Fundamentals of General Organic and Biological Chemistry 7

Fundamentals of General, Organic and Biological Chemistry 7 th Edition Chapter Four Molecular Compounds

Chapter 4 Goals Topics and Goals for the Chapter: • What is a covalent bond? Be able to define and describe the nature of covalent bonds and how they are formed. • How does the octet rule apply to covalent bond formation? Be able to use the octet rule to predict the numbers of covalent bonds formed by common main group elements. Be able to define single bond, double bond and triple bond. • What are the major differences between ionic and molecular bonds? Be able to compare the structures, compositions, and properties of ionic and molecular compounds. • How are molecular compounds represented? Be able to interpret molecular formulas and draw Lewis structures for molecules. Chapter 4 2013 Pearson 2

Chapter 4 Goals Cont. • What is the influence of valence-shell electrons on molecular shape? Be able to define VSEPR theory and be able to apply it to predict the molecular geometry around a central atom in a Lewis structure. • When are bonds and molecules polar? Be able to define electronegativity and polar covalent bond. Be able to understand the trends of increasing and decreasing electronegativity values in the periodic table. Be able to use electronegativity and molecular geometry to predict bond and molecular polarity. • How are binary molecular compounds named? Be able to correctly name binary molecular compounds. 2013 Pearson Education, Inc. Chapter 4 3 3

4. 1 Covalent Bonds Ionic bonds • Complete transfer of e- from one atom to another to form bond. Covalent bonds • Sharing of electrons between atoms to form bond. • Generally interactions between nonmetallic elements. Molecule – a group of atoms held together by covalent bonds. Nonmetals can achieve an octet by sharing an appropriate number of electrons in covalent bonds. Chapter 4 2013 Pearson 4

H 2 is the simplest covalent bond H∙ 1 s 1 e • • ∙H 1 s 1 e- When two H atoms come together, electrical interactions occur. Some interactions are repulsive—the two positively charged nuclei repel each other and the negatively charged electrons repel each other. Other interactions are attractive—each nucleus attracts the electrons and each electron attracts both nuclei. When the attractive forces are stronger than the repulsive forces, a covalent bond is formed and the atoms stay together. Chapter 4 2013 Pearson 5
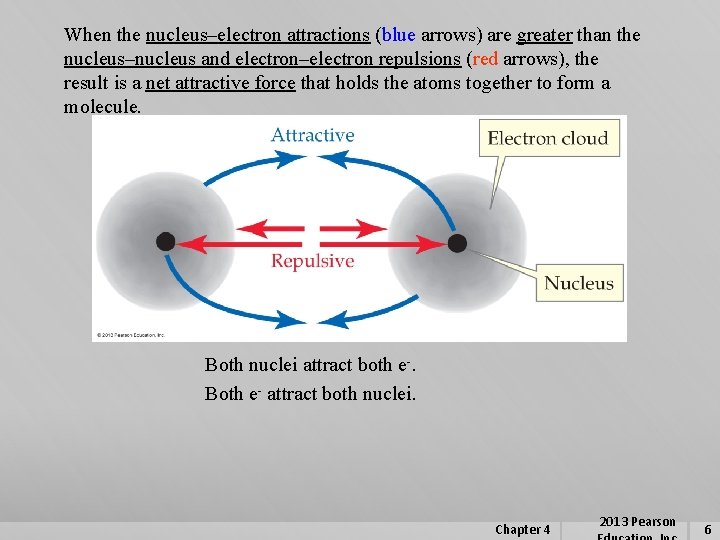
When the nucleus–electron attractions (blue arrows) are greater than the nucleus–nucleus and electron–electron repulsions (red arrows), the result is a net attractive force that holds the atoms together to form a molecule. Both nuclei attract both e-. Both e- attract both nuclei. Chapter 4 2013 Pearson 6
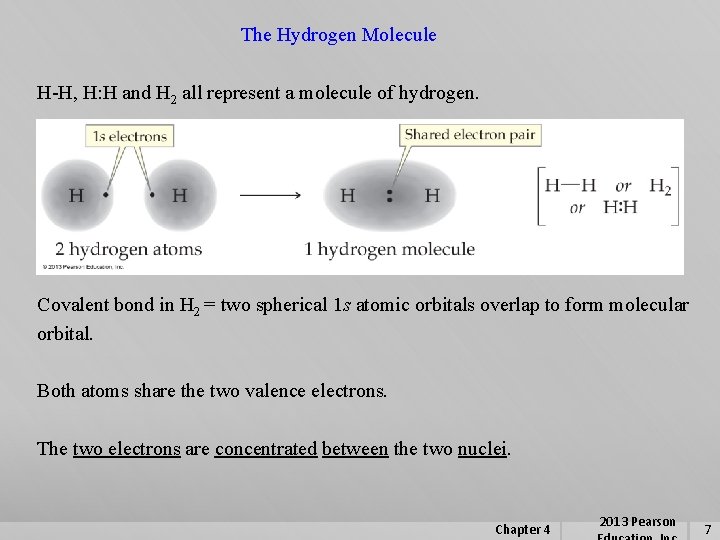
The Hydrogen Molecule H-H, H: H and H 2 all represent a molecule of hydrogen. Covalent bond in H 2 = two spherical 1 s atomic orbitals overlap to form molecular orbital. Both atoms share the two valence electrons. The two electrons are concentrated between the two nuclei. Chapter 4 2013 Pearson 7
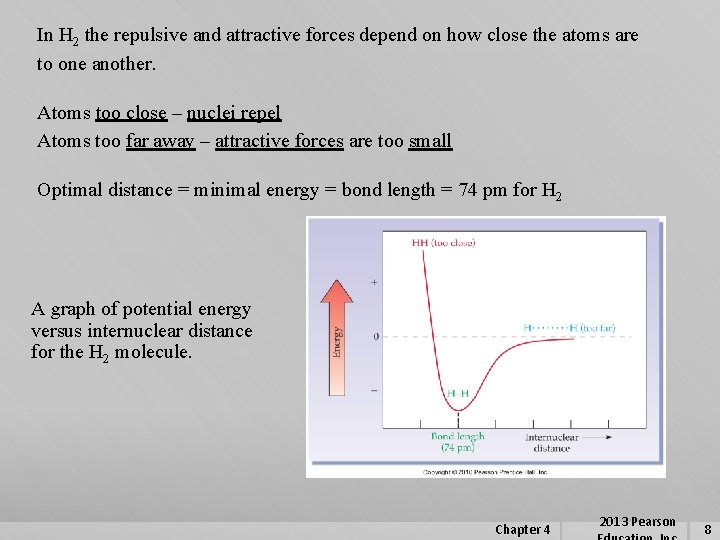
In H 2 the repulsive and attractive forces depend on how close the atoms are to one another. Atoms too close – nuclei repel Atoms too far away – attractive forces are too small Optimal distance = minimal energy = bond length = 74 pm for H 2 A graph of potential energy versus internuclear distance for the H 2 molecule. Chapter 4 2013 Pearson 8
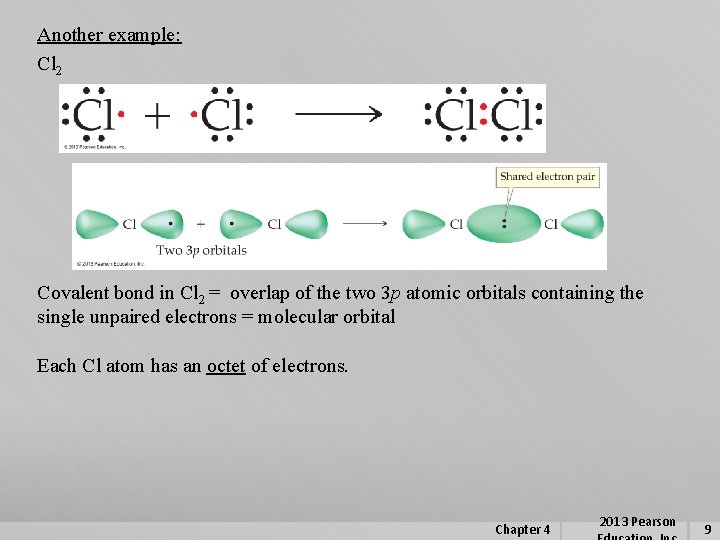
Another example: Cl 2 Covalent bond in Cl 2 = overlap of the two 3 p atomic orbitals containing the single unpaired electrons = molecular orbital Each Cl atom has an octet of electrons. Chapter 4 2013 Pearson 9

