Gas Laws Remember that gas has mass Pressure
Gas Laws Remember that gas has mass
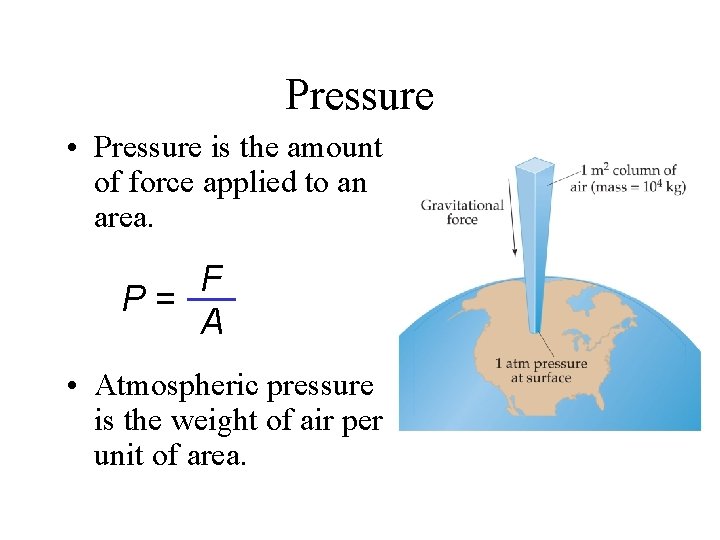
Pressure • Pressure is the amount of force applied to an area. F P= A • Atmospheric pressure is the weight of air per unit of area.

Pressure • What is pressure? – Accumulated force of the collisions of atoms • Pascals (Pa) or kilopascals (k. Pa) – 1 Pa = 1 newton/square meter = 1 N/m 2 • Bar – 1 bar = 105 Pa = 100 k. Pa • mm Hg or torr – These units are literally the difference in the height measured in mm of a mercury barometer. Atmospheres (atm) – Average value of atmospheric pressure at sea level 1 atm = 760 torr = 101. 325 k. Pa

How is Pressure Measured • Barometers and manometers – Use pressure to elevate a liquid • An open-end manometer is used to measure the difference between atmospheric pressure and that of a gas in a vessel. • A closed-end manometer will only measure the pressure of the gas inside the vessel. • Piezoelectric chips

Pressure Conversions • Normal atmospheric pressure at sea level and room temperature is referred to as standard temperature and pressure, or STP. • 1 atm = 760 torr = 760 mm. Hg = 14. 7 psi • 1 atm = 101, 325 Pa (use k. Pa) • Temperature – 25 ºC = 298 Kelvin USE KELVIN! ALL THE TIME! Kelvin = Celsius + 273 REMEMBER ME!

Gas Laws • There are three gas laws discovered independently that tell us how gases behave when certain variables are changed. • Boyle’s • Charles’ • Avogadro’s
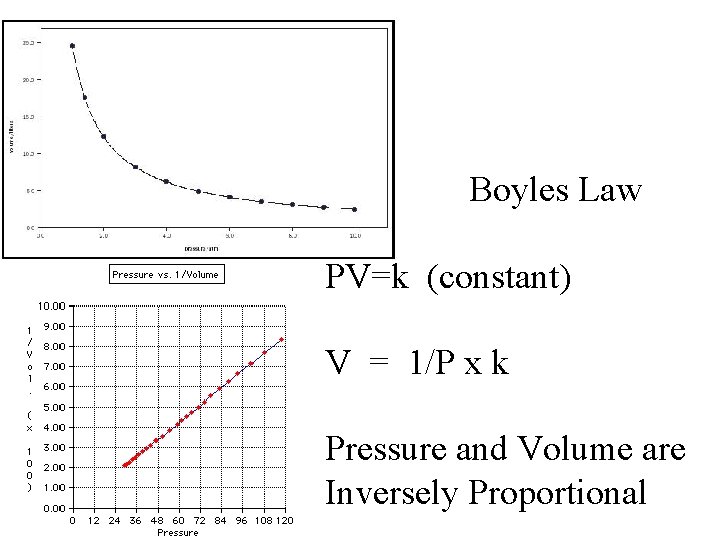
Boyles Law PV=k (constant) V = 1/P x k Pressure and Volume are Inversely Proportional
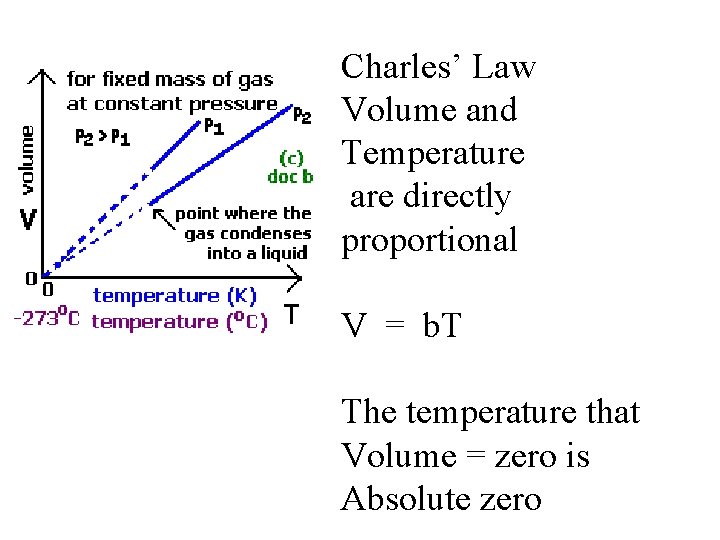
Charles’ Law Volume and Temperature are directly proportional V = b. T The temperature that Volume = zero is Absolute zero

Avogadro’s Law • Volume of a gas is directly proportional to the number of molecules • V = na ü V = volume in liters ü n = number of moles ü a = proportionality constant • Avogadro did not invent Avogadro’s number! It was named after him 50 years after his death

Ideal Gas Law • If • And PV = k V = b. T V = an • Then • PV = n. T x constant PV = n. RT

Ideal Gas Law • Ideal Gas Law is an Equation of State – Given any three, you can determine the fourth – It is empirically derived • It expresses what REAL gases approach – At low pressure – High temperature – Using KMT : • Why is low pressure and high temp conditions required for a gas to approach ideal conditions?

Gas Law Problems • Use the equation for all problems. • R = PV n. T • What is constant in the problem? • Derive the equation and solve.

