G 9 Measurements precision and accuracy Todays objectives

G 9: Measurements, precision and accuracy

Today’s objectives 1. Revising metric prefixes 2. Recognising laboratory equipment 3. Accuracy versus precision versus error

When does accuracy matter?



How sure can we be about our measurements? • Our measurements should be as accurate, precise and reliable as possible • Accuracy – how close a measurement is to the true value • Precision – how close the measurements are to each other (reproducibility) • Reliable - If someone else can repeat your measurements and get the same results then your results are reliable. Carrying out repeat measurements checks the reliability of your results.

Precision • Indicates repeatability of a measurement • Precision often also indicates how small a measurement is. Millimetres are more precise than centimetres because they are smaller.

Precision or accuracy?

Applying Precision • Scientists use decimal places OR significant figures to show precision of a measured quantity • In science, numbers are often rounded (up or down) to a defined number of significant figures OR a defined number of decimal points • You are already familiar with decimal places…

Decimal places versus significant figures When we round to decimals, we start counting as soon as we reach the decimal point. Round 17. 46789 to 2 decimal places 1 decimal place 2 decimal places, this will stay a 6 or go to a 7 depending on what the next number is

Rules for approximating to decimal places Rule One Determine what your rounding digit is and look to the right side of it. If that digit is 4, 3, 2, or 1, simply drop all digits to the right of it. Rule Two Determine what your rounding digit is and look to the right side of it. If that digit is 5, 6, 7, 8, or 9 add one to the rounding digit and drop all digits to the right of it.

Significant Figures Significant figure: ‘each of the digits of a number that are used to express it to the required degree of accuracy, starting from the first nonzero digit’

Rules for Counting Significant Figures 1. Nonzero integers always count as significant figures. 1457 has 4 significant figures 23. 3 has 3 significant figures 2. Zeros a. Leading zeros - never count 0. 0025 2 significant figures b. Captive zeros - always count 1. 008 4 significant figures c. Trailing zeros - count only if the number is written with a decimal point 100 1 significant figure 100. 3 significant figures 120. 0 4 significant figures Zeros after a decimal point ARE SIGNIFICANT They do not begin the number 25. 00 and 15. 10 have FOUR Significant Figures

Numbers in scientific notation always indicate accurately how many sig figs are present An EASY way to decide is to convert the number to SCIENTIFIC NOTATION (but keep and consider any zeros following a decimal point)… The number of digits in your ‘mantissa’ is your number of significant figures For example: 32701. 00 = 3. 270100 X 104 Note that in this example, the two zeros following the decimal point are also considered as significant…

Significant Figures When we round to significant figures, we start counting as soon as we reach a number that is not zero Round 17. 46789 to 2 significant figures 1 significant figure 2 significant figures, this will stay a 7 or go to a 8 depending on what the next number is All the numbers after will become zero Round 17. 46789 to 2 decimal places=17. 00

Calculations using significant figures • Your answer reflects the reliability of the LEAST PRECISE operation • An answer is no more precise than the least precise number in the calculation • Imagine a team race where you must finish together – the slowest member of the team determines where you finish….

Significant figures – addition/ subtraction The answer cannot have more decimal places than the least number of decimal places in the calculation • Add 15. 1 to 3 • The answer is 18

Significant Figures – Multiplication/Division Based on the given values, round your answer so that it has the same number of significant figures as the least precise number • Multiply 2. 0 and 3. 01 • Answer in correct significant figures is 6. 0

Making precise measurements in science • Measurements in science are not the same as pure numbers in mathematics. Measurements are never exactly precise and have uncertainty. • UNCERTAINTY is the range of values on either side of a measurement in which the actual measurement might lie. • The smaller the divisions are (the number of decimal places) on a measuring device, the more precise (repeatable) our measurements become and thus usually more accurate.
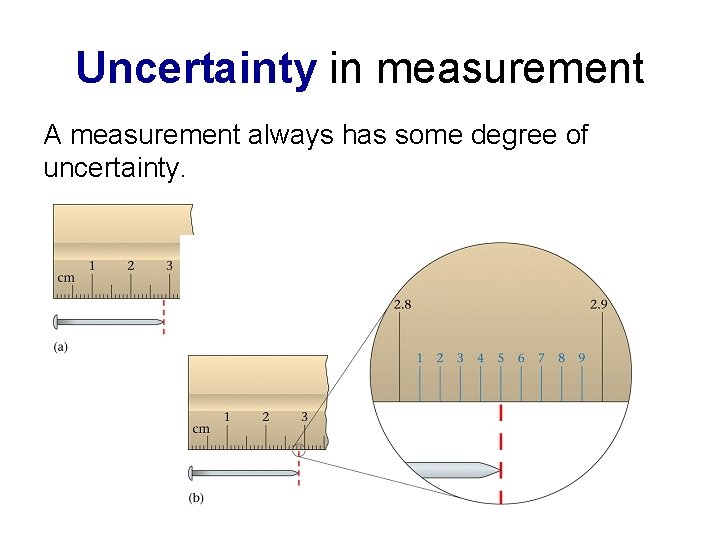
Uncertainty in measurement A measurement always has some degree of uncertainty.

Determining uncertainty • Uncertainty is determined by calculating one half of the smallest division of the measurement scale if the divisions are sufficiently spaced to estimate between them. • If the spaces between the divisions are too small to make a sensible estimate, then uncertainty is determined as the value of the smallest division.

How to record uncertainty Uncertainty is indicated using the symbol ± Whenever you use a measuring device, the measurement you write down must ALWAYS include the symbol ±, followed by the uncertainty value.
- Slides: 23