There are 7 diatomic elements – H 2, N 2, O 2, F 2, Cl 2, Br 2, I 2 Atoms are held together by covalent bonds. N 2 and O 2 - colorless, odorless, nontoxic gases. F 2 - a pale yellow, highly reactive gas. Br 2 - a dark red, toxic liquid I 2 - a violet, crystalline solid. Chapter 4 2013 Pearson 10
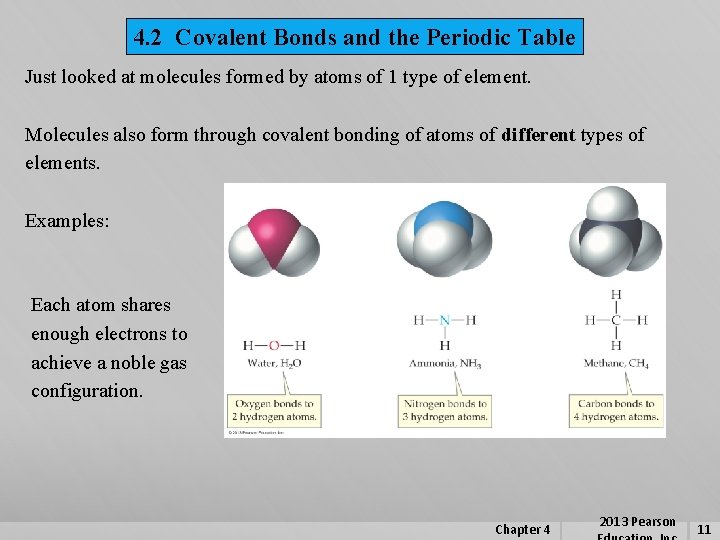
4. 2 Covalent Bonds and the Periodic Table Just looked at molecules formed by atoms of 1 type of element. Molecules also form through covalent bonding of atoms of different types of elements. Examples: Each atom shares enough electrons to achieve a noble gas configuration. Chapter 4 2013 Pearson 11

Figure 5. 4 summarizes the number of covalent bonds typically formed by common main group elements. In general, # of bonds formed follows fulfilling the octet rule. There are exceptions to the octet rule. Numbers in parentheses indicate possible numbers of bonds that result in exceptions to the octet rule. Chapter 4 2013 Pearson 12

Exceptions to the Octet Rule Boron Only has 3 e- it can share. Only forms 3 covalent bonds. Ex: BF 3 Elements in 3 rd row and below Have vacant d orbitals available. Can have greater than an octet of electrons around the atom. Chapter 4 2013 Pearson 13

