garnet Hornblende Plag garnet Metabasites Francis 2014 Metabasites

garnet Hornblende Plag garnet Metabasites Francis, 2014

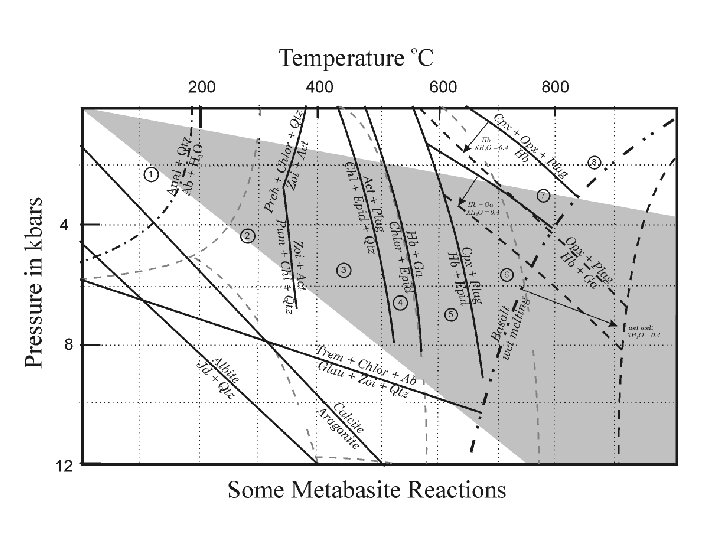
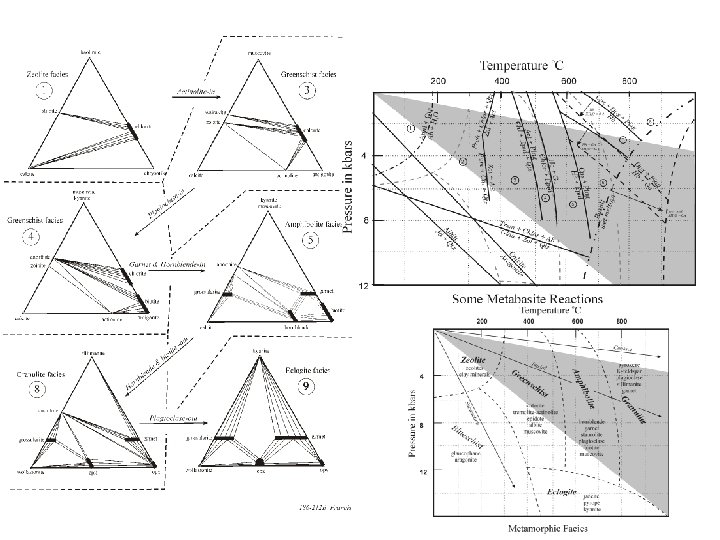
Metabasites: Mafic volcanics such as basalts andesites have more complicated and variable compositions than shales, containing significant quantities of Ca and Na, in addition to Si, Al, Mg Fe, K, and H 2 O. At least 8 components are necessary to describe such systems, and it is no longer possible to construct a simple projection scheme that is thermodynamically rigorous. The metamorphic mineral assemblages of mafic volcanic rocks are commonly portrayed in an ACF projection, but it should be remembered that, unlike the metapelite AFM diagram, 3 phase regions in the ACF diagram are not strictly invariant and crossing tie lines are not uncommon.

Zeolite Facies The beginning of metamorphism in volcanic rocks and volcanogenic sediments is marked by the development of zeolites in vesicles and fractures under conditions of shallow burial. In consequence, volcanic rocks change from being vesicular to amygdular. Apart from the filling of vesicules and void spaces with these secondary minerals, rocks in the zeolite facies look essentially unmetamorphosed, although they often appear somewhat weathered in hand specimen: dirty, brownish, and oxidized because the processes of weathering have continued into the zeolite facies. Massive samples may still be quite pristine, however, retaining their original igneous mineralogy, especially if they have been isolated from extensive weathering and are relatively dry. General Zeolite formula: Ca Zeolites Low T Chabazite Ca. Al 2 Si 4 O 12. 