Fundamentals of Biochemistry Donald Winslow St Gregorys University

Fundamentals of Biochemistry Donald Winslow St. Gregory's University 21 January 2014

Biochemistry ● Chemistry of carbon ● Organic compounds ● Monomers & polymers ● Types of organic molecules in cells ● Diversity of shapes ● Hydrolysis and dehydration synthesis
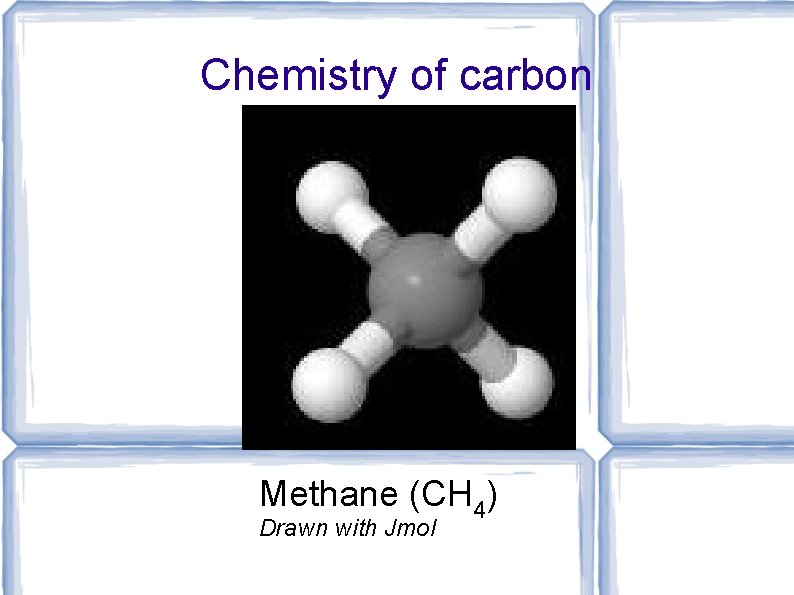
Chemistry of carbon Methane (CH 4) Drawn with Jmol

Organic compounds ● ● An organic compound is a compound containing carbon. Don't confuse with “organic” in “organic agriculture”. – Pesticides are organic compounds – “Organic” means no synthetic pesticides

Monomers and polymers ● What is a monomer? ● What is a polymer?

Monomers and polymers ● What is a monomer? – ● A small, unitary molecule What is a polymer? – Alanine, an amino acid Drawn with Jmol A large molecule built of monomers ● Often a chain of monomers Glycine and alanine can form a peptide bond, resulting in a dipeptide molecule. Many amino acids can link together to form a polypeptide, which can fold into a protein. Drawn with Jmol

What are the four main categories of organic compounds found in cells?

What are the four main categories of organic compounds found in cells? ● ● Lipids Carbohydrates Amino acids & proteins Nucleic acids
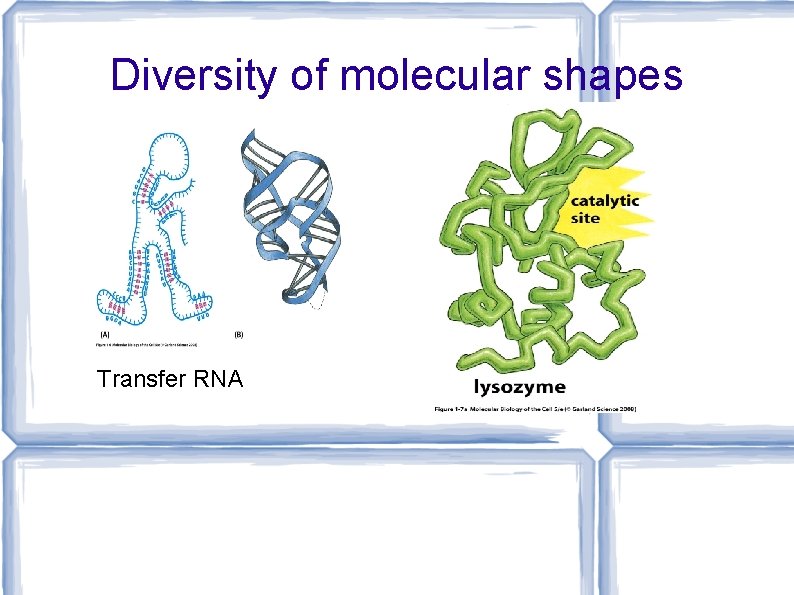
Diversity of molecular shapes Transfer RNA

Hydrolysis & dehydration synthesis ● Hydrolysis ● ● Splits polymer into smaller molecules, using a water molecule. Dehydration synthesis ● Builds polymer from two smaller molecules, releasing water.
- Slides: 10