A 125. 01 L balloon is at 250. 0 K It is heated to 350. 0 K. What is the volume? • R = PV n. T • What is constant? • Moles and pressure. • R = V 1 = V 2 T 1 T 2 • 175. 0 L

Gas Stoichiometry • One mole of any gas at STP (273 K, 1 atm) is 22. 4 liters. • True for Ideal Gases. • R = PV n. T • P = 1 atm, V = 22. 4 L, T = 273. 15 K, and moles (n) = 1. 0, then R = 0. 0821 L atm / mol K

Units of R • There are two common “R”’s – Besides the pirates “rrrrrr” • 0. 0821 L atm /mol k – Used in gas problems And • 8. 3145 L Kpa / mol K – used in thermo problems, whenever the answer is in joules

15. 0 TL (teraliter) of hydrogen gas at 450 K and 1488 torr was reacted with 273 Tg (teragram) of iron (III) oxide. • What is the reaction? • What is the limiting reactant? • How much iron will be formed? • What is the pressure of the water assuming the reaction tank is at the same conditions (temperature and volume) as the reactants?

Gas and Molar Mass • Whenever moles are used in a relationship – Like the ideal gas law • It can be thought of as “grams divided by molar mass” – Or – g molar mass (M)

Molar Mass of Gas • PV = n. RT • P = n. RT = (m/M) R T V V • P = (m)(RT) = d R T V M M m = mass, d = density (units = g/L)

Rearrange the Equation • P = d R T M • Molar mass = d R T P • m = mass, d = density (units g/L)

Dalton’s Law The total pressure of a gas mixture is the sum of the partial pressures of the gases if they were alone. Ptotal = P 1 + P 2 + P 3 +…. .

Dalton’s Law • The pressure is a combination of all partial pressures • It assumes gases have no influence on each other • Under what conditions do gases act ideally?

Mole Fraction • Mole Fraction is the fraction of the moles of one substance in a mixture compared to the total number of moles • Mole fraction • X 1 = n 1 ntotal n 1+ n 2+ n 3+ …… • If V and T are constant X 1 = P 1 or P 1 = X 1 • Ptotal

Gas Collected Over Water Gases collected over water always have some water vapor included due to evaporation. (Vapor pressure) If the water level in the flask is equal to the surrounding water, than the inside pressure is equal to the outside pressure. Pin = PO 2 + PH 2 O = P atmospheric

Vapor Pressure Explaining Vapor Pressure on a Molecular Level • Some of the molecules on the surface of a liquid have enough energy to escape the attraction of the bulk liquid. • These molecules move into the gas phase. • As the number of molecules in the gas phase increases, some of the gas phase molecules strike the surface and return to the liquid. • After some time the pressure of the gas will be constant at the vapor pressure.

Vapor Pressure Explaining Vapor Pressure on the Molecular Level • Dynamic Equilibrium: the point when as many molecules escape the surface as strike the surface. • Vapor pressure is the pressure exerted when the liquid and vapor are in dynamic equilibrium.

Kinetic-Molecular Theory of moving molecules developed to explain gas behavior. Assumptions: · Gases · · consist of a large number of molecules in constant random motion. Volume of individual molecules negligible compared to volume of container. Intermolecular forces (forces between gas molecules) negligible. Energy can be transferred between molecules, but total kinetic energy is constant at constant temperature. Average kinetic energy of molecules is proportional to temperature.

Kinetic-Molecular Theory • Kinetic molecular theory gives us an understanding of pressure and temperature on the molecular level. • Pressure of a gas results from the number of collisions per unit time on the walls of container

Kinetic-Molecular Theory • Magnitude of pressure given by how often and how hard the molecules strike. • Since the mass is small, the momentum of the atom is really small, however there a lot of atom • What ever increases the number of collisions will increase the pressure (more atoms in the same space) • What ever increases the kinetic energy of the particle will increase the pressure (Temperature increase) • Gas molecules have an average kinetic energy but each molecule has a different energy within a certain range. • As the temperature increases, the average kinetic energy of the gas molecules increases.

Kinetic-Molecular Theory Boltzman Distribution Colder gas Warmer gas

Kinetic Molecular Theory

Kinetic Molecular Theory

Kinetic-Molecular Theory • As kinetic energy increases, the velocity of the gas molecules increases. • Root mean square speed, speed u, is the speed of a gas molecule having average kinetic energy. It is calculated by taking the square root of the average of the squared speeds of the gas molecules in a gas sample. • Average kinetic energy, KE, is related to root mean square speed and the molar mass of the gas: 2 KE = 1/2 mu

Kinetic-Molecular Theory Application to the Gas Laws • As volume increases at constant temperature, temperature the average kinetic of the gas remains constant. Therefore, u is constant. However, volume increases so the gas molecules have to travel further to hit the walls of the container. Therefore, pressure decreases • If temperature increases at constant volume, volume the average kinetic energy of the gas molecules increases. Therefore, there are more collisions with the container walls and the pressure increases

Molecular Effusion and Diffusion If one particle has more mass than the other, it must be moving slower since they have the same KEavg! Different gases at the same temperature have different average speeds. The bigger particles are moving slower. Mathematically: The lower the molar mass, M, the higher the rms, u, for that gas at a constant temperature.

Using Equation • Velocity of a gas particle can be calculated • In AP exam, you will be given the equation: • urms= (3 RT) 1/2 M • R is 8. 3145 J/k • mol (from KE) • M is in Kg/mol ( molar mass x 10 -3) • Derivation on Pg 216

Molecular Effusion and Diffusion

Molecular Effusion and Diffusion Graham’s Law of Effusion

Molecular Effusion and Diffusion Graham’s Law of Effusion • Only those molecules that hit the small hole will escape through it. • Therefore, the higher the rms the more likelihood of a gas molecule hitting the hole. • We can show

Molecular Effusion and Diffusion and Mean Free Path • Diffusion of a gas is the spread of the gas through space. • Diffusion is faster for light gas molecules. • Diffusion is significantly slower than rms speed (consider someone opening a perfume bottle: it takes while to detect the odor but rms speed at 25 C is about 1150 mi/hr). • Diffusion is slowed by gas molecules colliding with each other. • Average distance of a gas molecule between collisions is called mean free path.

Molecular Effusion and Diffusion and Mean Free Path • At sea level, mean free path is about 6 10 -6 cm.

Ideal vs Real Gases • Size of atom doesn’t count • Molecules do not interact • Kinetic energy (velocity) is directly proportional to temperature • Size of atom does • Molecules do interact • Even non-polar molecules interact! • Velocity is not directly proportional (close but no cigar)

Real Gases: Deviations from Ideal Behavior • From the ideal gas equation, we have • For 1 mol of gas, PV/RT = 1 for all pressures. • In a real gas, PV/RT varies from 1 significantly. • The higher the pressure the more the deviation from ideal behavior.