6 H 2 O Wn. Tm. O 2 m. s. H 2 O W = Na, Ca, K, (Ba, Sr, …) T = Si, Al Na Zeolites Phillipsite Na 3 Al 3 Si 5 O 16. 6 H 2 O Stilbite Ca. Al 2 Si 7 O 18. 7 H 2 O High T Heulandite Ca. Al 2 Si 7 O 18. 6 H 2 O Analcime Na. Al. Si 2 O 6. H 2 O Laumontite Ca. Al 2 Si 4 O 12. 4 H 2 O Natrolite Na 4 Al 4 Si 6 O 20. 4 H 2 O Wairakite Ca. Al 2 Si 4 O 12. 2 H 2 O Albite - Feldspar Na. Al. Si 3 O 8
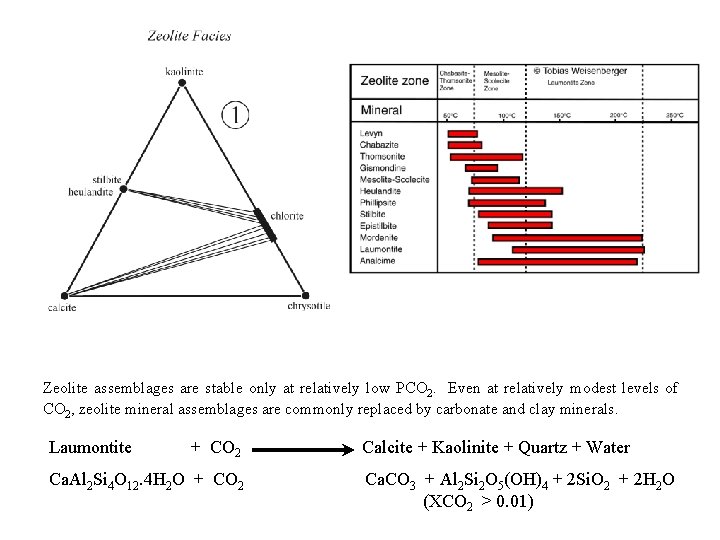
Zeolite assemblages are stable only at relatively low PCO 2. Even at relatively modest levels of CO 2, zeolite mineral assemblages are commonly replaced by carbonate and clay minerals. Laumontite + CO 2 Ca. Al 2 Si 4 O 12. 4 H 2 O + CO 2 Calcite + Kaolinite + Quartz + Water Ca. CO 3 + Al 2 Si 2 O 5(OH)4 + 2 Si. O 2 + 2 H 2 O (XCO 2 > 0. 01)

Prehnite - Pumpellyite sub-Facies The development of prehnite and pumpellyite in the both the groundmass and void spaces marks the beginning of the prehnite - pumpellyite facies in metavolcanic and metavolcanogenic sedimentary rocks. Volcanic rocks in this metamorphic facies begin to take on a greenish colour, although they typically are relatively unstrained and unrecrystallized, and commonly appear little metamorphosed in hand specimen, except for the development of a greenish colour. The presence of prehnite and pumpellyite are best recognized in thin section. Prehnite Ca 2 Al(Al. Si 3)O 10(OH)2 brittle mica Pumpellyite W 4 X(OH, O)Y 5 O(OH)3(TO 4)2(T 2 O 7)2. 