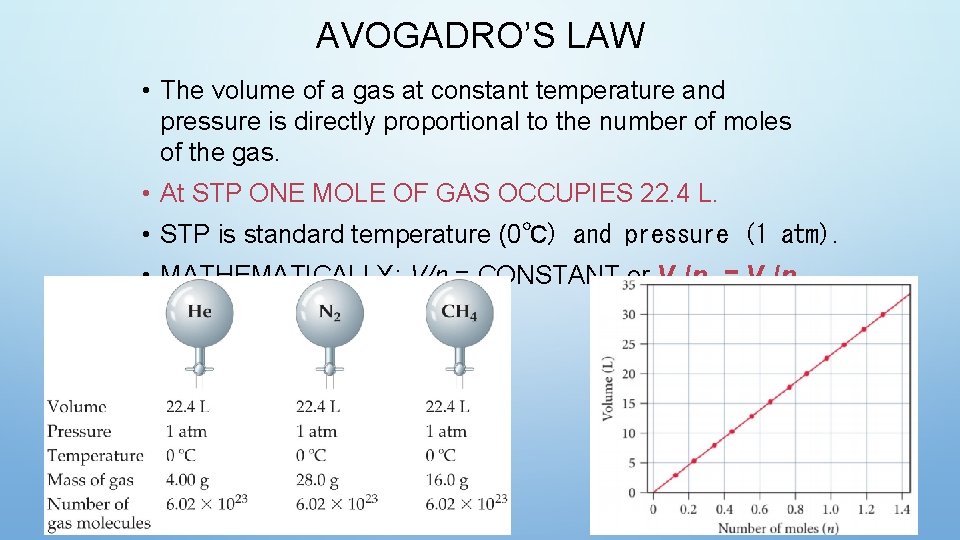
AVOGADRO’S LAW • The volume of a gas at constant temperature and pressure is directly proportional to the number of moles of the gas. • At STP ONE MOLE OF GAS OCCUPIES 22. 4 L. • STP is standard temperature (0℃) and pressure (1 atm). • MATHEMATICALLY: V/n = CONSTANT or V 1/n 1 = V 2/n 2

Avogadro’s Law Practice 8. 1 L

Gay-Lussac’s Law (Amonton’s Law) Pressure of a given mass of gas varies directly with the absolute temperature of the gas, when the volume is kept constant. P 1/T 1 = P 2/T 2

Combined Gas Law “Peas and veggies are on the table. ”

IDEAL GAS LAW • So far we’ve seen that • Combining these, we get V ∝ 1/P (Boyle’s law). V ∝ T (Charles’s law). V ∝ n (Avogadro’s law). n. T P V ∝ • Finally, to make it an equality, we use a constant of proportionality, R. • Rearranging the equation gives the Ideal Gas Equation: PV = n. RT.

Ideal Gas Law Practice 1 0. 57 mol

Ideal Gas Law Practice 2 Determine the mass of a sample of argon gas in a 3. 0 L container at 56 ℃ and a pressure of 897 torr. 0. 131 mol, 5. 2 g Ar

Gas Stoichiometry • The balanced equation tells us relative amounts of moles in a reaction. • We can also relate volume for gases. • Use PV = n. RT for gas A to get moles A and use the mole ratio from the balanced equation to get moles of substance B.

Gas Stoichiometry Practice 1 34. 0 L CO 2 at STP

Gas Stoichiometry Practice 2 CH 4(g) + 2 O 2(g) → CO 2(g) + 2 H 2 O(g) 2. 47 L

DENSITY OF GASES If we divide both sides of the ideal gas equation by V and by RT, we get n/V = P/RT Also, moles × molar mass = mass n * MM = mass If we multiply both sides by mass and substituting n. MM on the right side of the equation, we get mass/V = MMP/RT and mass/V is density, D; the result is: D = MMP/RT (One needs to know only the molar mass, the pressure, and the temperature to calculate the density of a gas. )