4. 3 Multiple Covalent Bonds Atoms can share more than 1 electron. Double and triple covalent bonds can form between two atoms. CO 2 and N 2 are covalent molecules. How do the atoms obtain a full octet? Single bonds? Chapter 4 2013 Pearson 14
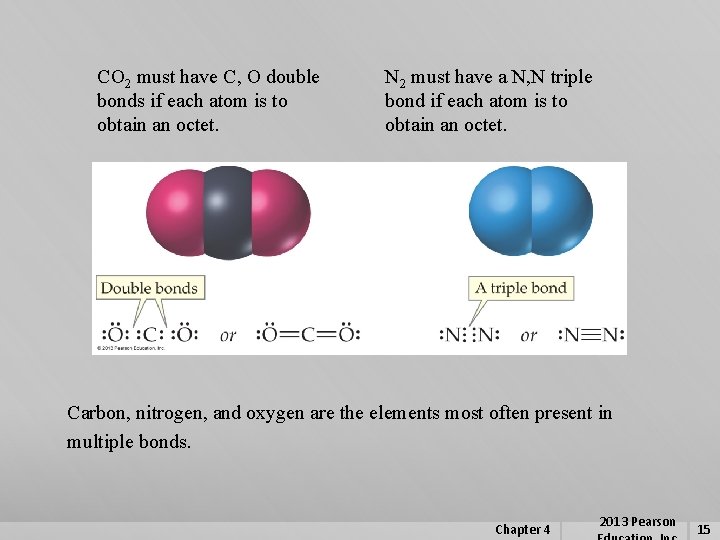
CO 2 must have C, O double bonds if each atom is to obtain an octet. N 2 must have a N, N triple bond if each atom is to obtain an octet. Carbon, nitrogen, and oxygen are the elements most often present in multiple bonds. Chapter 4 2013 Pearson 15
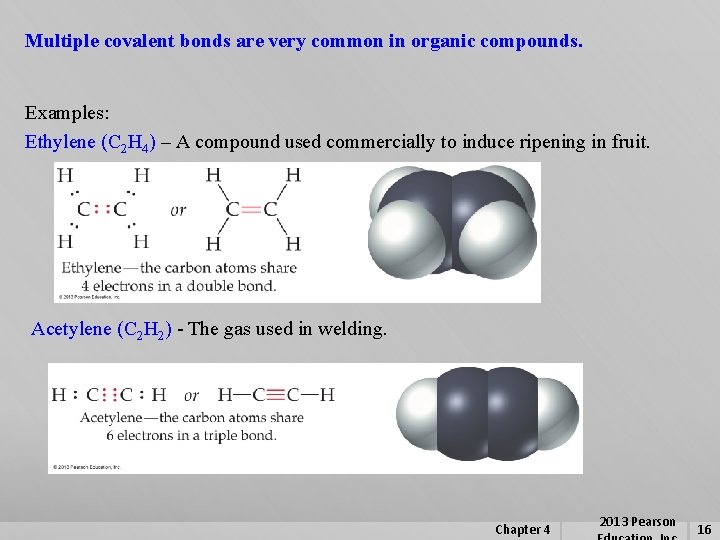
Multiple covalent bonds are very common in organic compounds. Examples: Ethylene (C 2 H 4) – A compound used commercially to induce ripening in fruit. Acetylene (C 2 H 2) - The gas used in welding. Chapter 4 2013 Pearson 16
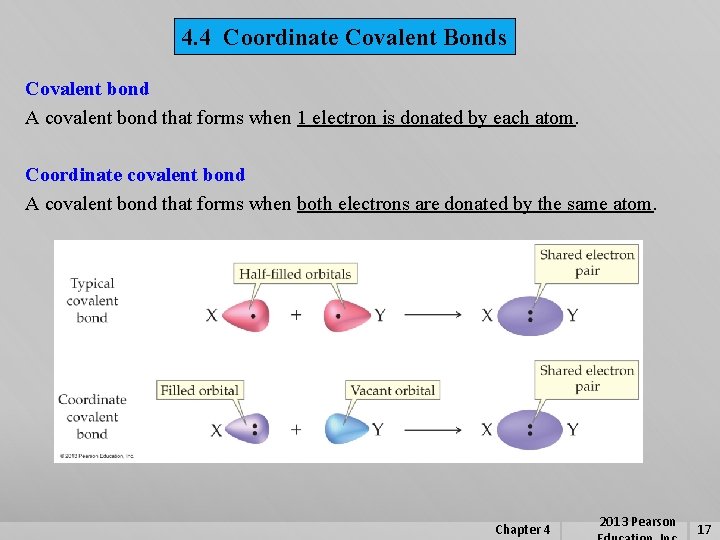
4. 4 Coordinate Covalent Bonds Covalent bond A covalent bond that forms when 1 electron is donated by each atom. Coordinate covalent bond A covalent bond that forms when both electrons are donated by the same atom. Chapter 4 2013 Pearson 17

4. 5 Characteristics of Molecular Compounds Molecules have forces between molecules – intermolecular forces We will look at intermolecular forces more closely in Chapter 8. If molecules have weak intermolecular forces, the molecules are very weakly attracted to one another. The molecular substance is a gas at ordinary temperatures. As intermolecular forces increase, the substances tend to be liquids and solids. Intermolecular forces State weak gases < liquids < strong solids Chapter 4 2013 Pearson 18
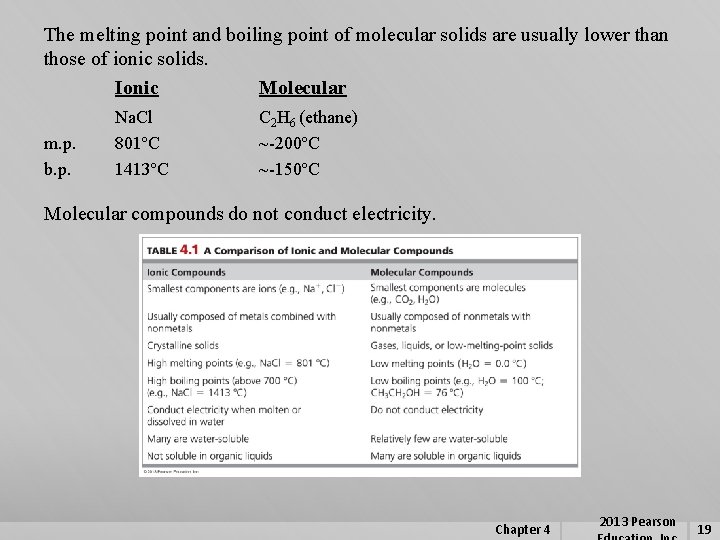
The melting point and boiling point of molecular solids are usually lower than those of ionic solids. Ionic Molecular m. p. b. p. Na. Cl 801ºC 1413ºC C 2 H 6 (ethane) ~-200ºC ~-150ºC Molecular compounds do not conduct electricity. Chapter 4 2013 Pearson 19
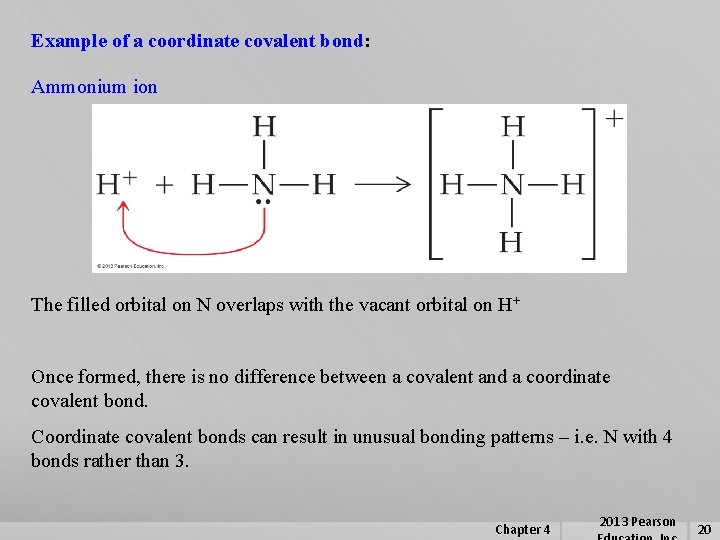
Example of a coordinate covalent bond: Ammonium ion The filled orbital on N overlaps with the vacant orbital on H+ Once formed, there is no difference between a covalent and a coordinate covalent bond. Coordinate covalent bonds can result in unusual bonding patterns – i. e. N with 4 bonds rather than 3. Chapter 4 2013 Pearson 20