Real Gases • P= n. RT V • P = n. RT V – nb • The molecules actually take up space • Pobs = P’ - factor = P’ – a(n/V)2 • P = n. RT – a(n/V)2 V – nb • Molecules attract

Van der Waals Equation • Corrected version of the ideal gas law. • Uses two constants: a and b – which are experimentally determined and will be given for real gas calculations. • These constants “correct” the pressure and volume from ideal to real.

Van der Waals equation This equation is a modification of the ideal gas relationship. It accounts for attractive forces and molecular volume. an 2 P + 2 (V - nb) = n. RT V ( ) Correction for Molecular volume Correction for attractive forces between molecules

Clearly, not all gases behave ideal.

Even the same gas acts differently at different temperatures.

Real Gases • The assumptions of the kinetic-molecular theory break down at low temperature and high pressure. • Increased collisions between particles change the ideal behavior.

Values for a, b Gas a (atm ∙L 2)/mol 2 b (L/mol) He 0. 0341 0. 0237 Ne 0. 211 0. 0171 Kr 2. 32 0. 0398 Xe 4. 19 0. 0511 CO 2 3. 59 0. 0427 CH 4 2. 25 0. 0428 NH 3 4. 17 0. 0371 H 2 O 5. 46 0. 0305

Van der Waals Equation • If 1. 000 mol of an ideal gas were confined to 22. 41 L at 0. 0 ºC, it would exert a pressure of 1. 000 atm. Use the van der Waals equation and the values of a and b for Cl 2 to estimate the pressure exerted by 1. 000 mol of Cl 2 in 22. 41 L at 0. 0 ºC. • a = 6. 49 L 2 -atm/mol 2 • b = 0. 0562 L/mol

Molecular Comparison of Liquids & Solids • Converting a gas into a liquid or solid requires the molecules to get closer to each other: – cool or compress. • Converting a solid into a liquid or gas requires the molecules to move further apart: – heat or reduce pressure. • The forces holding solids and liquids together are called intermolecular forces.

Phase Changes Energy Changes Accompanying Phase Changes

Molecular Comparison of Liquids & Solids

• Solid - the attractive forces are stronger than the kinetic energy of the particles – The particles are held in position • Gas - the attractive forces are weak compared to their kinetic energy – particles move freely, are far apart, and have almost no influence on one another. • Liquid - the attractive forces between particles pull the particles close together – The particles have considerable freedom to move about.

Why Aren’t All Substances Gases? • Democritus’ theory of atoms was dismissed because why don’t all these particles fall apart like sand? • Why don’t all these particles fall apart like sand? • If there is nothing holding molecules together, then they should be free to go where ever. Just like an ideal gas.

Dipole-Dipole • Why does a molecule have a dipole? • When two molecules approach one another – Positive and negative sides are attracted – This attraction restricts the movement or – it takes more energy to be a gas (break the attractive force)

Dipole–dipole forces: • The positive and negative ends of polar molecules – are attracted to one another by dipole–dipole forces. – molecules have higher boiling points than nonpolar molecules of similar size. Copyright © 2010 Pearson Education, Inc. Chapter Eight 57

Dipoles line up To minimize repulsion And maximize attraction The closer the molecules The more important Intermolecular forces

Hydrogen Bond • Hydrogen make particularly strong dipoles – It is a very small atom so it can get real close • Relatively strong intermolecular force • The unusual properties of water are due to hydrogen bonding

Hydrogen bond • O, N, or F atom and a positively polarized hydrogen atom bonded to another electronegative O, N, or F. • An interaction between an unshared electron pair and the polarized hydrogen • Hydrogen bonds occur in both water and ammonia.

O O H H H H O O O H H Intermolecular bonds are responsible for the “condensed states


Boiling Points Smaller atoms are more electronegative, so they have more polar bonds. H – bonding is more effective so they have higher boiling points The higher the molecular weight The higher the boiling point. Ask why!


London Dispersion Forces • Why does a noble gas condense into a liquid? – It has no polarity – It is not reactive – What attracts one atom to another?

London Dispersion Forces • On average, the electron distribution in a nonpolar molecule is symmetrical. • At any instant, it may be unsymmetrical, resulting in a temporary polarity that can attract neighboring molecules. • All molecules, regardless of structure, experience London dispersion forces. – Only polar molecules experience dipole-dipole

London Dispersion Forces Can be viewed as an “induced dipole” Copyright © 2010 Pearson Education, Inc. Chapter Eight 67


Induced Dipoles • When non-polar molecules approach – The negative electron clouds repel – Inducing a dipole – Which allows the molecules to interact • Helium freezes at 3 K – Have to move really slowly to induce a dipole

Van Der Waals Forces • The longer the chain, the higher the boiling point • The chains get tangled like spaghetti • Takes more energy to break intermolecular tangles or in other words • It has a higher boiling point

Which has the higher boiling point, melting point and why? • • Heptane or Octane 1 -Decanol or 1 - octanol Ammonia or methyl amine (NH 2 CH 3) Hydrogen sulfide or hydrogen oxide Hydrogen selenide or hydrogen telluride Decane or 2, 3 diethyl hexane (isomer of decane) Xenon or krypton

Liquids • Physical properties of liquids are determined mainly by the nature of their intermolecular forces

Some Properties of Liquids Viscosity • Viscosity is the resistance of a liquid to flow. • A liquid flows by sliding molecules over each other. • The stronger the intermolecular forces, the higher the viscosity. Karo syrup vs water Cold oil vs hot oil Surface Tension • Bulk molecules (those in the liquid) are equally attracted to their neighbors. beads of water on a newly waxed car meniscus in graduated cylinder

Viscosity Properties • Resistance to flow. of liquids • This increases with increased intermolecular attractions. CH 3 CH 2 CH 3 CH CH 2 CH CH 2 OH OH OH Increasing viscosity • Also, liquids composed of long, flexible molecules can entwine, resulting in increased

Some Properties of Liquids Surface Tension


Some Properties of Liquids Surface Tension • Surface molecules are only attracted inwards towards the bulk molecules. – Therefore, surface molecules are packed more closely than bulk molecules. • Surface tension is the amount of energy required to increase the surface area of a liquid. • Cohesive forces bind molecules to each other. • Adhesive forces bind molecules to a surface.

Properties of liquids Surface Tension • Force in the surface of a liquid that makes the area of the surface as small as possible. Molecules at the surface interact only with neighbors inside the liquid.

Properties of liquids Capillary action • It is the competition between two forces. Cohesive forces • The attractions between molecules of a substance. Adhesive forces • Attractions between molecules of different substances.

Properties of liquids Capillary action Capillary tube meniscus Mercury Cohesive is larger than adhesive. Water Adhesive is larger than cohesive.

