Gas Laws Honors Chemistry Hannah Moore Gases Compressible

Gas Laws Honors Chemistry Hannah Moore

Gases � Compressible ◦ Sparse � Pressure ◦ ◦ – force per unit area mm. Hg or Torr Atmospheres (atm) 760 mm. Hg = 1 atm = 760 Torr Pascals – very small unit of measure � 1 atm = 101. 3 k. Pa

Conversion of Units � How many mm. Hg is 1. 25 atm? � How many Pascals are in 622 mm. Hg?

Gas Laws � Boyle’s Law Pressure ◦ Volume of an ideal gas varies inversely with pressure when under a constant Temperature Volume

Boyle’s Law � Useful �A form (if # moles and T are constant): balloon has a pressure of 1. 00 atm and a volume of 2. 00 L. How far must the pressure be reduced for the volume to change to 4. 00 L?

Gas Laws � Charles’ Law ◦ The volume of an ideal gas varies directly with the Kelvin Temperature when under a constant Pressure Volume(L) V=k. T y=mx m=k Temperature (K)

Charles’ Law � Useful �A Form (constant # moles and P): Helium balloon has a volume of 12. 5 L at 25 °C. What is its volume at 55 °C?

Gas Laws � Avogadro’s Law ◦ The Volume of an ideal gas varies directly with its number of moles when under constant Temperature and Pressure.

Avogadro’s Law � Useful form (constant P & T) � Consider a balloon which has a volume of 6. 5 L and contains 3. 2 g of He. If an additional 1. 5 g of He are added, what is the volume?

Gas Laws � 4 ◦ ◦ Variables involved in relationships Pressure (P) Volume (V) Temperature (T) Moles of gas (n) � Ideal Gas Law ◦ Property of gases above 0°C and near 1 atm
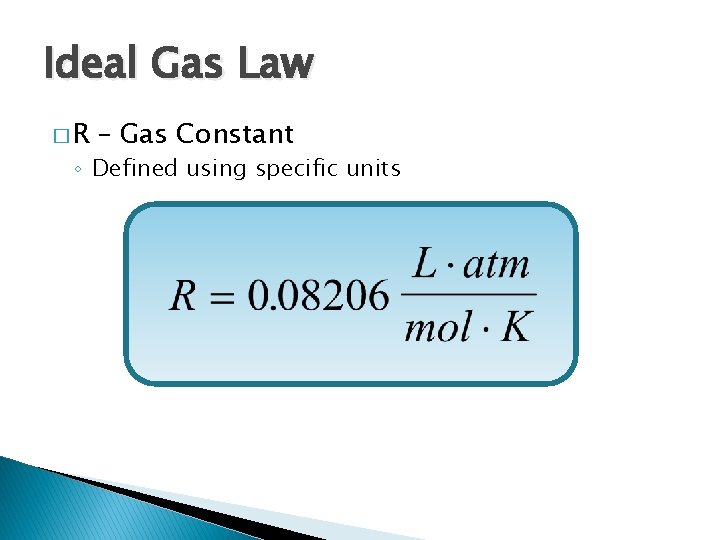
Ideal Gas Law �R – Gas Constant ◦ Defined using specific units

STP � Standard Temperature and Pressure ◦ 0°C and 1 atm

Ideal Gas Law � What is the pressure of 1. 45 mol of a gas if the volume is 20. 0 L and the Temperature is 25. 0°C?

Ideal Gas Law � What volume is occupied by 11. 0 g of CO 2 at 25 °C and 371 Torr?

Ideal Gas Law �A gas has a mass of 3. 20 g and occupies 2. 00 L at 17. 0°C and 380. Torr. What is its Molar Mass? ◦ V=2. 00 L ◦ T=290. 2 K ◦ P=0. 500 atm

Ideal Gas Law � What � 1 is the volume of 1 mol of a gas at STP? mole of an ideal gas occupies 22. 41 Liters @ STP

Molar Volume � What is the density of Fluorine gas at STP?
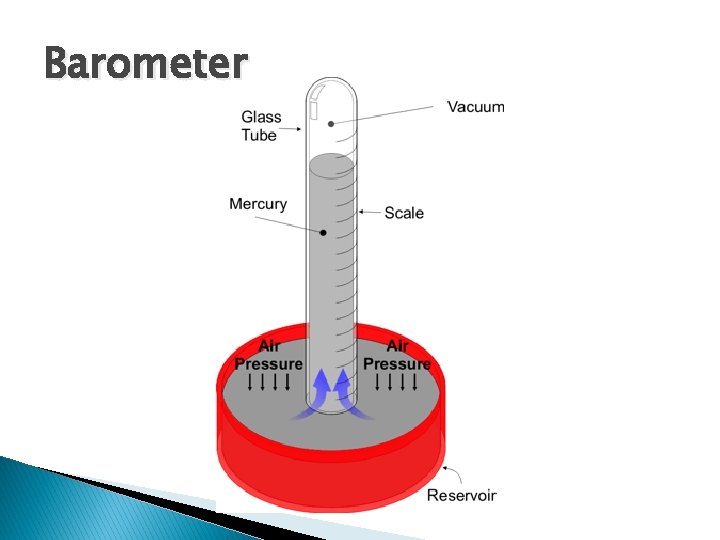
Barometer

Gas Stoichiometry � What volume of Hydrogen gas is produced in the reaction of 25. 0 g of Zinc with HCl(aq) at STP?

