Gas Laws Dalton Boyle Before starting l Gases

Gas Laws Dalton & Boyle

Before starting…. l Gases behave in different ways & obey “laws” l But when we are discussing gases we assume that we are dealing with an ideal gas. l An ideal gas follows the gas laws at all conditions of temperature & pressure l There are however exceptions!

Dalton’s Law of Partial Pressure: l At constant vol & temp, the total pressure exerted by a mixture of gases is equal to the sum of the partial pressures l PTotal = P 1 + P 2 + P 3…. . = + + Units: mm. Hg or atm or k. Pa 760 mm. Hg = 1 atm = 101. 3 k. Pa
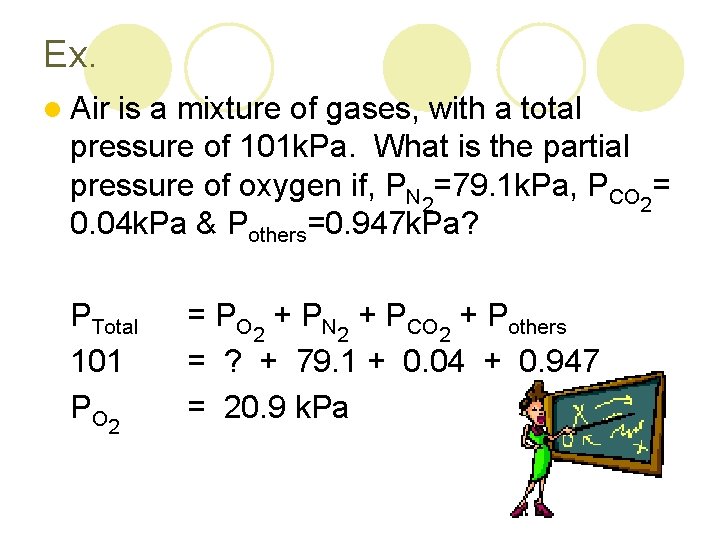
Ex. l Air is a mixture of gases, with a total pressure of 101 k. Pa. What is the partial pressure of oxygen if, PN 2=79. 1 k. Pa, PCO 2= 0. 04 k. Pa & Pothers=0. 947 k. Pa? PTotal 101 P O 2 = PO 2 + PN 2 + PCO 2 + Pothers = ? + 79. 1 + 0. 04 + 0. 947 = 20. 9 k. Pa

Try #3 pg 225 PTotal = PO 2 + PN 2 + PHe PTotal = 20 + 46. 7 + 26. 7 PTotal = 93. 4 k. Pa

Boyle’s Law: l For a given mass of a gas at a constant temp, the volume of a gas varies inversely with pressure P 1 V 1 = P 2 V 2 l So… ¡ if volume decreases, then pressure increases ¡ if volume increases, then pressure decreases
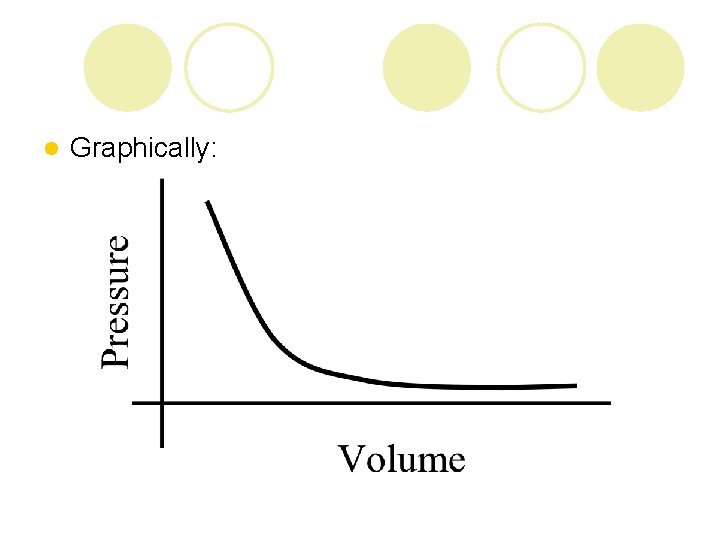
l Graphically:

Ex. l. A balloon is filled with 30 L of He gas at 100 k. Pa. What is the volume when the balloon rises to an altitude where the pressure is 25 k. Pa? P 1 V 1 = P 2 V 2 (100 k. Pa)(30 L) = (25 k. Pa)(V 2) V 2 = (100 k. Pa)(30 L) (25 k. Pa) V 2 = 120 L

Ex. l The pressure on 2. 50 L of anesthetic gas is changed from 100 k. Pa to 40 k. Pa. What will be the new volume if the temperature remains constant? P 1 V 1 = P 2 V 2 (100 k. Pa)(2. 50 L) = (40 k. Pa)(V 2) V 2 = (100 k. Pa)(2. 50 L) (40 k. Pa) V 2 = 6. 25 L
Try: l pg 241 # 20, 21, 22
- Slides: 10