4. 6 Molecular Formulas and Lewis Structures Different Structural Representations for Molecules Molecular formula – A formula that shows the numbers and kinds of atoms in one molecule of a compound. Condensed structure – Provides minimal structural information, actual bonds not shown but inferred. Structural formula – Provides minimal structural information, bonds to atoms shown as lines. Space-filling model – More realistic, shows bonding, relative size of atoms, 3 -D structure Ball and Stick Model – Shows bonding, relative size of atoms, 3 -D structure Lewis Structure - A molecular representation that shows both the connections among atoms and the locations of lone-pair valence electrons. Chapter 4 2013 Pearson 21
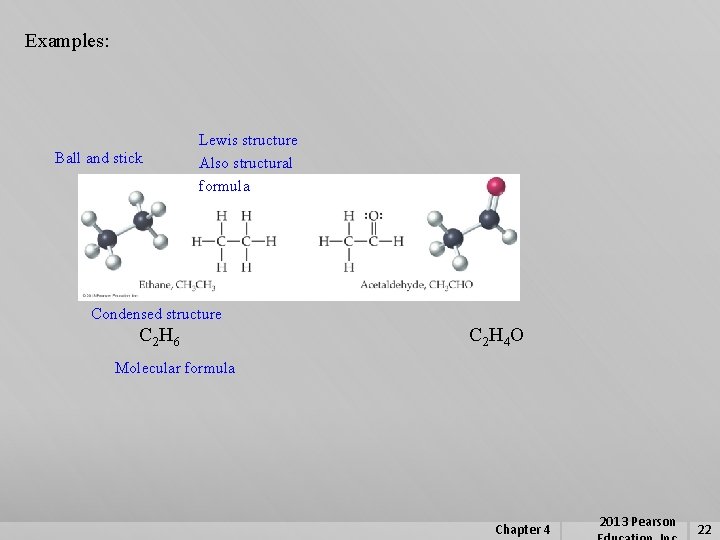
Examples: Ball and stick Lewis structure Also structural formula Condensed structure C 2 H 6 C 2 H 4 O Molecular formula Chapter 4 2013 Pearson 22
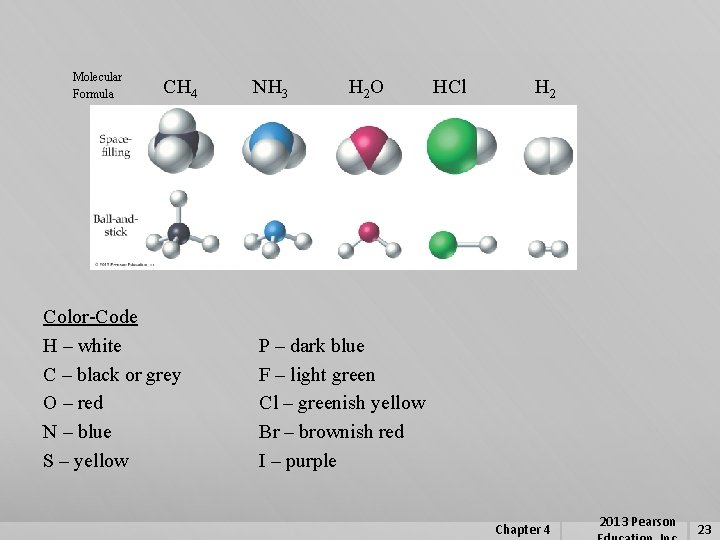
Molecular Formula CH 4 Color-Code H – white C – black or grey O – red N – blue S – yellow NH 3 H 2 O HCl H 2 P – dark blue F – light green Cl – greenish yellow Br – brownish red I – purple Chapter 4 2013 Pearson 23

Examples of Lewis Structures Chapter 4 2013 Pearson 24
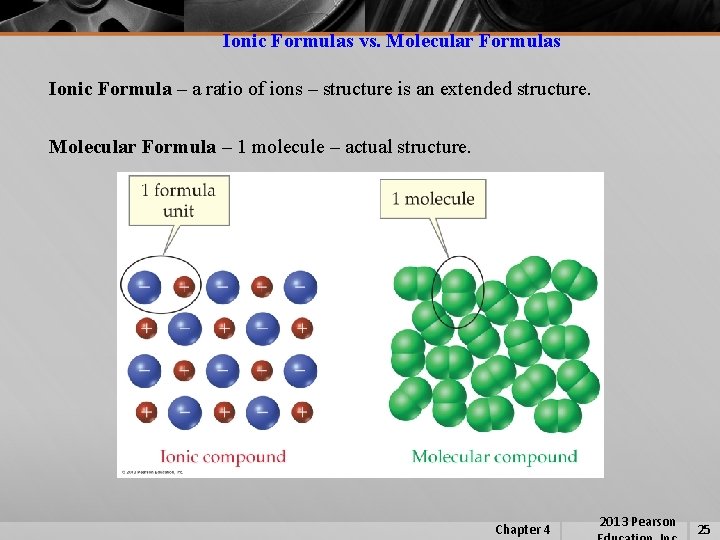
Ionic Formulas vs. Molecular Formulas Ionic Formula – a ratio of ions – structure is an extended structure. Molecular Formula – 1 molecule – actual structure. Chapter 4 2013 Pearson 25