2 H 2 O sorosilicate T = Si, Al Y = Al, Fe 3+, Ti 4+ X = Mn, Fe 2+, Mg, Al, Fe 3+ W = Ca, K, Na Lawsonite Ca. Al 2(OH)2 Si. O 2. H 2 O
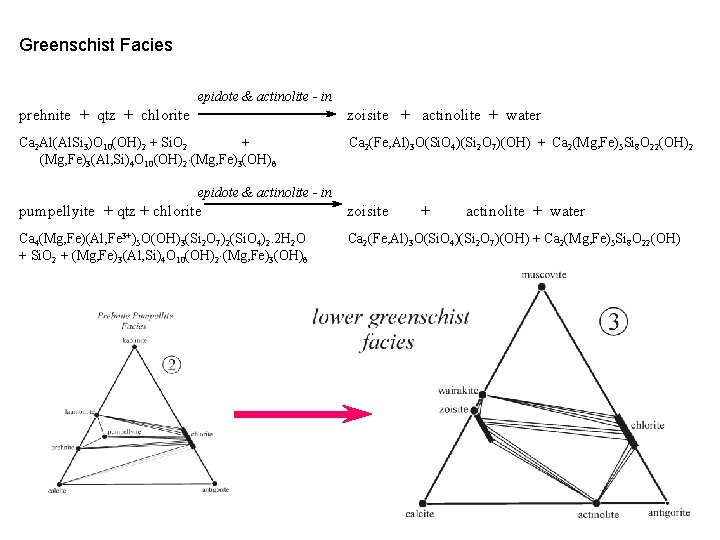
Greenschist Facies epidote & actinolite - in prehnite + qtz + chlorite zoisite + actinolite + water Ca 2 Al(Al. Si 3)O 10(OH)2 + Si. O 2 + (Mg, Fe)3(Al, Si)4 O 10(OH)2 (Mg, Fe)3(OH)6 Ca 2(Fe, Al)3 O(Si. O 4)(Si 2 O 7)(OH) + Ca 2(Mg, Fe)5 Si 8 O 22(OH)2 epidote & actinolite - in pumpellyite + qtz + chlorite zoisite + actinolite + water Ca 4(Mg, Fe)(Al, Fe 3+)5 O(OH)3(Si 2 O 7)2(Si. O 4)2. 2 H 2 O + Si. O 2 + (Mg, Fe)3(Al, Si)4 O 10(OH)2 (Mg, Fe)3(OH)6 Ca 2(Fe, Al)3 O(Si. O 4)(Si 2 O 7)(OH) + Ca 2(Mg, Fe)5 Si 8 O 22(OH)
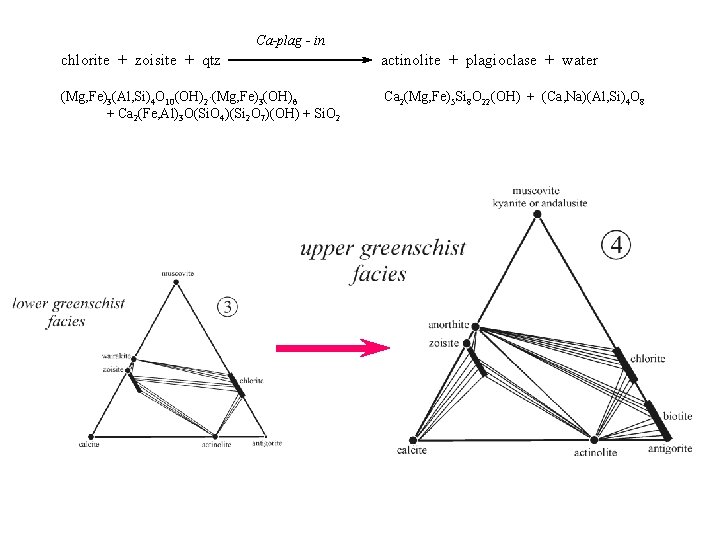
Ca-plag - in chlorite + zoisite + qtz actinolite + plagioclase + water (Mg, Fe)3(Al, Si)4 O 10(OH)2 (Mg, Fe)3(OH)6 + Ca 2(Fe, Al)3 O(Si. O 4)(Si 2 O 7)(OH) + Si. O 2 Ca 2(Mg, Fe)5 Si 8 O 22(OH) + (Ca, Na)(Al, Si)4 O 8
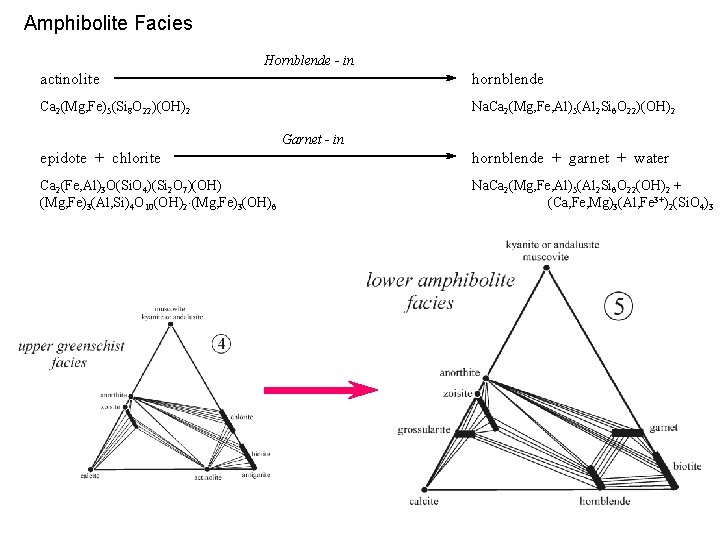
Amphibolite Facies Hornblende - in actinolite hornblende Ca 2(Mg, Fe)5(Si 8 O 22)(OH)2 Na. Ca 2(Mg, Fe, Al)5(Al 2 Si 6 O 22)(OH)2 Garnet - in epidote + chlorite hornblende + garnet + water Ca 2(Fe, Al)3 O(Si. O 4)(Si 2 O 7)(OH) (Mg, Fe)3(Al, Si)4 O 10(OH)2 (Mg, Fe)3(OH)6 Na. Ca 2(Mg, Fe, Al)5(Al 2 Si 6 O 22(OH)2 + (Ca, Fe, Mg)3(Al, Fe 3+)2(Si. O 4)3
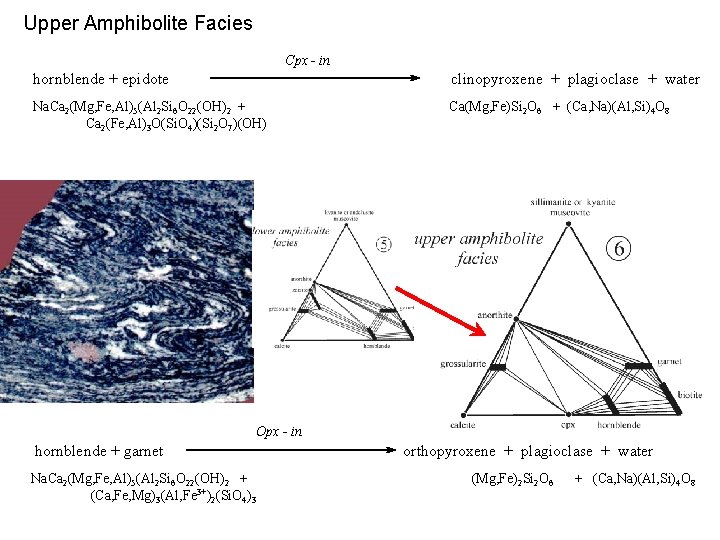