DENSITY & MOLECULAR MASS OF A GAS We can rearrange the equation to solve for molar mass: MM =DRT/P “Molar Mass kitty cat puts DRT over P. ”

Density of Gases Practice 32. 0 g/mol

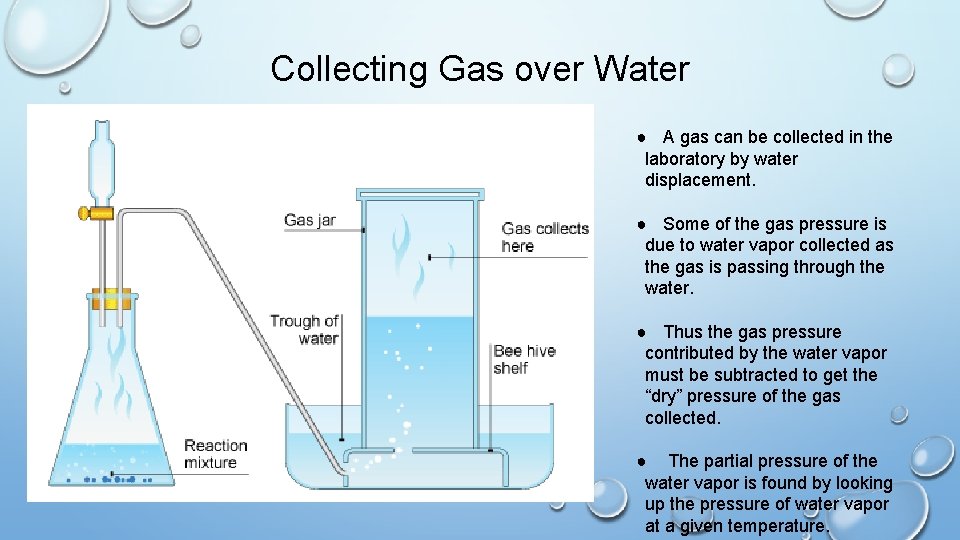
Collecting Gas over Water ● A gas can be collected in the laboratory by water displacement. ● Some of the gas pressure is due to water vapor collected as the gas is passing through the water. ● Thus the gas pressure contributed by the water vapor must be subtracted to get the “dry” pressure of the gas collected. ● The partial pressure of the water vapor is found by looking up the pressure of water vapor at a given temperature.

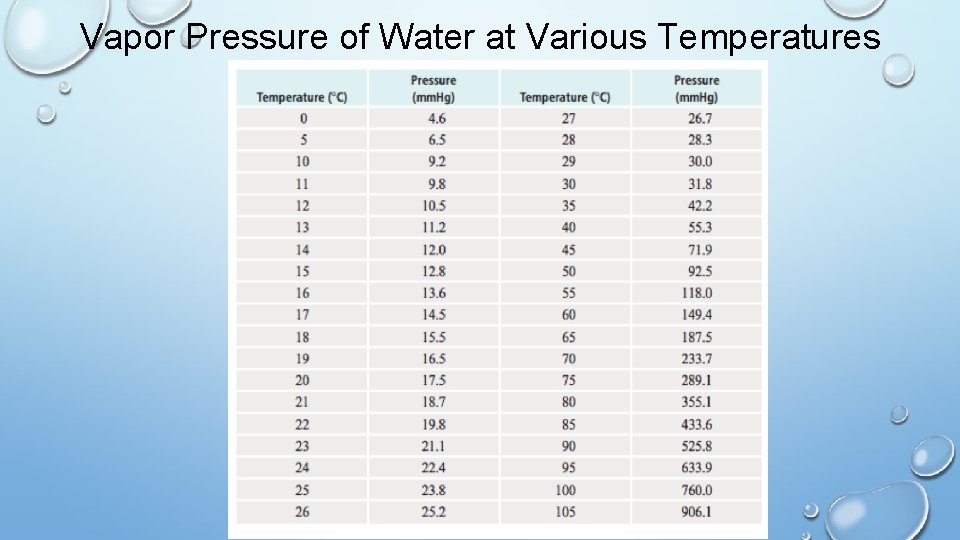
Vapor Pressure of Water at Various Temperatures