4. 7 Drawing Lewis Structures To draw a Lewis structure you must know how the atoms are connected to one another. Two approaches are used for drawing Lewis structures. Approach 1 – Useful for organic molecules Common bonding patterns are followed by atoms. C forms four covalent bonds. N forms three covalent bonds and has one lone pair of electrons. O forms two covalent bonds and has two lone pairs of electrons. Halogens form one covalent bond and have three lone pairs of electrons. H forms one covalent bond. H and halogens – 1 bond = must be at the ends of structures. Example: No C-H-C, or C-F-C bonds in this approach. Chapter 4 2013 Pearson 26

Approach 2 – A general stepwise procedure which works for all molecules A slightly more detailed handout on the steps is provided in class and available on Blackboard. Basis of approach – Ignore information regarding which atom donated electrons to form a bond. Focus on the total number of atoms and the total number of valence electrons. Step 1 - Count the total number of valence electrons Molecule – Add valence electrons for all atoms Polyanion – Add an electron for each negative charge Polycation – Subtract an electron for each positive charge Step 2 - Draw a skeleton structure for the species, joining atoms by single lines to represent covalent bonds For some species there might be more than one possible structure if you are given a formula. Most of the molecules we deal with have a central atom bonded to two or more terminal atoms (NH 4+, SO 2, CCl 4). The central atom is usually written first in the formula followed by terminal atoms. Chapter 4 2013 Pearson 27

Approach 2 – A general stepwise procedure which works for all molecules - Continued Step 3 - Determine the number of valence electrons still available for distribution and add electrons so that each peripheral atom connected to the central atom gets an octet. (H = 2, not 8 electrons) That is, deduct two valence electrons for each single bond written in Step 2. and place the remaining electrons around the peripheral atoms in the structure. Step 4 - Place all remaining electron lone pairs on the central atom. Step 5 - If the central atom does not yet have an octet after all electrons have been assigned, take a lone pair from a neighboring atom and form a multiple bond to the central atom. Note: This will NOT change the number of electrons counted towards the octet on the peripheral atom. You are simply “sharing” the two electrons. Chapter 4 2013 Pearson 28

When determining the Lewis structure for a molecule – Approach 2 will ALWAYS give you the correct structure. Practicing drawing Lewis structures: O 3 – ozone NO 3 - Try CO 2 on your own. Chapter 4 2013 Pearson 29
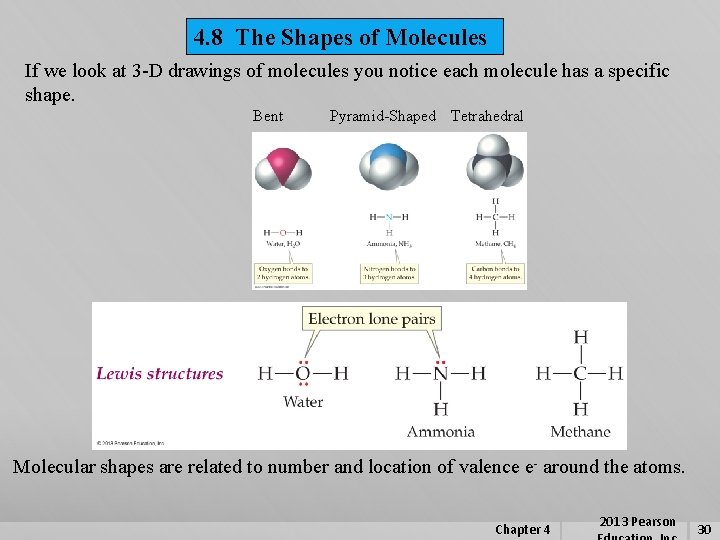
4. 8 The Shapes of Molecules If we look at 3 -D drawings of molecules you notice each molecule has a specific shape. Bent Pyramid-Shaped Tetrahedral Molecular shapes are related to number and location of valence e- around the atoms. Chapter 4 2013 Pearson 30

We can predict molecular shapes! Use VSEPR Model Valence Shell Electron Pair Repulsion Model According to VSEPR: Valence e- pairs (in bonds and unshared) surrounding an atom repel each other. Bonds and unshared e- orient themselves as far away from one another as possible to minimize repulsion. They take advantage of all available space. Chapter 4 2013 Pearson 31

