Further Types of Beta Decay Beta Decay So

Further Types of Beta Decay

Beta Decay So far we have met β- decay. One example of this decay is the decay of the nuclide Notice that the proton number of the beta particle is -1 Carbon 14 is a very important nuclide in radioactive dating of organic remains. Remember that the decay is essentially the decay of a neutron within the carbon-14 nucleus.

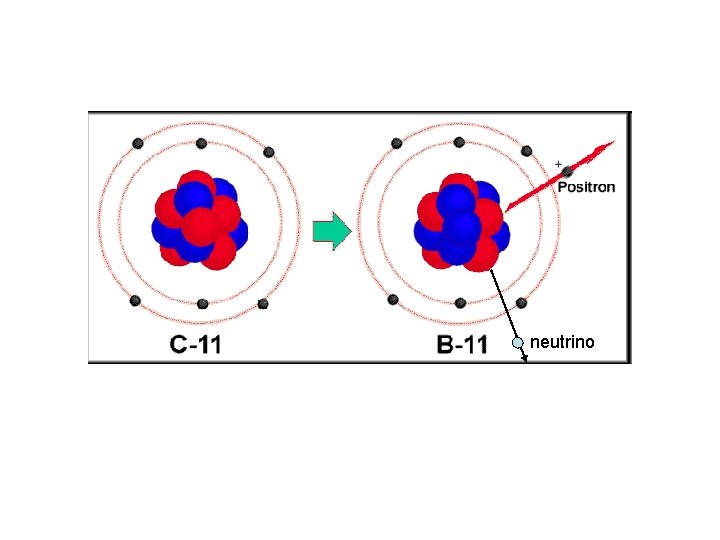
Beta Decay It is quite possible for the REVERSE process to occur. That is for a proton to “flip” and become a neutron: This is found to happen in the decay of another artificially made carbon isotope: The product of the decay is What is the charge on the β particle emitted? Another particle must be emitted in this process. Can you name it? Can you write an equation for the decay? Notice that the proton number of the positron produced is +1

β(+) decay (positron emission) • β(+) decay occurs when a proton is converted to a neutron with the emission of a positron ( an anti- electron). • Notice that the positron in β(+) decay is accompanied by the emission of a neutrino. • Positron emission does not occur on earth naturally, but positron emitting nuclides can be created in nuclear processes. Common positron emitters Mass number (A) C 11 N-13 13 O-15 15 F-18 18 Write decay equations for each nuclide in the table
neutrino
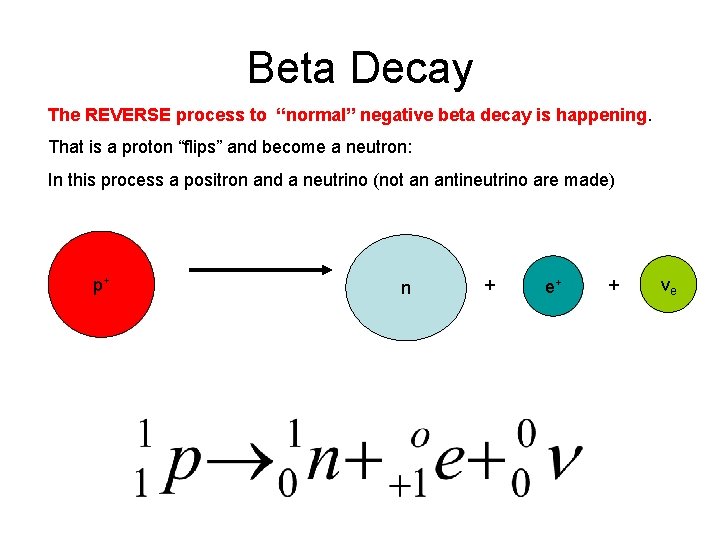
Beta Decay The REVERSE process to “normal” negative beta decay is happening. That is a proton “flips” and become a neutron: In this process a positron and a neutrino (not an antineutrino are made) p+ n + e+ + νe