Upper Amphibolite Facies Cpx - in hornblende + epidote clinopyroxene + plagioclase + water Na. Ca 2(Mg, Fe, Al)5(Al 2 Si 6 O 22(OH)2 + Ca 2(Fe, Al)3 O(Si. O 4)(Si 2 O 7)(OH) Ca(Mg, Fe)Si 2 O 6 + (Ca, Na)(Al, Si)4 O 8 Opx - in hornblende + garnet Na. Ca 2(Mg, Fe, Al)5(Al 2 Si 6 O 22(OH)2 + (Ca, Fe, Mg)3(Al, Fe 3+)2(Si. O 4)3 orthopyroxene + plagioclase + water (Mg, Fe)2 Si 2 O 6 + (Ca, Na)(Al, Si)4 O 8
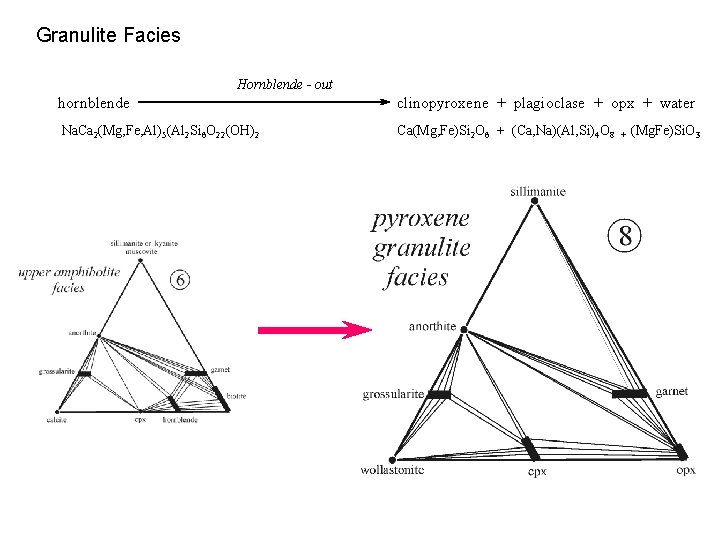
Granulite Facies Hornblende - out hornblende clinopyroxene + plagioclase + opx + water Na. Ca 2(Mg, Fe, Al)5(Al 2 Si 6 O 22(OH)2 Ca(Mg, Fe)Si 2 O 6 + (Ca, Na)(Al, Si)4 O 8 + (Mg. Fe)Si. O 3
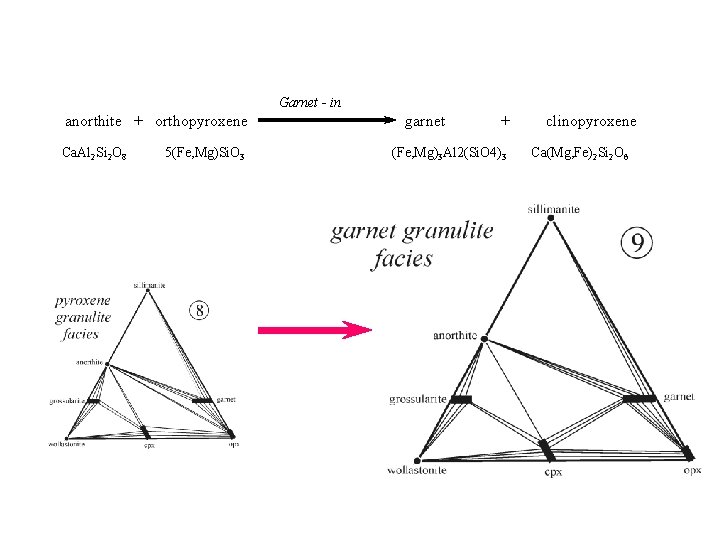
Garnet - in anorthite + orthopyroxene Ca. Al 2 Si 2 O 8 5(Fe, Mg)Si. O 3 garnet + (Fe, Mg)3 Al 2(Si. O 4)3 clinopyroxene Ca(Mg, Fe)2 Si 2 O 6
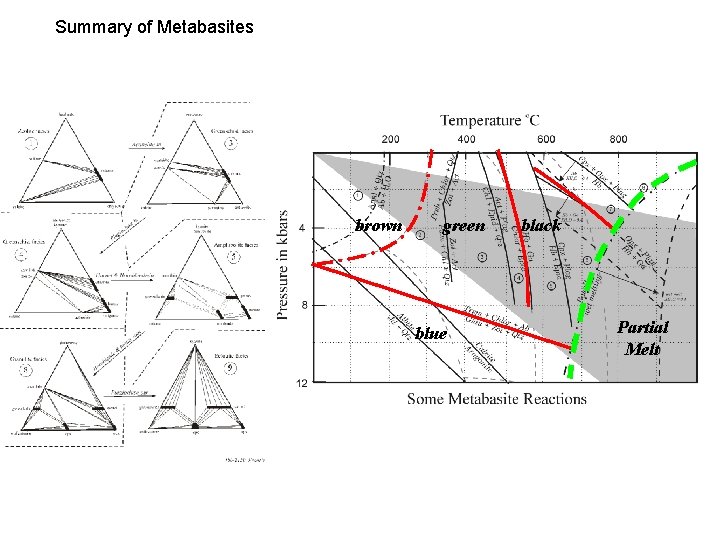
Summary of Metabasites brown green blue black Partial Melt
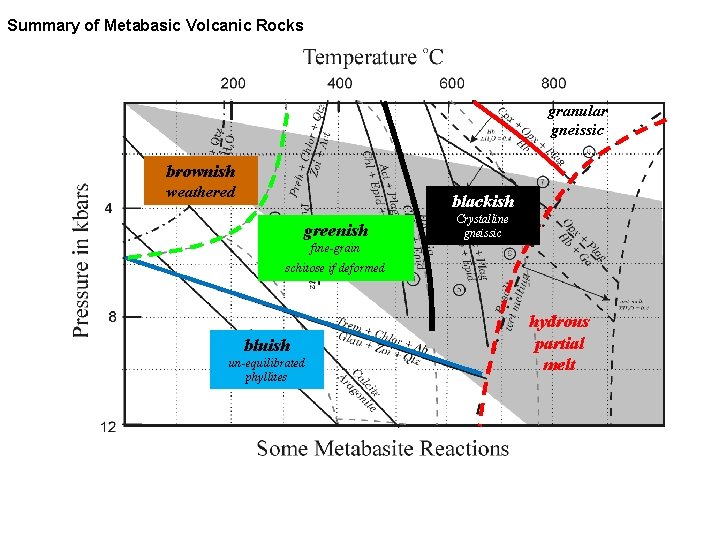
Summary of Metabasic Volcanic Rocks granular gneissic brownish weathered blackish greenish Crystalline gneissic fine-grain schitose if deformed bluish un-equilibrated phyllites hydrous partial melt
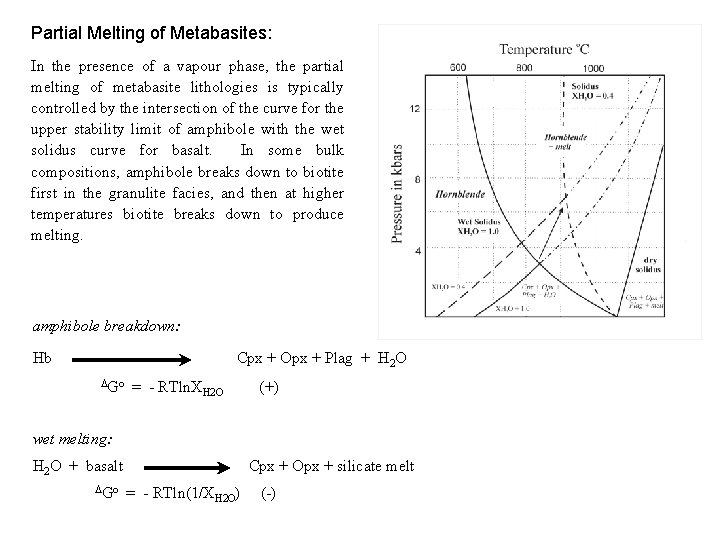
Partial Melting of Metabasites: In the presence of a vapour phase, the partial melting of metabasite lithologies is typically controlled by the intersection of the curve for the upper stability limit of amphibole with the wet solidus curve for basalt. In some bulk compositions, amphibole breaks down to biotite first in the granulite