VSEPR Model 3 steps to apply model to determine molecular shape of a molecule: Step 1: Draw a Lewis structure of the molecule and identify the atom whose geometry is of interest. In a simple molecule – The atom of interest will be the central atom. Step 2: Count the number of electron charge clouds (areas of electron density) surrounding the atom of interest. Single bond = 1 e- cloud Double bond = 1 e- cloud Triple bond = 1 e- cloud Lone e- pair = 1 e- cloud Step 3: Predict molecular shape by assuming that the charge clouds orient in space so that they are as far away from one another as possible. Chapter 4 2013 Pearson 32
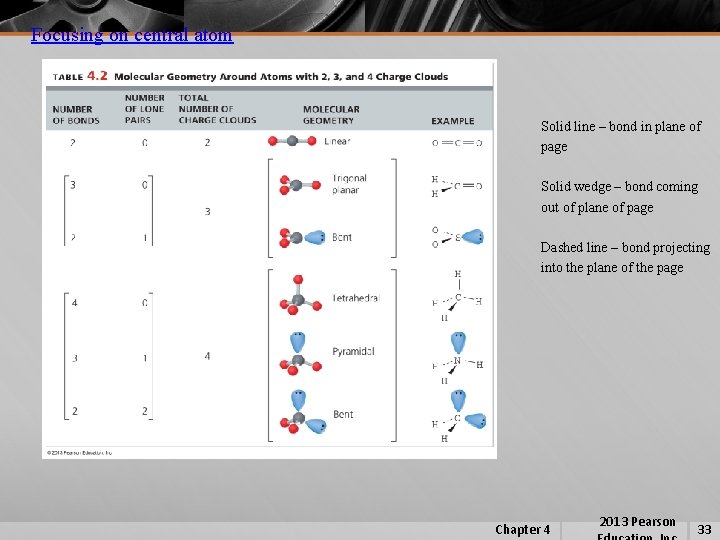
Focusing on central atom Solid line – bond in plane of page Solid wedge – bond coming out of plane of page Dashed line – bond projecting into the plane of the page Chapter 4 2013 Pearson 33
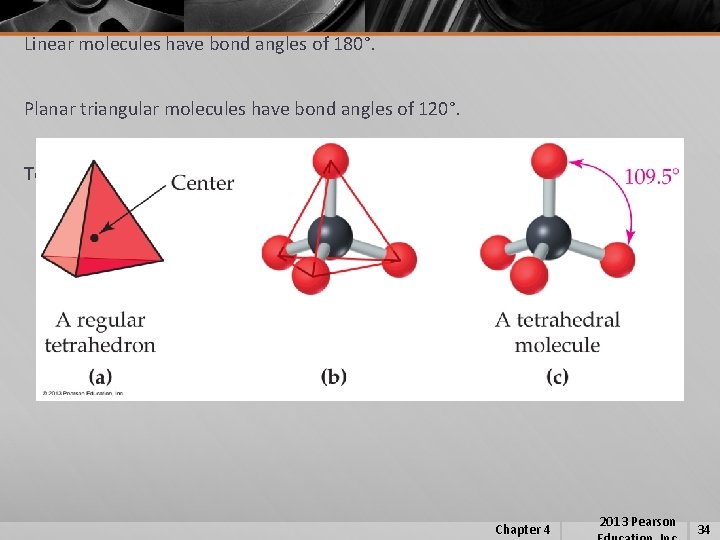
Linear molecules have bond angles of 180°. Planar triangular molecules have bond angles of 120°. Tetrahedral molecules have bond angles of 109. 5°. Chapter 4 2013 Pearson 34
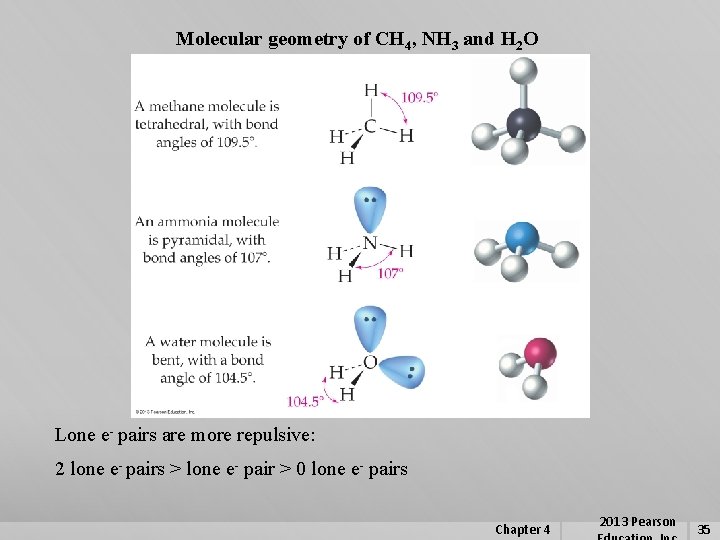
Molecular geometry of CH 4, NH 3 and H 2 O Lone e- pairs are more repulsive: 2 lone e- pairs > lone e- pair > 0 lone e- pairs Chapter 4 2013 Pearson 35
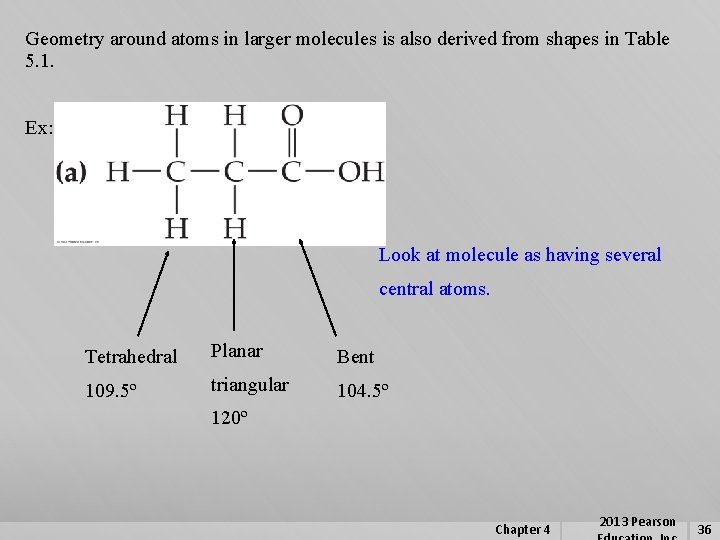
Geometry around atoms in larger molecules is also derived from shapes in Table 5. 1. Ex: Look at molecule as having several central atoms. Tetrahedral Planar Bent 109. 5º triangular 104. 5º 120º Chapter 4 2013 Pearson 36
Question: What is the molecular geometry of NO 3 -? Chapter 4 2013 Pearson 37

4. 9 Polar Covalent Bonds and Electronegativity Electrons in a covalent bond occupy the region between bonded atoms. Bond polarity – Describes sharing of e- between atoms. Non-polar covalent bond – Electrons shared equally between two atoms. Atoms need to be identical Ex: Cl 2, H 2, N 2, O 2 Polar covalent bond – Electrons shared unequally between two atoms. Atoms are not identical Bonding e- are attracted more strongly by one atom than by the other. Ex: H-O-H and H-Cl Chapter 4 2013 Pearson 38

