G MIXTURES Mixture Combination of 2 or more

G. MIXTURES Mixture Combination of 2 or more pure substances that are not chemically combined Substances are held together by physical forces, not chemical NO CHEMICAL CHANGE TAKES PLACE Each item retains its properties in the mixture They can be separated physically

G. MIXTURES CONT. Can be classified as: Heterogeneous mixture Homogeneous mixture

H. HETEROGENEOUS V. HOMOGENEOUS Heterogeneous Mixture A mixture that does not blend Example: Sand mixed with water, soup Homogeneous smoothly throughout Mixture A mixture that has constant composition Homogeneous mixture = SOLUTION Example: Salt dissolved in water, Gatorade throughout

CHECK Homogeneous or heterogeneous mixture? Vinegar b) Iron filings in sand c) Chicken noodle soup d) Muddy water a)
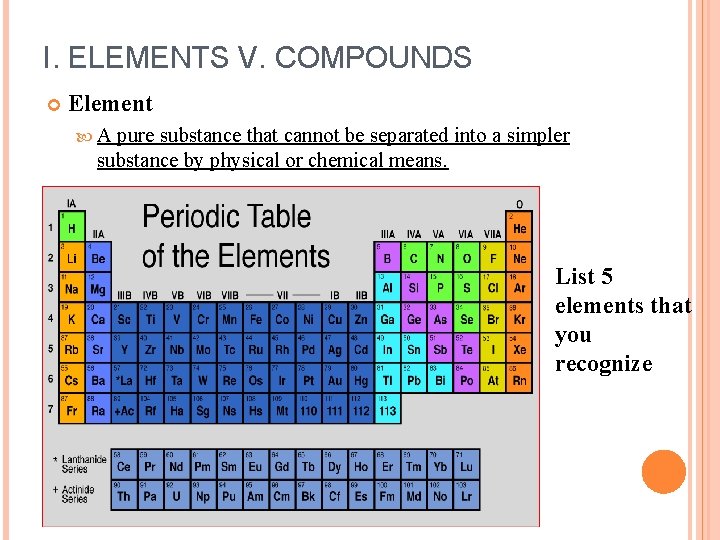
I. ELEMENTS V. COMPOUNDS Element A pure substance that cannot be separated into a simpler substance by physical or chemical means. List 5 elements that you recognize

I. ELEMENTS V. COMPOUNDS CONT. Compounds Combination of two or more different elements that are combined chemically Made of elements in a specific ratio that is always the same Has a chemical formula Can only be separated by chemical means, not physically

CHECK Element a) b) c) d) e) or Compound? Neon (Ne) Water (H 2 O) Salt (Na. Cl) Carbon Dioxide (CO 2) Sulfur (S)
CLASSIFICATION OF MATTER Matter changes
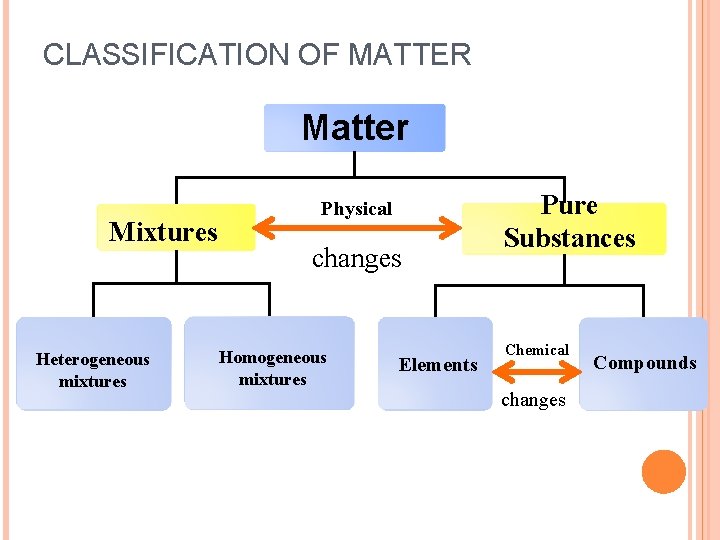
CLASSIFICATION OF MATTER Matter Mixtures Heterogeneous mixtures Physical changes Homogeneous mixtures Elements Pure Substances Chemical changes Compounds
- Slides: 9