facies, and then at higher temperatures biotite breaks down to produce melting. amphibole breakdown: Hb Cpx + Opx + Plag + H 2 O Go = - RTln. XH 2 O (+) wet melting: H 2 O + basalt Go Cpx + Opx + silicate melt = - RTln(1/XH 2 O) (-)
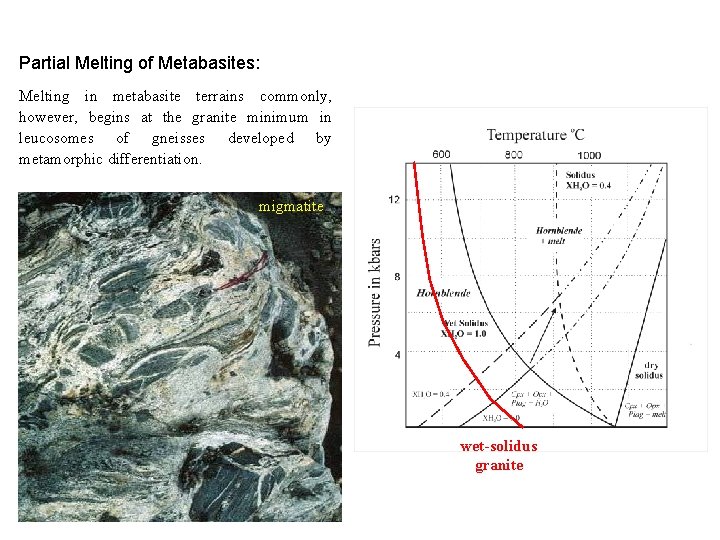
Partial Melting of Metabasites: Melting in metabasite terrains commonly, however, begins at the granite minimum in leucosomes of gneisses developed by metamorphic differentiation. migmatite wet-solidus granite
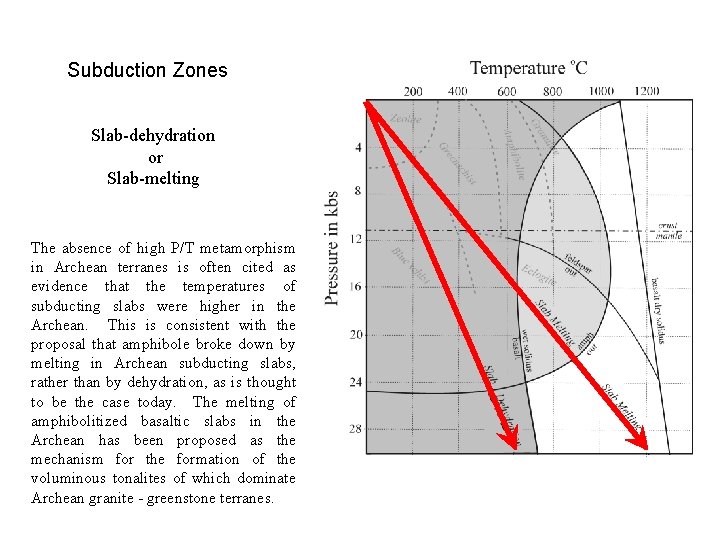
Subduction Zones Slab-dehydration or Slab-melting The absence of high P/T metamorphism in Archean terranes is often cited as evidence that the temperatures of subducting slabs were higher in the Archean. This is consistent with the proposal that amphibole broke down by melting in Archean subducting slabs, rather than by dehydration, as is thought to be the case today. The melting of amphibolitized basaltic slabs in the Archean has been proposed as the mechanism for the formation of the voluminous tonalites of which dominate Archean granite - greenstone terranes.
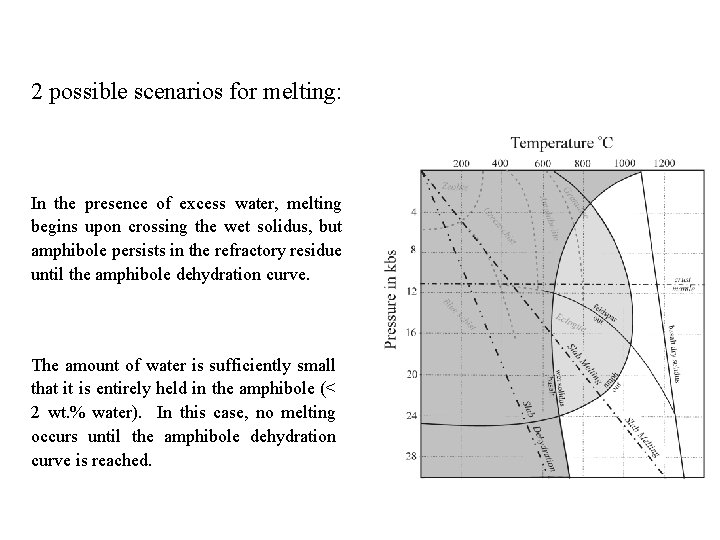
2 possible scenarios for melting: In the presence of excess water, melting begins upon crossing the wet solidus, but amphibole persists in the refractory residue until the amphibole dehydration curve. The amount of water is sufficiently small that it is entirely held in the amphibole (< 2 wt. % water). In this case, no melting occurs until the amphibole dehydration curve is reached.