Molecules with Polar Covalent Bonds Molecules are neutral overall but they have + and – areas. d+ = partial positive placed on more positive atom d- = partial negative placed on more negative atom H-O-H H-Cl d+ d- d+ d+ d- Chapter 4 2013 Pearson 39
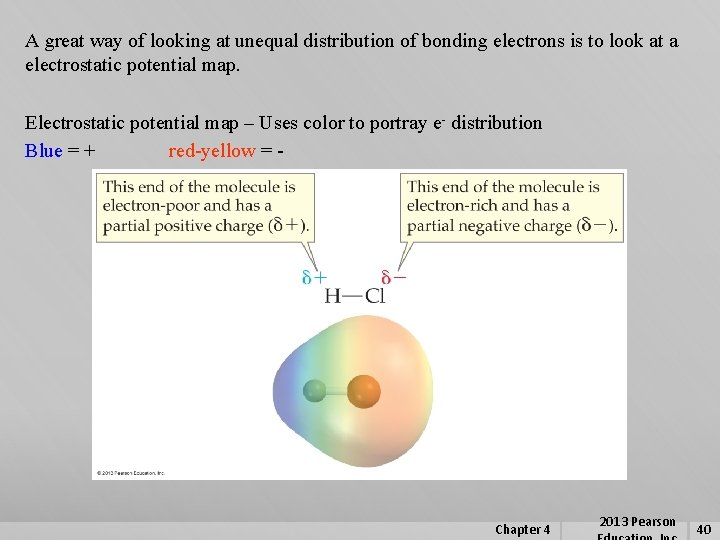
A great way of looking at unequal distribution of bonding electrons is to look at a electrostatic potential map. Electrostatic potential map – Uses color to portray e- distribution Blue = + red-yellow = - Chapter 4 2013 Pearson 40

Electronegativity We can determine bond polarities and predict the occurrence of covalent vs. ionic bonding in a molecule by comparing electronegativities of atoms. Electronegativity – The ability of an atom in a molecule to attract e- to itself. The electronegativity scale is based on giving the most electronegative element, F, an assigned value of 4. Electronegativity increases across a period. Electronegativity decreases down a group. Noble gases are not assigned values. Chapter 4 2013 Pearson 41
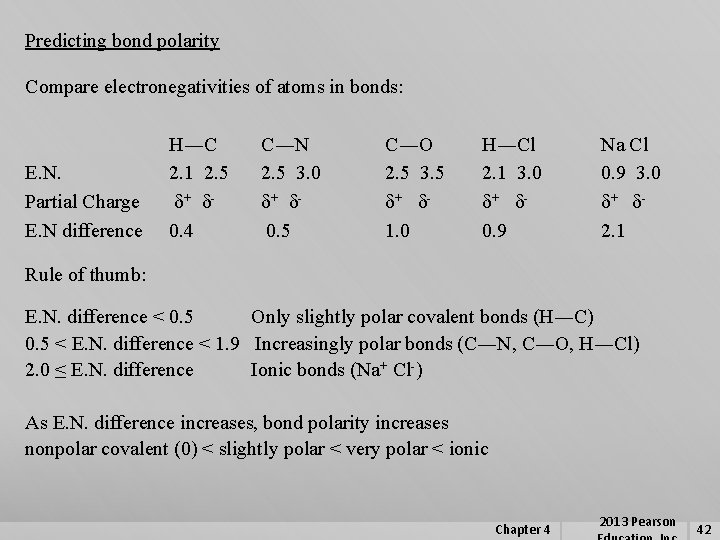
Predicting bond polarity Compare electronegativities of atoms in bonds: E. N. Partial Charge E. N difference H―C 2. 1 2. 5 d+ d 0. 4 C―N 2. 5 3. 0 d+ d 0. 5 C―O 2. 5 3. 5 d+ d 1. 0 H―Cl 2. 1 3. 0 d+ d 0. 9 Na Cl 0. 9 3. 0 d+ d 2. 1 Rule of thumb: E. N. difference < 0. 5 Only slightly polar covalent bonds (H―C) 0. 5 < E. N. difference < 1. 9 Increasingly polar bonds (C―N, C―O, H―Cl) 2. 0 ≤ E. N. difference Ionic bonds (Na+ Cl-) As E. N. difference increases, bond polarity increases nonpolar covalent (0) < slightly polar < very polar < ionic Chapter 4 2013 Pearson 42
Sample problem: Predict whether the H-S bond in H 2 S is nonpolar covalent, polar covalent or ionic. Chapter 4 2013 Pearson 43

4. 10 Polar Molecules Just as we can describe bonds as being polar or nonpolar, we can describe molecules as being polar or nonpolar. Polarity of molecules – Due to the sum of all individual bond polarities and lone pair contributions in the molecule. The physical properties of a molecule are dependent on the polarity of the molecule. Dipoles or polarity can be represented by an arrow pointing to the negative end of the molecule with a cross at the positive end resembling a + sign. d+ d- Chapter 4 2013 Pearson 44
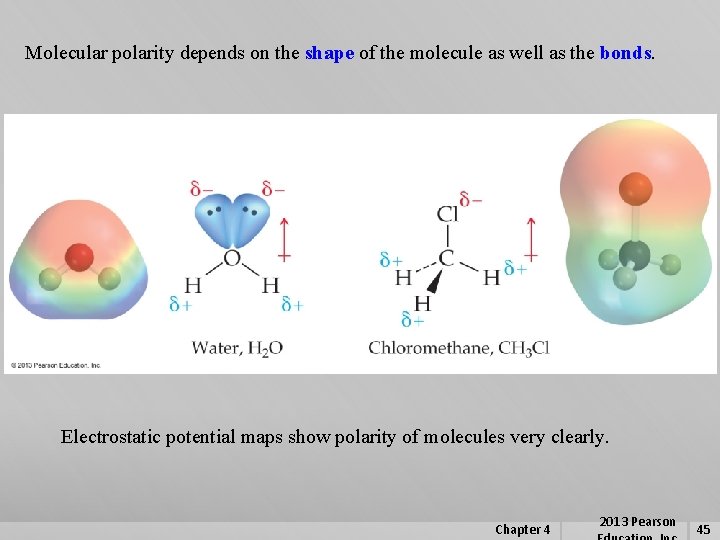
Molecular polarity depends on the shape of the molecule as well as the bonds. Electrostatic potential maps show polarity of molecules very clearly. Chapter 4 2013 Pearson 45
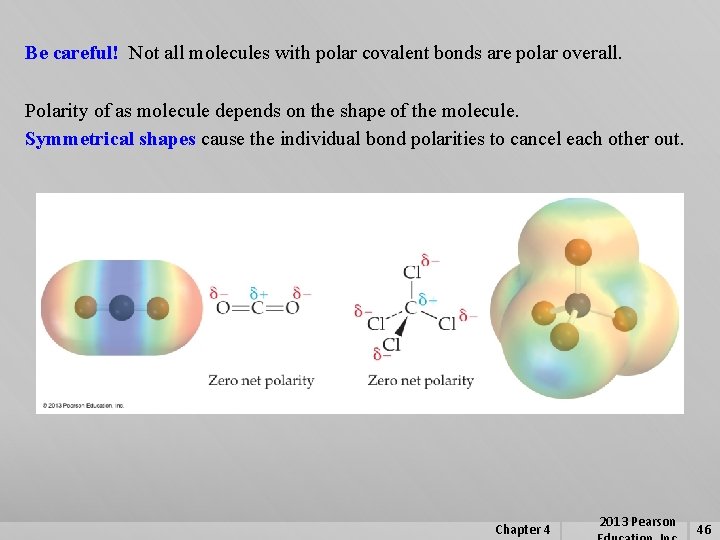
Be careful! Not all molecules with polar covalent bonds are polar overall. Polarity of as molecule depends on the shape of the molecule. Symmetrical shapes cause the individual bond polarities to cancel each other out. Chapter 4 2013 Pearson 46
A few practice problems: Determine whether the following molecules are polar: BH 3 SO 32 - 2013 Pearson Education, Inc. Chapter 4 47 47