Some Properties of Liquids Surface Tension • Meniscus is the shape of the liquid surface. – If adhesive forces are greater than cohesive forces, the liquid surface is attracted to its container more than the bulk molecules. Therefore, the meniscus is U-shaped (e. g. water in glass). – If cohesive forces are greater than adhesive forces, the meniscus is curved downwards. • Capillary Action: When a narrow glass tube is placed in water, the meniscus pulls the water up the tube.

Properties of liquids Diffusion • This takes place in both liquids and gases. It is the spontaneous mixing of materials that results from the random motion of molecules.

Water is Weird • • Most abundant substance on earth’s surface You are 60% water High heat capacity High boiling point Lower density solid than liquid High surface tension High heat of vaporization Universal solvent

Vapor Pressure • What does it mean when something evaporates? • What does it mean when something boils? • What is vapor pressure again?

Rate of Escape = Rate of Return

Equilibrium ----time-

What is Vapor Pressure? • Gas has mass lab – Water has a vapor pressure of 17. 2 mm. Hg@20 C • Pressure is the accumulated collisions – More molecules mean more collisions • The warmer the water – The higher the vapor pressure

Different Compounds have Different Vapor Pressures • Why? • How could you test? – Qualitative – Quantitative

Evaporation • In order for one molecule to escape – It has to break the intermolecular attractions – It has to have enough kinetic energy to leave – Why does one molecule have enough energy to leave and another does not? • It has to do with the concept of average
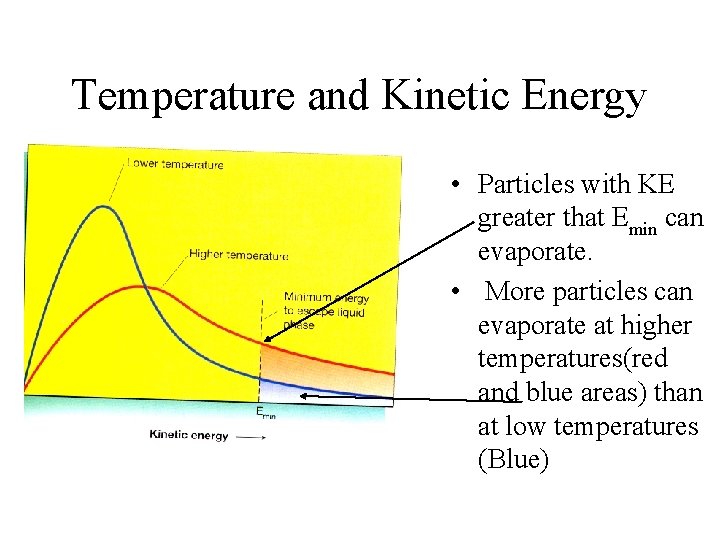
Temperature and Kinetic Energy • Particles with KE greater that Emin can evaporate. • More particles can evaporate at higher temperatures(red and blue areas) than at low temperatures (Blue)

Boiling • Higher temps = higher kinetic energy – More escaping molecules – When the pressure of the escaping molecules exceeds atmospheric pressure – The solution is said to boil – Vapor pressure = atmospheric pressure

Atmosphere is exerting pressure or colliding with particles. When the vapor pressure exceeds atmospheric pressure, it boils

What is Boiling, Condensing, Melting, Freezing? • Heat of Vaporization – k. J/mol to go from liquid to gas – Energy to overcome all intermolecular interactions • Heat of Fusion – k. J/mol to go from liquid to solid – Energy to be able to move past your neighbor

Copyright © 2010 Pearson Education, Inc. Chapter Eight 94

Phase Changes Heating Curves
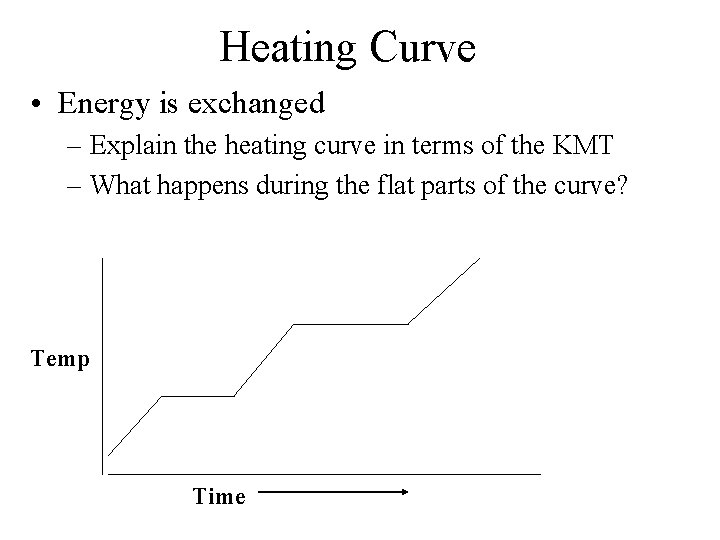
Heating Curve • Energy is exchanged – Explain the heating curve in terms of the KMT – What happens during the flat parts of the curve? Temp Time

Phase Diagrams • Phase diagram: plot of pressure vs. Temperature summarizing all equilibria between phases. • Given a temperature and pressure, phase diagrams tell us which phase will exist. • Features of a phase diagram: – Triple point: temperature and pressure at which all three phases are in equilibrium. – Vapor-pressure curve: generally as pressure increases, temperature increases. – Critical point: critical temperature and pressure for the gas. – Melting point curve: as pressure increases, the solid phase is favored if the solid is more dense than the liquid. – Normal melting point: melting point at 1 atm.

Phase Diagrams • Any temperature and pressure combination not on a curve represents a single phase.

Phase Changes Critical Temperature and Pressure • Gases liquefied by increasing pressure at some temperature. • Critical temperature: temperature the minimum temperature for liquefaction of a gas using pressure. • Critical pressure: pressure the minimum pressure required for liquefaction at the critical temperature.