Collecting Gas over Water Example A sample of solid potassium chlorate (KCl. O 3) was heated in a test tube and decomposed by the following reaction: 2 KCl. O 3 (s) → 2 KCl (s) + 3 O 2 (g) The oxygen produced was collected by water displacement at 22 ℃ at a total pressure of 754 torr. The volume of the gas collected was 0. 650 L, and the vapor pressure of water at 22 ℃ is 19. 8 torr. Calculate the partial pressure of O 2 gas collected and the mass of KCl. O 3 in the sample that was decomposed. PO 2 = 734 torr; (0. 0173 mol KCl. O 3) 2. 12 g KCl. O 3

Collecting Gas over Water Example 2 KCl. O 3 (s) → 2 KCl (s) + 3 O 2 (g)

Collecting Gas over Water Practice Nitrogen gas was produced in a reaction and collected over water. A 45. 2 m. L sample of gas was collected over water at 25 ℃. The reading on the barometer in the lab was 0. 97 atm. The vapor pressure of water is 23. 76 mm. Hg at 25 ℃. Find the mass of nitrogen gas collected. 0. 048 g

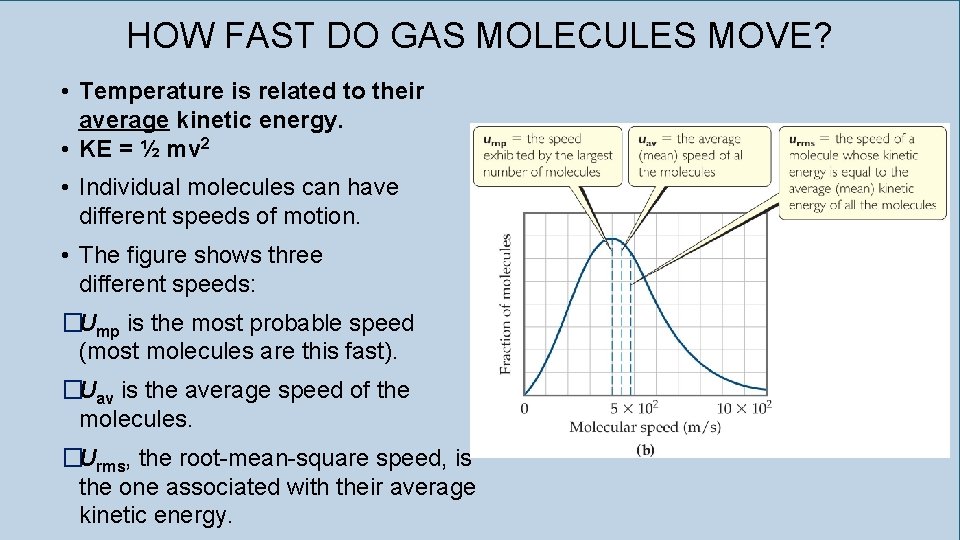
HOW FAST DO GAS MOLECULES MOVE? • Temperature is related to their average kinetic energy. • KE = ½ mv 2 • Individual molecules can have different speeds of motion. • The figure shows three different speeds: �Ump is the most probable speed (most molecules are this fast). �Uav is the average speed of the molecules. �Urms, the root-mean-square speed, is the one associated with their average kinetic energy.