4. 11 Naming Binary Molecular Compounds Binary compounds – Formulas of molecular compounds with two different types of atoms. Rules: 1. Formulas are written with the least electronegative element first a. Metals always written before nonmetals. b. Nonmetals further to the left on the periodic table are written first. N before O 2. When naming you must identify exactly how many atoms of each element are in the molecular formula. 3. Prefixes are used to indicate the number of atoms of each element that combine. Chapter 4 2013 Pearson 48

Naming Binary Molecular Compounds Rules Cont. 4. Two steps for naming: a. Name the first element in the formula. b. Name the second element in the formula. Use an –ide ending as for anions. For both of the above steps use a prefix to indicate the number of atoms, if needed. Note: The prefix mono can be omitted for the first element. (It is assumed to be one if given no prefix. ) Chapter 4 2013 Pearson 49
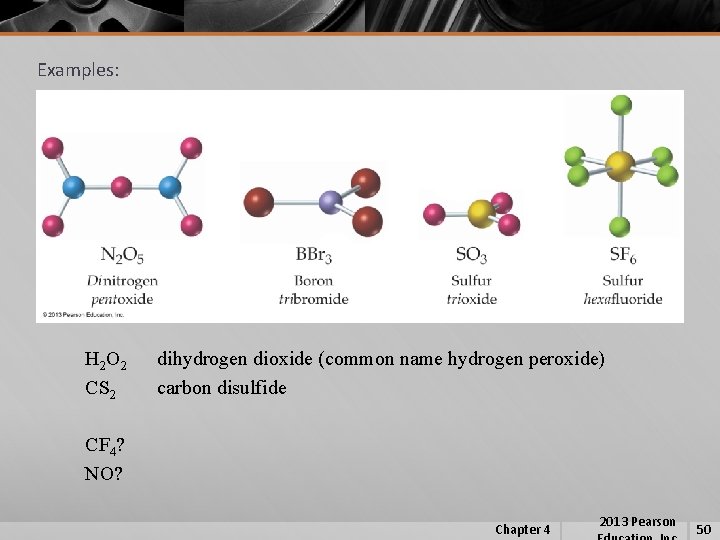
Examples: H 2 O 2 CS 2 dihydrogen dioxide (common name hydrogen peroxide) carbon disulfide CF 4? NO? Chapter 4 2013 Pearson 50
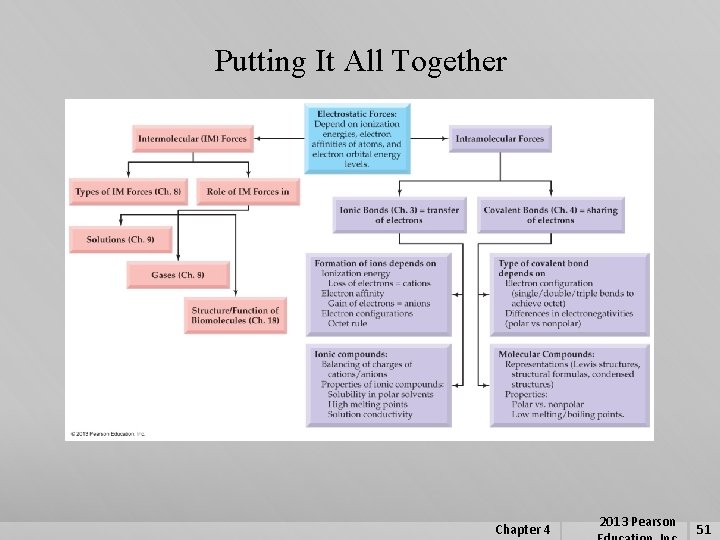
Putting It All Together Chapter 4 2013 Pearson 51

Chapter Summary • A covalent bond is formed by the sharing of electrons between atoms rather than by the complete transfer of electrons from one atom to another. • Two shared electrons are a single bond , four are a double bond, and six are a triple bond. • The group of atoms held together by covalent bonds is called a molecule. • When a lone pair of electrons on one atom overlaps a vacant orbital on another atom a coordinate covalent bond is formed. • An atom shares enough electrons to reach a noble gas configuration. • Molecular formulas show the numbers and kinds of atoms in a molecule. • Lewis structures show atoms are connected in molecules. • Covalent bonds are indicated as lines between atoms, and valence electron lone pairs are shown as dots. 2013 Pearson Education, Inc. Chapter 4 52 52

Chapter Summary Cont. • Molecules have shapes that can be predicted using the VSEPR model. • The electronic geometry of atoms with 2 electron charge clouds is linear, with 3 it is planar triangular, and with 4 it is tetrahedral. • Bonds between atoms are polar if the bonding electrons are not shared equally between the atoms. • The ability of an atom to attract electrons is electronegativity. It is highest on the upper right of the periodic table and lowest on the lower left. • Molecular polarity is the sum of all individual bond polarities and lone pair contributions in a molecule. • Molecular compounds usually have lower melting points and boiling points than ionic compounds, many are water insoluble, and they do not conduct electricity when melted or dissolved. Chapter 4 2013 Pearson 53
- Slides: 53