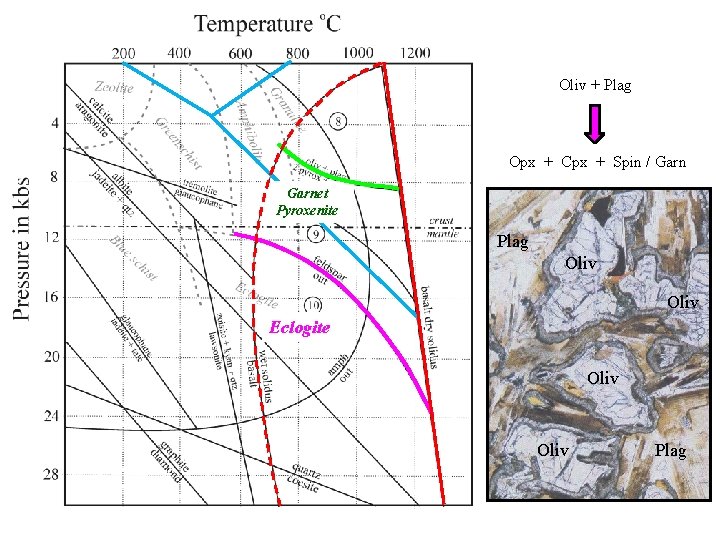
Oliv + Plag Opx + Cpx + Spin / Garnet Pyroxenite Plag Oliv Eclogite Oliv Plag

The Basalt - Eclogite Transition: Eclogites are high pressure rocks in which the low pressure plagioclase – pyroxene mineralogy of basalts and gabbros is converted to an assemblage of Jadeiite -rich clinopyroxene called omphacite and pyrope-rich garnet ± kyanite or quartz. Plagioclase is unstable and cannot be present. The complete transition from granulite to eclogite facies occurs over a 5 kb pressure range, beginning with the: • appearance of Garnet: Pyroxene Granulite Garnet Granulite and ending with: • disappearance of Plagioclase: Garnet Granulite Eclogite Does the basalt – eclogite transition correspond to the base of the crust (MOHO)? eclogite

MOHO Only two known rock types have the required density to match that inferred for the mantle underlying the MOHO. Eclogite ( = 3. 4 - 3. 6), a rock composed of clinopyroxene and garnet which has the same chemical composition as basalt, but a different mineralogy because it has crystallized at high pressure. Peridotite ( = 3. 2 - 3. 4), a rock consisting predominantly of olivine (60 - 80%), with lesser amounts of orthopyroxene, clinopyroxene, and spinel. The composition of peridotite is much richer in Mg and poorer in Al and Si than basalt, thus olivine (Y 2 TO 4) predominates over pyroxene (YTO 3) as the ferromagnesian mineral, and feldspar is minor or absent.
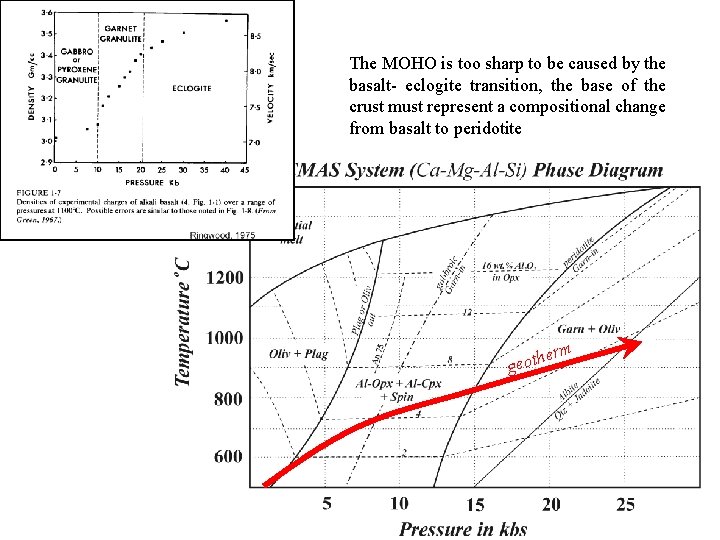
The MOHO is too sharp to be caused by the basalt- eclogite transition, the base of the crust must represent a compositional change from basalt to peridotite herm t o e g
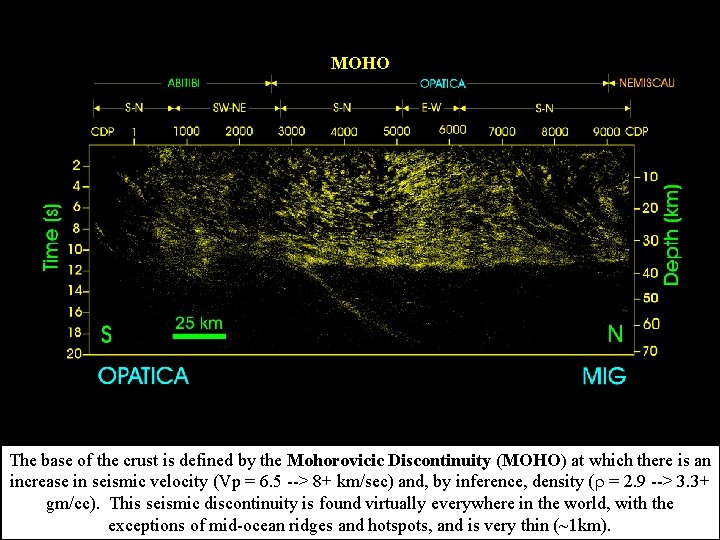
MOHO The base of the crust is defined by the Mohorovicic Discontinuity (MOHO) at which there is an increase in seismic velocity (Vp = 6. 5 --> 8+ km/sec) and, by inference, density ( = 2. 9 --> 3. 3+ gm/cc). This seismic discontinuity is found virtually everywhere in the world, with the exceptions of mid-ocean ridges and hotspots, and is very thin (~1 km).