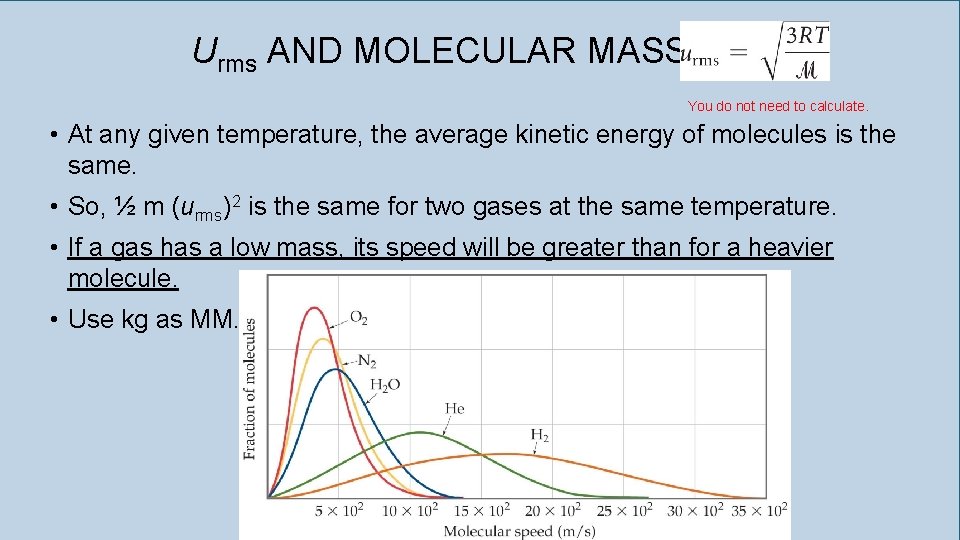
Urms AND MOLECULAR MASS You do not need to calculate. • At any given temperature, the average kinetic energy of molecules is the same. • So, ½ m (urms)2 is the same for two gases at the same temperature. • If a gas has a low mass, its speed will be greater than for a heavier molecule. • Use kg as MM.

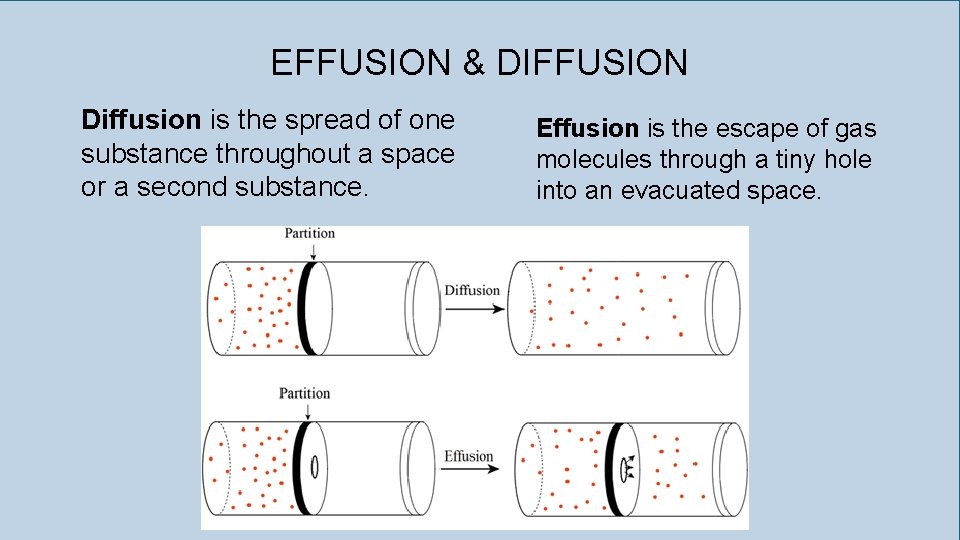
EFFUSION & DIFFUSION Diffusion is the spread of one substance throughout a space or a second substance. Effusion is the escape of gas molecules through a tiny hole into an evacuated space.

GRAHAM’S LAW DESCRIBES DIFFUSION & EFFUSION • Graham’s law relates the molar mass of two gases to their rate of speed of travel. • The “lighter” gas always has a faster rate of speed. • Diffusion experiment: HCl + NH 3 r = rate of gas 1 r = rate of gas 2 M 1 = molar mass You do not need to calculate.