Phase Diagram of Water Pressure Super critical fluid Freezing Melting liquid solid Evaporation Condensation gas Triple Point Sublimation Deposition Temperature
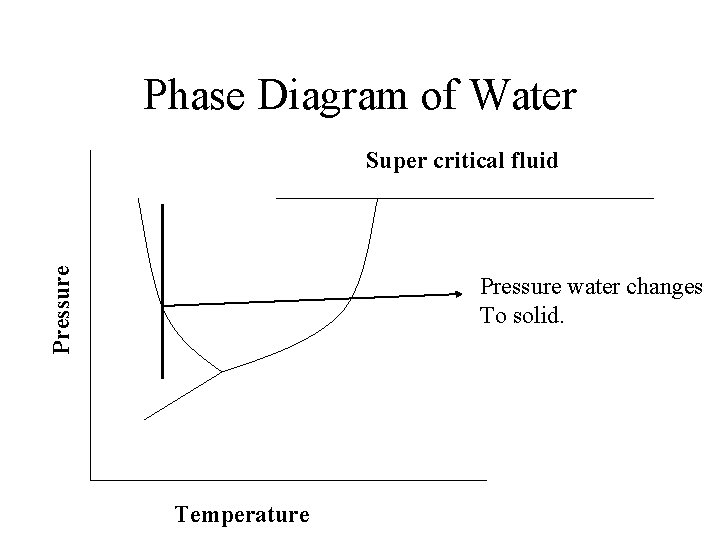
Phase Diagram of Water Pressure Super critical fluid Pressure water changes To solid. Temperature

“Normal” Boiling and Melting Point 1 atm Pressure 100 C Temperature

Vapor Pressure Volatility, Vapor Pressure, and Temperature

Water Vapor Pressure ln P k. Pa Pressure, 0 3. 0 20 3. 440 3. 860 4. 280 4. 6 100 The Clausius-Clapeyron Equation 00. 002720 400. 003260 80 0. 0037 100 Temperature ºC 1/Kelvin temp

Structures of Solids Unit Cells • Crystalline solid: solid well-ordered, definite arrangements of molecules, atoms or ions. • Crystals have an ordered, repeated structure. • The smallest repeating unit in a crystal is a unit cell. • Unit cell is the smallest unit with all the symmetry of the entire crystal. • Three-dimensional stacking of unit cells is the crystal lattice.

Hydrogen Bonds in H 2 O snowflake

Structures of Solids Unit Cells

Unit Cells Structures of Solids Three common types of unit cell. • Primitive(simple) cubic, atoms at the corners of a simple cube – each atom shared by 8 unit cells; • Body-centered cubic(bcc), atoms at the corners of a cube plus one in the center of the body of the cube – corner atoms shared by 8 unit cells, center atom completely enclosed in one unit cell; • Face-centered cubic(fcc), atoms at the corners of a cube plus one atom in the center of each face of the cube – corner atoms shared by 8 unit cells, face atoms shared by 2 unit cells.

Structures of Solids Unit Cells

Space-Filling Cubic Cells
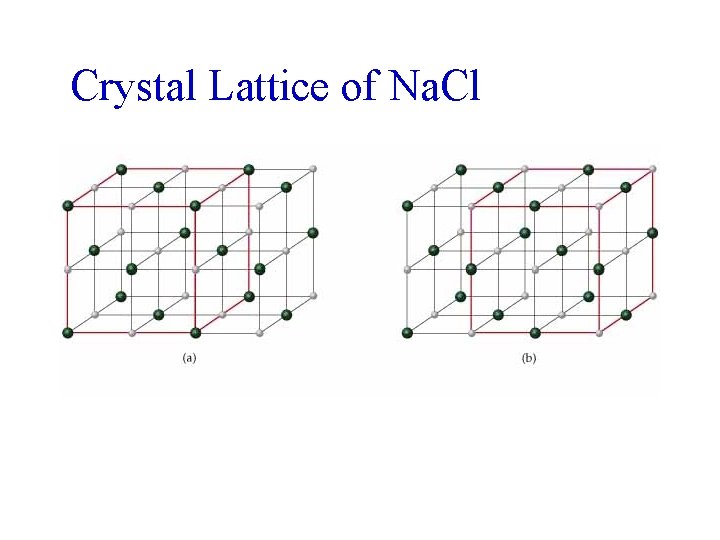
Crystal Lattice of Na. Cl

Unit Cell of Na. Cl

Structures of Solids Crystal Structure of Sodium Chloride • Face-centered cubic lattice. • Two equivalent ways of defining unit cell: – Cl- (larger) ions at the corners of the cell, or – Na+ (smaller) ions at the corners of the cell. • The cation to anion ratio in a unit cell is the same for the crystal. In Na. Cl each unit cell contains same number of Na+ and Cl- ions. • Note the unit cell for Ca. Cl 2 needs twice as many Clions as Ca 2+ ions.

Sample Unit Cells

Structures of Solids Close Packing of Spheres • Solids have maximum intermolecular forces. • Molecules, atoms or ions can be modeled by spheres. • Crystals are formed by close packing of the molecules, atoms or ions. • We rationalize maximum intermolecular force in a crystal by the close packing of spheres. • When spheres are packed as closely as possible, there are small spaces between adjacent spheres. The spaces are called interstitial holes • Other atoms can sometimes fit into these holes.

Hexagonal Close Packed Spheres

X-Ray Crystallography

Types of Solids

Diamond and Graphite

Cross Section of a Metal

Solids • Amorphous solids – “super cooled” liquids – glass, rubber, many plastics – gets softer and softer as heated

Solids • • Metallic solids - gold, silver Molecular solids - wax, rubber, plastic Ionic Solids - sodium chloride Covalent-network solids - diamond, graphite

Types and Properties of Solids Type Network Metallic Group 18 Molecular Ionic Structure Atom Molecule Ion Type of Bond Directional Covalent Bonds Nondir. Delocalized elctrons London Dispersion Forces Dipole LD Van d. Waals Ionic Properties Hard High MP Insulator Wide range of Very low MP mp, hardness Conductor Soft, Low MP Insulator Hard High MP Insulator Example Silver, iron Ice, Dry ice Na. Cl KF Diamond Argon

Metal Alloys • Alloy is a mixture of elements with metallic properties • Substitutional – replaces one element with the other in the structure of the solid – High grade steel – replace some irons with chromium, vanadium, titanium, etc • Interstitial – fits inside the structure of the solid – Steel – carbon fits inside of iron atoms

Semiconductors • Silicon and germanium most common • Doping – add elements (like an alloy) to change the conductivity • n-type semiconductor – has more conductivity • p-type semiconductor – has less conductivity • p-n junction and transitiors

A solution In a solution • The solute can’t be filtered out. • The solute always stays mixed. • Particles are always in motion. • Volumes may not be additive. • A solution will have different properties than the solvent • A solution consists of two component types. • solvent - component in the greater concentration • solute component in the lesser amount (You may have more than one. )

Physical states of solutions • Solutions can be made that exist in any of the three states. • Solid solutions • dental fillings, 14 K gold, sterling silver • • Liquid solutions • saline, vodka, vinegar, sugar water • Gas solutions • the atmosphere, anesthesia gases

Predicting Solubilites “Like dissolves like. ” Materials with similar polarity are soluble in each other. Dissimilar ones are not. • Miscible - Liquids that are soluble in each other in all proportions such as ethanol and water. • Immiscible - Liquids that are not soluble in each other such as hexane and water.