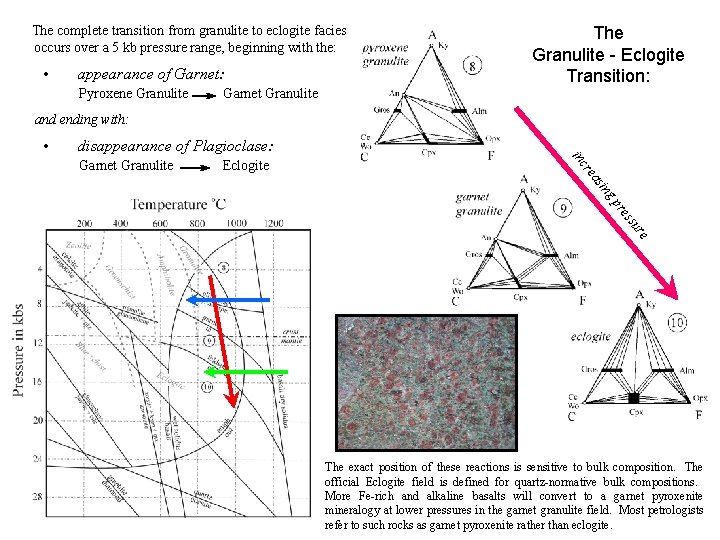
The complete transition from granulite to eclogite facies occurs over a 5 kb pressure range, beginning with the: • appearance of Garnet: Pyroxene Granulite Garnet Granulite The Granulite - Eclogite Transition: and ending with: • disappearance of Plagioclase: ing as re Eclogite inc Garnet Granulite e ur ss e pr The exact position of these reactions is sensitive to bulk composition. The official Eclogite field is defined for quartz-normative bulk compositions. More Fe-rich and alkaline basalts will convert to a garnet pyroxenite mineralogy at lower pressures in the garnet granulite field. Most petrologists refer to such rocks as garnet pyroxenite rather than eclogite.
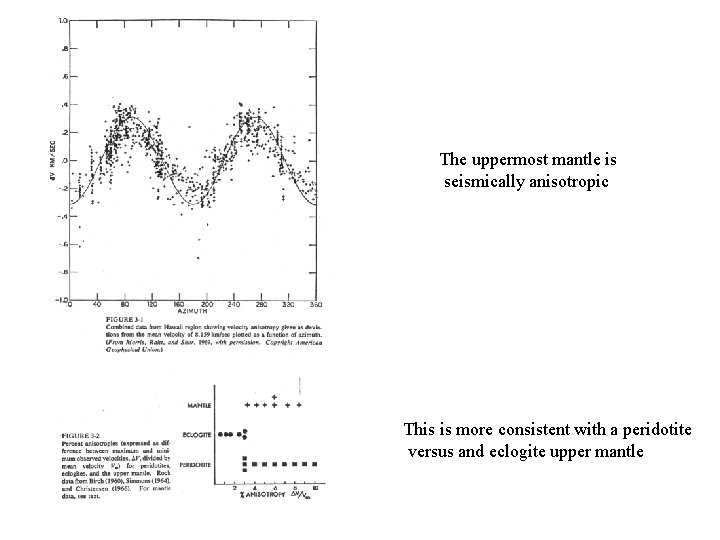
The uppermost mantle is seismically anisotropic This is more consistent with a peridotite versus and eclogite upper mantle
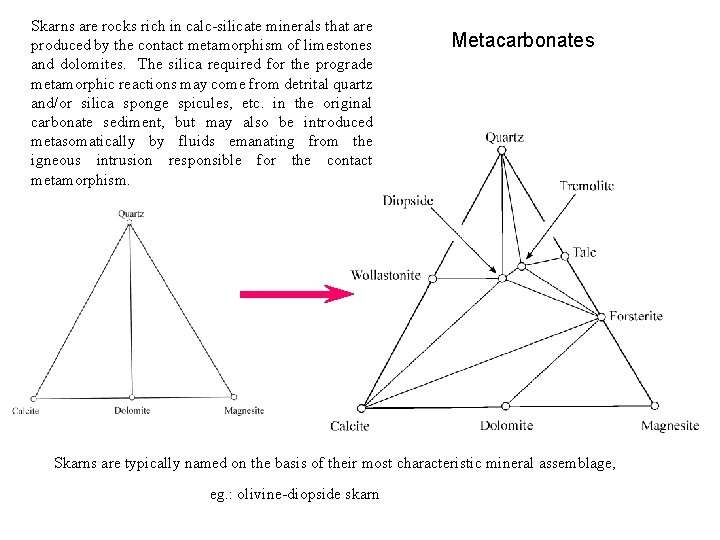
Skarns are rocks rich in calc-silicate minerals that are produced by the contact metamorphism of limestones and dolomites. The silica required for the prograde metamorphic reactions may come from detrital quartz and/or silica sponge spicules, etc. in the original carbonate sediment, but may also be introduced metasomatically by fluids emanating from the igneous intrusion responsible for the contact metamorphism. Metacarbonates Skarns are typically named on the basis of their most characteristic mineral assemblage, eg. : olivine-diopside skarn

Prograde Metacarbonate Reactions 3 Ca. Mg(CO 3)2 + 4 Si. O 2 + H 2 O Dolomite Talc - in Qtz Mg 3 Si 4 O 10(OH)2 + 3 Ca. CO 3 + 3 CO 2 Talc 5 Ca. Mg(CO 3)2 + 8 Si. O 2 + H 2 O Dolomite Greenwood Classification Tremolite - in Calcite Ca 2 Mg 5 Si 8 O 22(OH)2 + 3 Ca. CO 3 +7 CO 2 Qtz Tremolite Calcite #6 Calcite Diopside - in Ca 2 Mg 5 Si 8 O 22(OH)2 + 3 Ca. CO 3 + 2 Si. O 2 5 Ca. Mg. Si 2 O 6 + 3 CO 2 + H 2 O Tremolite #6 #4 Diopside Olivine - in Ca 2 Mg 5 Si 8 O 22(OH)2 + 11 Ca. Mg(CO 3)2 8 Mg 2 Si. O 4 + 13 Ca. CO 3 + 9 CO 2 + H 2 O Tremolite Olivine Dolomite Ca 2 Mg 5 Si 8 O 22(OH)2 + Ca. CO 3 Tremolite Calcite Ca. CO 3 + Si. O 2 Calcite Tremolite-out Qtz Wollastonite-in #4 Calcite Mg 2 Si. O 4 + 3 Ca. Mg. Si 2 O 6 + CO 2 + H 2 O Olivine Ca. Si. O 3 + CO 2 Wollastonite #4 Diopside #3
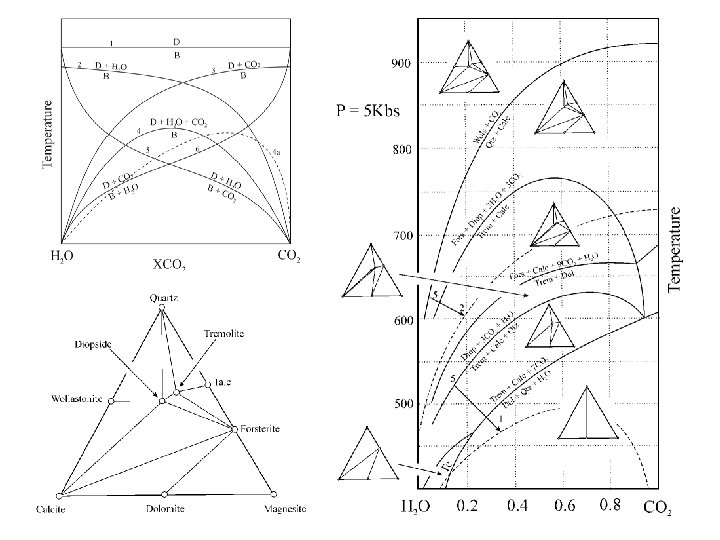

Buffering The metamorphic mineral assemblages developed in rocks are sensitive to the degree to which rocks behave as closed systems. Two end member situations may be imagined: • Internally Buffered Systems The rock behaves as a closed system, in which the activities of all components within the system are buffered by the bulk composition. In such systems, the compositions of the fluid phase may vary greatly as metamorphic reactions release additional volatiles. • Externally Buffered Systems The rock behaves as a completely open