All of the nuclides below undergo beta-plus decay write an equation for each process. You do not need to show charges if you show proton number “positron emission tomography (PET) scans involve the injection of a short lived isotope (often iodine 121) into the body as a “tracer” in the blood stream Positrons are emitted annihilated by collision with electrons in the body. The resulting gamma rays are detected.
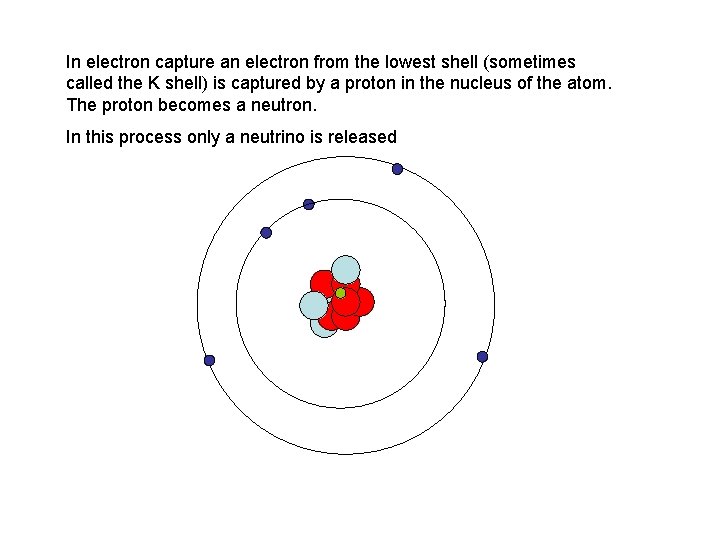
In electron capture an electron from the lowest shell (sometimes called the K shell) is captured by a proton in the nucleus of the atom. The proton becomes a neutron. In this process only a neutrino is released
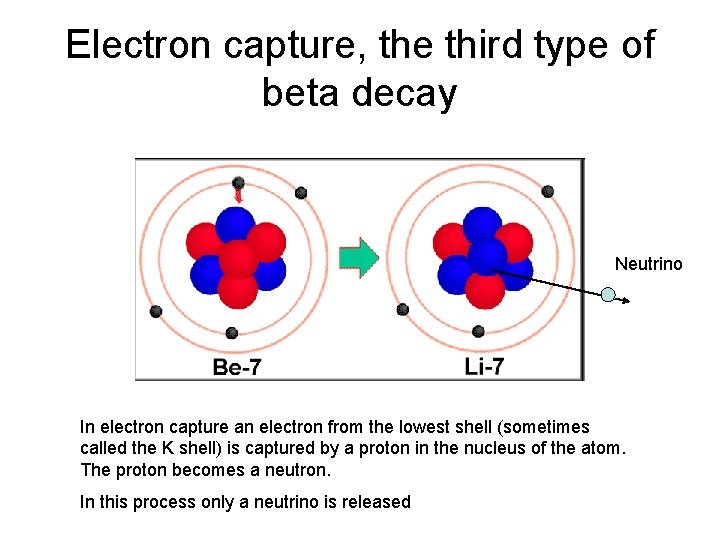
Electron capture, the third type of beta decay Neutrino In electron capture an electron from the lowest shell (sometimes called the K shell) is captured by a proton in the nucleus of the atom. The proton becomes a neutron. In this process only a neutrino is released

Electron Capture Can you finish the equations for these nuclides undergoing electron capture?

Beta decay and the weak interaction • It became obvious that beta decay could not be the result of the strong force as antineutrinos and electrons are not affected by the strong force. • There must be a fourth force in nature responsible for beta decay. This force is called the weak force (or weak interaction). We will see why it is weak later.

Another Conservation Law • We have seen a few things are conserved in all these processes – energy, and charge being two of these quantities • By allocating a number to each of the leptons we can see another quantity is being conserved
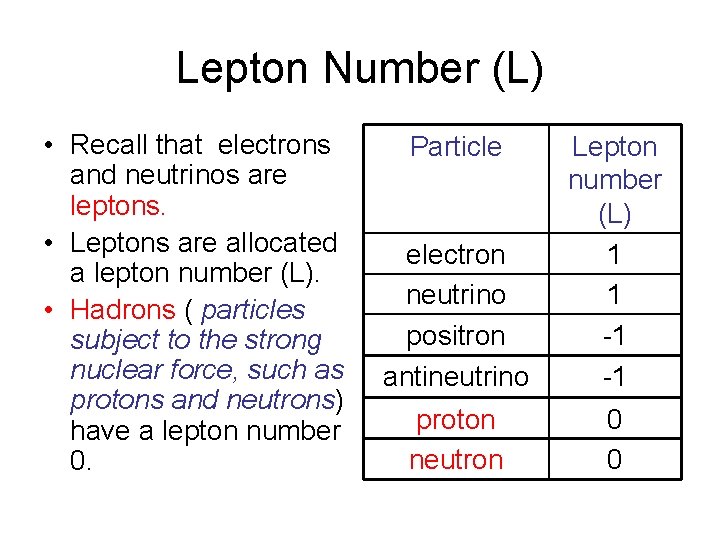
Lepton Number (L) • Recall that electrons and neutrinos are leptons. • Leptons are allocated a lepton number (L). • Hadrons ( particles subject to the strong nuclear force, such as protons and neutrons) have a lepton number 0. Particle electron neutrino positron antineutrino Lepton number (L) 1 1 -1 -1 proton neutron 0 0

Beta(-) Decay And Lepton Number Lepton number is always conserved in any process. Beta decay is no exception Lepton no (L) 0 + =0 0 1 + ( -1) =0

K capture The general formula for K capture is: 1. Show that lepton number is preserved in this process. 2. Show that in the beta minus decay and in electron capture that lepton number is preserved. positron emission Beta minus decay 3. Write general formulas for beta minus and electron capture like the general formula for K capture above.
- Slides: 15