Solubility A measure of how much of a solute can be dissolved in a solvent. Common unit - grams / 100 m. L Factors affecting solubility Temperature Pressure Polarity

A saturated solution contains the maximum amount of a solute that will dissolve in a given solvent at a specific temperature. An unsaturated solution contains less solute than the solvent has the capacity to dissolve at a specific temperature. A supersaturated solution contains more solute than is present in a saturated solution at a specific temperature. Sodium acetate crystals rapidly form when a seed crystal is added to a supersaturated solution of sodium acetate. 130

How much stuff is in a mixture? • Molarity (M) moles per liter solution • Normality (N) equivalents per liter solution – 1 M H 2 SO 4 has 2 X the H+ than 1 M HCl – It has a normality of 2 N vs 1 N for 1 M HCL • Molality (m) moles per kilogram solvent • Mole Fraction (X) is ratio of moles to total moles

Concentration Units The concentration of a solution is the amount of solute present in a given quantity of solvent or solution. Percent by Mass mass of solute % by mass = mass of solute + mass of solvent x 100% mass of solute x 100% = mass of solution Mole Fraction (X) moles of A XA = sum of moles of all components 132

Concentration Units Continued Molarity (M) moles of solute M = liters of solution Molality (m) m = moles of solute mass of solvent (kg) 133

In practice we often make a “stock” solution of a chemical and dilute it to a desired level A solution is prepared by diluting 30. 00 m. L of a 0. 400 M solution of Ca. Cl 2 to a final volume of 0. 500 L. What is the final concentration of [Ca. Cl 2] in this solution? What is the final concentration of [Ca+2]? What is the final concentration of [Cl-1]? . 0300 L x 0. 400 M =. 012 moles 0. 500 L [Ca+2] = 0. 024 M [Cl-1] = 2 x 0. 024 M = 0. 048 M =. 024 molar Ca. Cl 2

What is the molality of a 5. 86 M ethanol (C 2 H 5 OH) solution whose density is 0. 927 g/m. L? m = moles of solute mass of solvent (kg) M = moles of solute liters of solution Assume 1 L of solution: 5. 86 moles ethanol = 270 g ethanol 927 g of solution (1000 m. L x 0. 927 g/m. L) mass of solvent = mass of solution – mass of solute = 927 g – 270 g = 657 g = 0. 657 kg m = moles of solute mass of solvent (kg) = 5. 86 moles C 2 H 5 OH 0. 657 kg solvent = 8. 92 m 135

What is the molality of a 5. 86 M ethanol (C 2 H 5 OH) solution whose density is 0. 927 g/m. L? m = moles of solute mass of solvent (kg) M = moles of solute liters of solution Assume 1 L of solution: 5. 86 moles ethanol = 270 g ethanol 927 g of solution (1000 m. L x 0. 927 g/m. L) mass of solvent = mass of solution – mass of solute = 927 g – 270 g = 657 g = 0. 657 kg m = moles of solute mass of solvent (kg) = 5. 86 moles C 2 H 5 OH 0. 657 kg solvent = 8. 92 m 136

Calculate molarity, molality, mole fraction Concentrated HCl has a density of 1. 19 g/ml and is 38% HCl in water (mass percent) Molarity 1000 m. L of solution has a mass of 1190 g. 38 x 1190 = 452. 2 g HCl 452. 2 g x 1 mole = 12. 4 moles 36. 4 g HCl 12. 4 moles in one liter = 12. 4 molar Molality In one liter 12. 4 moles HCl in 737. 8 g Or 0. 7378 Kg of water 12. 4 mole = 16. 8 m. 7378 Kg Mole Fraction In one liter of solution there is 452. 2 g HCl = 12. 4 moles In one liter there is 1190 – 452. 2 g = 737. 8 g water = 41. 0 moles water X HCl = 12. 4 / (12. 4 + 41. 0) x 100 = 23. 2 %

Energy of Solutions • Remember lattice energies? What are they? E = k (Q 1 Q 2) r • When an ionic solid dissolves in water, the lattice energy is overcome. How? – The water surrounds the ions and hydrates them – The water has to get in between the ions or – The ions have to get in between the water molecules

Three Steps to Dissolution • Step 1 – Separate the solute into individual components – Expanding the solute • Step 2 – Overcoming the intermolecular forces in the solvent to make room for the solute – Expanding the solvent • Step 3 – Allowing the solute and solvent to interact to form the solution

Three types of interactions in the solution process: • solvent-solvent interaction • solute-solute interaction • solvent-solute interaction Molecular view of the formation of solution Hsoln = H 1 + H 2 + H 3 140

Heat of Solution Exothermic Endothermic

Heat of Solution Depends on… P Solv P Solu P Solv NP Solu NP Solv NP Solu H 1 H 2 H 3 Hsol Outcome Large Small Solution forms Small Large Neg Small Large No positive solution Small Large No positive solution Solution forms

Factors Affecting Solubility • The structure of a compound determines what it will dissolve • If it is non-polar, lots of C – H bonds, then it will dissolve in non-polar solvents. • If it is polar or ionic, it will dissolve in polar solvents • The polarity of solvent can be measured by its dielectric constant. The higher the constant the more polar it is

Fat Soluble Chemicals • Fat soluble vitamins A, D, E and K. These are nonpolar structures. They can be stored in fat, because fat is non-polar. – If you eat too much of these, you can get sick because they accumulate in fatty tissue. • DDT, the insecticide, is fat soluble. It is in all our bodies, even if it has not been sprayed in the US since the 70’s. – DDT bioacculmulates. As you move up the food chain, the animals store the DDT in their fat. When they are eaten, all the DDT goes to the predator, who then stores it in his fat. As you move up the food chain, there is a greater accumulation of DDT.

Water Soluble Vitamins • Some vitamins are water soluble. – They are excreted. – If we do not replenish them, they lower their concentration quickly. • Vitamin C is a good example – British navy called Limies because they brought limes – Without the water soluble vitamin C, they were prone to getting scurvy.

Why did the white bear dissolve in water? Because it was polar.