system with respect to fluids such that the activity of water (and other volatiles) is buffered or controlled outside the system. In extreme cases, the composition of the fluid phase may be fixed, and control the mineral assemblage in the rock. In general, a degree of freedom is lost for each component that is externally buffered, which is typically reflected in a reduction in the number of phases in stable mineral assemblages. The phase rule becomes: F = C–P + N where C = only the number of internally buffered chemical components, and N = 2 (P, T) + the number of externally buffered chemical components.

In reality, neither of these two end member cases is typical, but something intermediate prevails. The key parameter is the fluid / rock ratio, the total weight of fluid that has passed through a unit weight of rock. In systems in which the fluid / rock ratio is low, the bulk composition of the rock will control the composition of the fluid phase. However, systems that experience a high through put of fluids from the outside will be transformed so as to equilibrate with the fluid. Rocks that reflect such situations are usually characterized by phase assemblages that have apparently high variance (ie. a low number of phases for the number of apparent components). Extreme examples are mono-mineralic chlorite alteration assemblages in the fluid pathways responsible for exhalative ore deposits. Consider the system Mg. O – H 2 O – CO 2 Magnesite + H 2 O Brucite Mg. CO 3 H 2 O Mg(OH)2 84 18 58 + CO 2 44 molecular wt. So that, in a system with 99 gms magnesite and 1 gm fluid (a. CO 2 = XCO 2 = 0. 02), If at approximately 550 o. C: 1 gm magnesite + 0. 214 gms H 2 O 0. 691 gms brucite + 0. 524 gms CO 2 Then the amount of fluid would increase to 1. 31 gms and the activity of CO 2 would be: XCO 2 = 0. 544 / 1. 31 = 0. 42
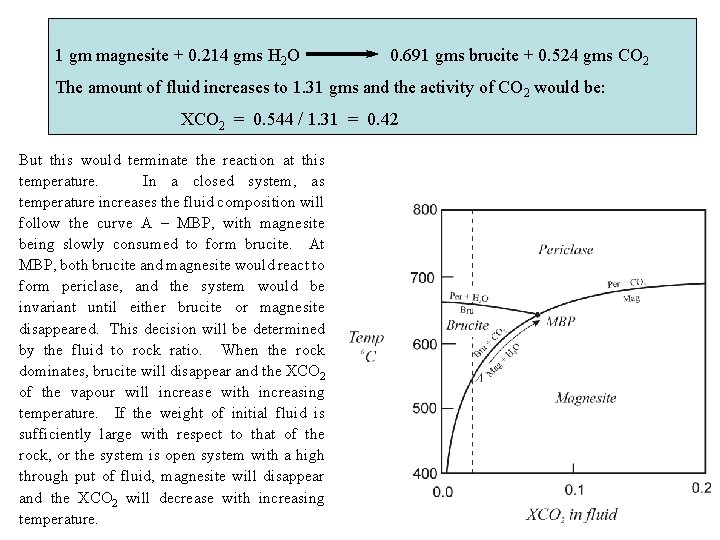
1 gm magnesite + 0. 214 gms H 2 O 0. 691 gms brucite + 0. 524 gms CO 2 The amount of fluid increases to 1. 31 gms and the activity of CO 2 would be: XCO 2 = 0. 544 / 1. 31 = 0. 42 But this would terminate the reaction at this temperature. In a closed system, as temperature increases the fluid composition will follow the curve A – MBP, with magnesite being slowly consumed to form brucite. At MBP, both brucite and magnesite would react to form periclase, and the system would be invariant until either brucite or magnesite disappeared. This decision will be determined by the fluid to rock ratio. When the rock dominates, brucite will disappear and the XCO 2 of the vapour will increase with increasing temperature. If the weight of initial fluid is sufficiently large with respect to that of the rock, or the system is open system with a high through put of fluid, magnesite will disappear and the XCO 2 will decrease with increasing temperature.
- Slides: 32