Gas Stoichiometry � Consider the combustion reaction of Benzene (C 6 H 6). If 20. 0 L of O 2 is provided at 22 °C and 5. 00 atm, how much Benzene will be consumed?

Gas Stoichiometry �A 4. 00 L vessel is filled with 1. 00 atm of CH 4 and 4. 00 atm of O 2 at 300 °C. A spark causes the complete combustion. What mass of CO 2 is produced?

Dalton’s Law � Gas Laws apply to all pure gases � Gas Laws apply independently in a mixture ◦ Partial Pressure � Dalton’s Law of Partial Pressures

Dalton’s Law � Each gas acts independent of the other gases ◦ No interaction between gases � All gases in a mixture are under identical conditions

Dalton’s Law � Fraction of moles in a gas mixture is the same as the fraction of pressures

Dalton’s Law �A sample of KCl. O 3 is decomposed. The Oxygen gas is collected over water at 22 °C (Water has a vapor pressure of 21 Torr). The total pressure is 754 Torr, determine the oxygen pressure.

Dalton’s Law � What are the partial pressures of a mixture of 12. 45 g H 2, 60. 67 g N 2 and 2. 38 g NH 3 at 25. 5°C in a 2. 50 L container?

Dalton’s Law �A scuba diver’s tank contains 300. g of Helium and 1600. g of Oxygen. If the total pressure in the tank is 8. 0 atm, what is the partial pressure of each gas?
Kinetic Molecular Theory �A gas consists of small particles in continuous random motion

Kinetic Molecular Theory � The volume of the individual gases is negligible relative to the total volume ◦ Most of the gas is empty space
Kinetic Molecular Theory � Gases collide with each other and the walls of the container; All collisions are elastic

Kinetic Molecular Theory � Gas particles are independent of each other and their attractive forces are negligible ◦ Liquids and solids form due to attractive forces between atoms, ions and molecules

Kinetic Molecular Theory � The average kinetic energy of a gas is directly proportional to its Temperature ◦ Temperature causes the atoms/molecules in a gas to move Effects of temperature are seen in the velocity of the particles

Kinetic Molecular Theory �A gas consists of small particles in continuous random motion � The volume of the individual gases is negligible relative to the total volume � Gases collide with each other and the walls of the container; All collisions are elastic � Gas particles are independent of each other and their attractive forces are negligible � The average kinetic energy of a gas is directly proportional to its Temperature
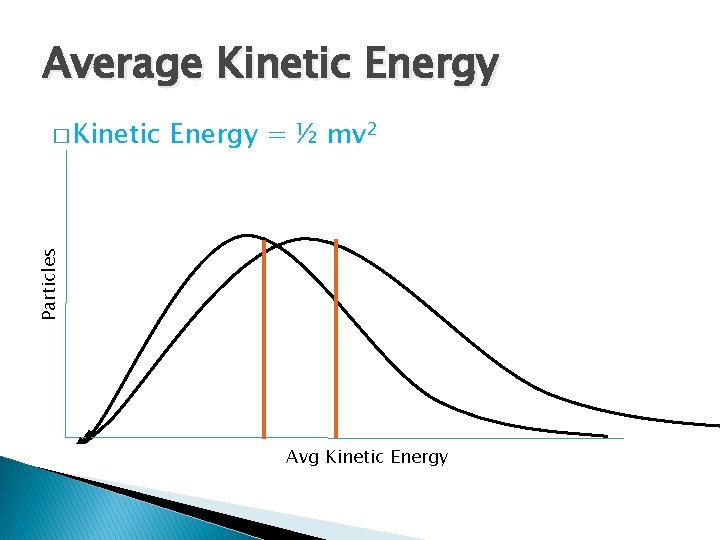
Average Kinetic Energy = ½ mv 2 Particles � Kinetic Avg Kinetic Energy

Real Gases � Two major assumptions ◦ Gases have no volume ◦ Gases have no attractive forces � Fails ◦ High Pressure �Volume becomes important �Attractive forces important �Gases Become Liquid ◦ Low Temperature �Attractive forces important

Summary � Ideal Gas Law ◦ PV=n. RT � Dalton’s Law ◦ Total Pressure is the sum of the partial pressures � Kinetic Molecular Theory ◦ Gas particles are moving, have no volume or attractive forces and increase speed with T ◦ Average velocity of molecules in a gas is important � Gas Stoichiometry ◦ Moles can be determined from gas variables
- Slides: 36