Pressure and Solubility of Gases The solubility of a gas in a liquid is proportional to the pressure of the gas over the solution (Henry’s law). c is the concentration (M) of the dissolved gas c = k. P P is the pressure of the gas over the solution k is a constant for each gas (mol/L • atm) that depends only on temperature low P high P low c high c 147

Henry’s Law • The partial pressure of a gas above a solution is proportional to its concentration P=k. C P = partial pressure in atm k = constant = L • atm mol C = molarity of solution (moles/liter) • Explain using KMT

What is concentration of CO 2 in a soda if the partial pressure of above the soda is CO 2 is 5. 0 atm? • CCO 2 = PCO 2 k. CO 2 = 5 atm = 0. 16 mol 32 L atm/mol L

Temperature Effects • Solids – Dissolve faster at higher temperatures – Many solids have a higher solubility at higher temperatures • Not all. Many sulfates do not • The only way to find out is to measure/experiment • Gases – Less soluble at higher temps – Explain use KMT

Water • As the temperature increases, the levels of dissolved gases lowers • Oxygen is an important gas for aquatic life – Dissolved oxygen (DO) can be measured – Warmer water holds less oxygen – Fish need the oxygen – Warm water kills certain types of fish

Solubility (g solute/100 g H 2 O) Notice at 90 C there is more solute than solvent! There is almost no difference In cold and hot solubility This solid has a lower solubility In hot water than cold Temperature ºC

Temperature and Solubility Solid solubility and temperature solubility increases with increasing temperature solubility decreases with increasing temperature 153

Fractional crystallization is the separation of a mixture of substances into pure components on the basis of their differing solubilities. Suppose you have 90 g KNO 3 contaminated with 10 g Na. Cl. Fractional crystallization: 1. Dissolve sample in 100 m. L of water at 600 C 2. Cool solution to 00 C 3. All Na. Cl will stay in solution (s = 34. 2 g/100 g) 4. 78 g of PURE KNO 3 will precipitate (s = 12 g/100 g). 90 g – 12 g = 78 g 154
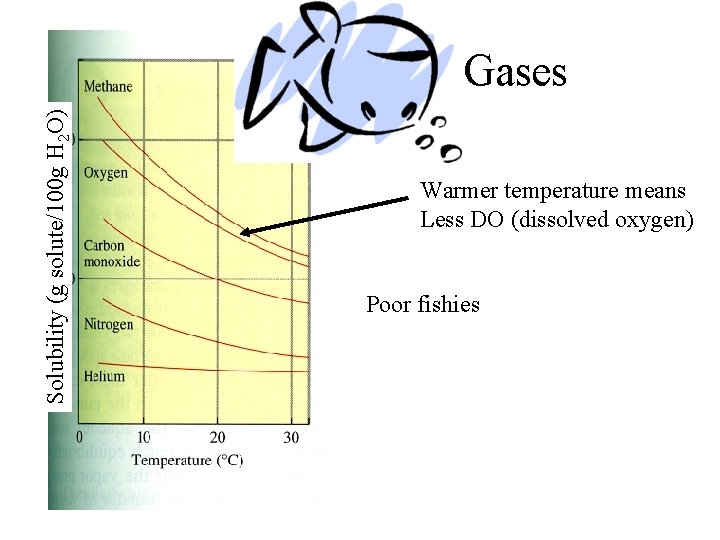
Solubility (g solute/100 g H 2 O) Gases Warmer temperature means Less DO (dissolved oxygen) Poor fishies

Pressure and solubility of gases • Increasing the pressure of a gas above a liquid increases the concentration of the gas. • This shifts the equilibrium, driving more gas into the liquid.

Pressure and solubility of gases cg = kpgas This law is accurate to within 1 -3% for slightly soluble gases and pressures up to one atmosphere. Solubility (g/100 g water) Henry’s Law At constant temperature, the solubility of a gas is directly proportional to the pressure of the gas above the solution. 0. 010 O 2 0. 005 N 2 He 0. 000 0 1 Pressure (atm) 2

Solubility of some substances Substance Temperature o. C Solubility g/100 m. L water Na. Cl (s) 100 39. 12 Pb. Cl 2 (s) 100 3. 34 Ag. Cl (s) 100 0. 00021 CH 3 CH 2 OH (l) 0 -100 infinity CH 3 CH 2 OCH 2 CH 3 (l) 15 8. 43 O 2 (g) 60 0. 0023 CO 2 (g) 40 0. 097 SO 2 (g) 40 5. 41

Saturation • When a solution contains as much solute as it can at a given temperature. • Unsaturated - Can still dissolve more. • Saturated - Have dissolved all you can. • Supersaturated - Temporarily have dissolved too much. • Precipitate - Excess solute that falls out of solution.

Saturated Solutions • At saturation, the solute is in dynamic equilibrium. The concentration is constant. • • • Solute species are constantly in motion, moving in and out of solution.

Properties of� aqueous solutions • There are two general classes of solutes. • Electrolytic • ionic compounds in polar solvents • dissociate in solution to make ions • conduct electricity • may be strong (100% dissociation) or weak (less than 10%, ) • Nonelectrolytic • do not conduct electricity • solute is dispersed but does not dissociate

Colligative properties “Bulk” properties that change when you add a solute to make a solution. • Based on how much you add but not what the solute • is. Effect of electrolytes is based on number of ions produced. Colligative properties • vapor pressure lowering • freezing point depression • boiling point elevation • osmotic pressure

Colligative Properties Lowering the Vapor Pressure

Vapor pressure lowering The introduction of a nonvolatile solute will reduce the vapor pressure of the solvent in the resulting solution. • The vapor pressure of a nonvolatile component is essentially zero. • It does not contribute to the vapor pressure of the solution. • However, the solution’s vapor pressure is dependent on the solute mole fraction.

Colligative Properties Raoult’s Law The partial pressure exerted by solvent vapor above a solution, PA, equals the mole fraction of the solvent in the solution, A , times the vapor pressure of the pure solvent, PA. Recall Dalton’s Law:

Colligative Properties Solute’s Effect on Phase Diagram

Boiling point elevation • When you add a nonvolatile solute to a solvent, the boiling point goes up. This is because the vapor pressure has been lowered. Tbp = Kbp x molality • The boiling point will continue to be elevated as you add more solute until you reach saturation. Examples Cooking pasta in salt water Antifreeze

Freezing point depression • When you add a solute to a solvent, the freezing point goes down. Tfp = Kfp x molality • The more you add, the lower it gets. • This will only work until you reach saturation. Examples “Salting” roads in winter Making ice cream antifreeze

Ionic vs. covalent substances Ionic substances have a greater effect per mole than covalent. • 1 mol/kg of water for glucose = 1 molal • 1 mol/kg of water for Na. Cl = 2 molal ions • 1 mol/kg of water for Ca. Cl 2 = 3 molal ions Effects are based on the number of particles! Tbp or fp = i. Kbp or fp x molality Where i is the van’t Hoff factor that compares the measured ∆Tbp or fp / calculated ∆Tbp or fp as nonelectrolyte

Ionic vs. covalent substances The ideal van’t Hoff factor for Na. Cl is 2, because it consists of 1 mole Na 1+ ions and 1 mole Cl 1 - ions. Oppositely charged ions in solution collide and briefly stick together as one particle. This lowers the ideal van’t Hoff factor. The more dilute a solution is and the lower the charges of the ions formed, the closer the value of i is to the ideal van’t Hoff factor.