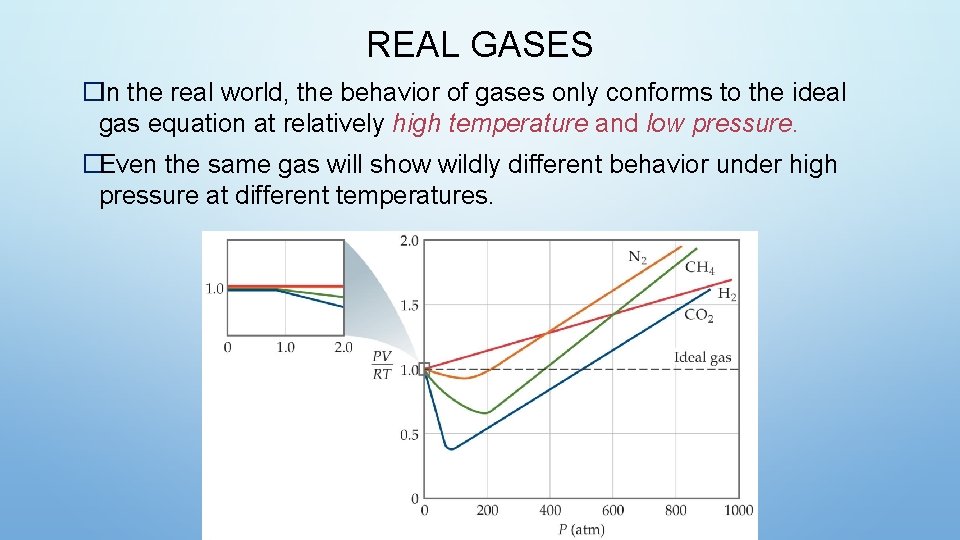
REAL GASES �In the real world, the behavior of gases only conforms to the ideal gas equation at relatively high temperature and low pressure. �Even the same gas will show wildly different behavior under high pressure at different temperatures.

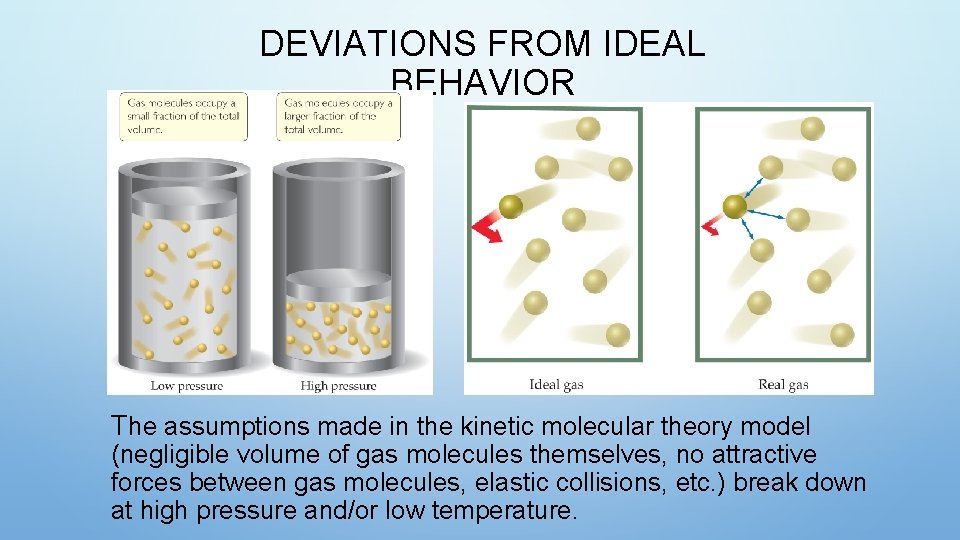
DEVIATIONS FROM IDEAL BEHAVIOR The assumptions made in the kinetic molecular theory model (negligible volume of gas molecules themselves, no attractive forces between gas molecules, elastic collisions, etc. ) break down at high pressure and/or low temperature.

CORRECTIONS FOR NON-IDEAL BEHAVIOR • The ideal gas equation can be adjusted to take these deviations from ideal behavior into account. • The corrected ideal-gas equation is known as the van der waals equation. • The pressure adjustment is due to the fact that molecules attract and repel each other. • The volume adjustment is due to the fact that molecules occupy some space on their own.