Osmosis • The movement of a solvent through a semipermeable membrane from a dilute solution to a more concentrated one. • Semipermeable membranes, membranes such as cell walls, only allow small molecules and ions to go through.

Osmosis Eventually the pressure difference between the arms stops osmosis.

Osmotic pressure The pressure required to stop osmosis. osmotic pressure ( ) = i. MRT i = van’t Hoff factor M = molar concentration T = temperature in Kelvin R = gas law constant Since molarity is moles/liter, this equation is just a modified form of the gas law equation. n. RT V

Osmotic Pressure Three conditions can exist for living cells. Concentration is the same on both sides. – isotonic Concentration is greater on the inside. – – hypertonic cell hypotonic solution Concentration is greater on the outside. – – hypotonic cell hypertonic solution

Cell in isotonic solution A red blood cell and plasma have the same osmotic pressure.

Cells in hypertonic solution If the level of salt in the plasma is too high, the cell collapses. Crenation - water is drawn out of the cell.

Cells in hypotonic solution If the level of salt in the plasma is too low, the cell swells and ruptures. Hemolysis - water is drawn into the cell.

Dialysis • The process where solvent and other small molecules can pass through a membrane. • Similar to osmosis but the ‘holes’ in the membrane are larger. As a result, even hydrated ions can pass through. • The method relies on: diffusion osmosis ultrafiltration

Dialysis By passing large amounts of a pure solvent past the membrane, we can flush out all but the largest components. pure water in water, ions and small molecule out

Colloids Homogeneous mixtures of two or more substances which are not solutions. The substances are present as larger particles than those found in solution. Dispersing medium - The substance in a colloid found in the greater extent. Dispersed phase - The substance found in the lesser extent.

Colloids • For solutions, ions and molecules have a size of about 10 -7 cm. • In colloids, the particles are larger, with sizes from 10 -7 to 10 -5 cm. • The colloidal particles are still too small to settle out of solution due to gravity. • There are several types of colloids depending on the physical state of the dispersing medium and the dispersed phase.
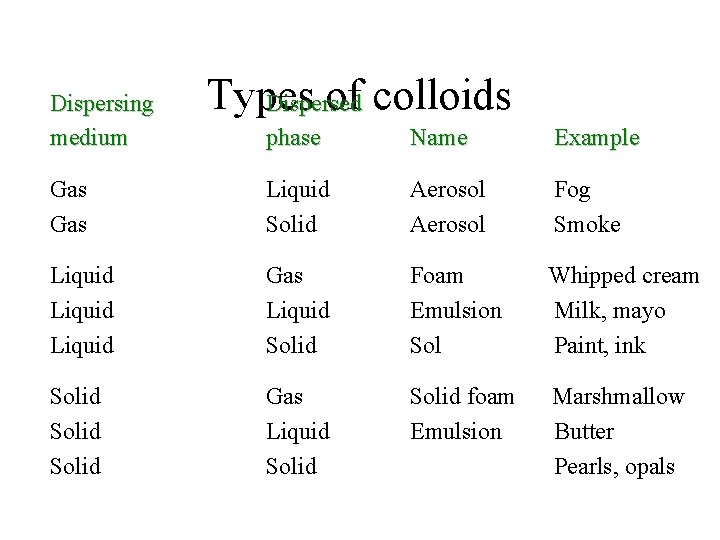
Dispersing medium Types of colloids Dispersed phase Name Example Gas Liquid Solid Aerosol Fog Smoke Liquid Gas Liquid Solid Foam Emulsion Sol Whipped cream Milk, mayo Paint, ink Solid Gas Liquid Solid foam Emulsion Marshmallow Butter Pearls, opals

Colloids • Tyndall effect - the ability of a colloid to scatter light. A beam of light can be seen passing through a colloid.

Tyndall Effect Light is scattered by the colloidal-sized particles.

Colloids • Hydrophilic and Hydrophobic Colloids • Many times water is the dispersing medium in colloids. The dispersed phase can be either: • “Water loving” colloids: hydrophilic. • • “Water hating” colloids: hydrophobic.

Colloids • Hydrophilic and Hydrophobic Colloids • Molecules arrange themselves so that hydrophobic portions are oriented towards each other. • If a large hydrophobic macromolecule (giant molecule) needs to exist in water (ex. proteins), hydrophobic portions embed themselves into the macromolecule leaving the hydrophilic ends to interact with water.
Colloids • Hydrophilic and Hydrophobic Colloids

Colloids • Hydrophilic and Hydrophobic Colloids • Typical hydrophilic groups are polar (containing C-O, O-H, N-H bonds) or charged. • Hydrophobic colloids need to be stabilized in water by adding a surfactant that reduces the water’s surface tension and permits mixing to occur.

Colloids • Hydrophilic and Hydrophobic Colloids • Adsorption: when something sticks to a surface we say that it is adsorbed. • If ions are adsorbed onto the surface of a colloid, the colloids appears hydrophilic and is stabilized in water. • Consider a small drop of oil in water. • Add to the water sodium stearate.

Colloids • Hydrophilic and Hydrophobic Colloids

Colloids • Hydrophilic and Hydrophobic Colloids • Sodium stearate has a long hydrophobic tail (CH 3(CH 2)16 -) and a small hydrophilic head (CO 2 -Na+). • The hydrophobic tail can be absorbed into the oil drop, leaving the hydrophilic head on the surface. • The hydrophilic heads then interact with the water and the oil drop is stabilized in water.

Colloids • Hydrophilic and Hydrophobic Colloids

Colloids • Hydrophilic and Hydrophobic Colloids • Most dirt stains on people and clothing are oil-based. Soaps are molecules with long hydrophobic tails and hydrophilic heads that remove dirt by stabilizing the colloid in water. • Bile excretes substances like sodium stereate that forms an emulsion with fats in our small intestine. • Emulsifying agents help form an emulsion.

Colloids • Removal of Colloidal Particles • Colloid particles are too small to be separated by physical means (e. g. filtration). • Colloid particles are coagulated (enlarged) until they can be removed by filtration.

Colloids • Removal of Colloidal Particles • Methods of coagulation: – heating (colloid particles move and are attracted to each other when they collide); – adding an electrolyte (neutralize the surface charges on the colloid particles). • Dialysis: using a semipermeable membranes separate ions from colloidal particles.

Suspension • In a suspension, the particles temporarily remain mixed because of collisions with the much smaller particles of the solvent. They appear to move in a zig-zag pattern, called Brownian Motion • In suspensions, the particles are larger than 10 -4 cm, which can be viewed under a microscope. • The suspended particles will eventually settle out of the mixture due to gravity.
- Slides: 196