Functional Groups Things attached to carbon chains Functional

Functional Groups Things attached to carbon chains

Functional Groups Atoms other than hydrogen or carbon covalently bonded to a carbon atom in an organic molecule. n Most commonly oxygen, nitrogen, or the halogens. n The presence of a functional group drastically changes the chemical properties of a molecule. n

Different Functional groups with a 2 carbon chain Ethane- gas (found in natural gas) n Ethanol- grain alcohol (drinkable) n Ethanoic acid- vinegar n Diethyl ether- starting fluid n Chloro fluoro ethane (CFC’s used as refrigerants) n Ethanal- foul smelling liquid (similar to formaldehyde) n
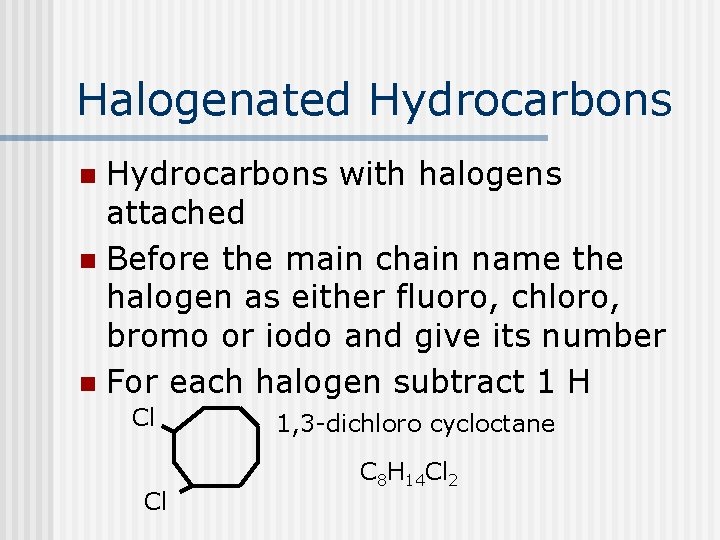
Halogenated Hydrocarbons with halogens attached n Before the main chain name the halogen as either fluoro, chloro, bromo or iodo and give its number n For each halogen subtract 1 H n Cl Cl 1, 3 -dichloro cycloctane C 8 H 14 Cl 2
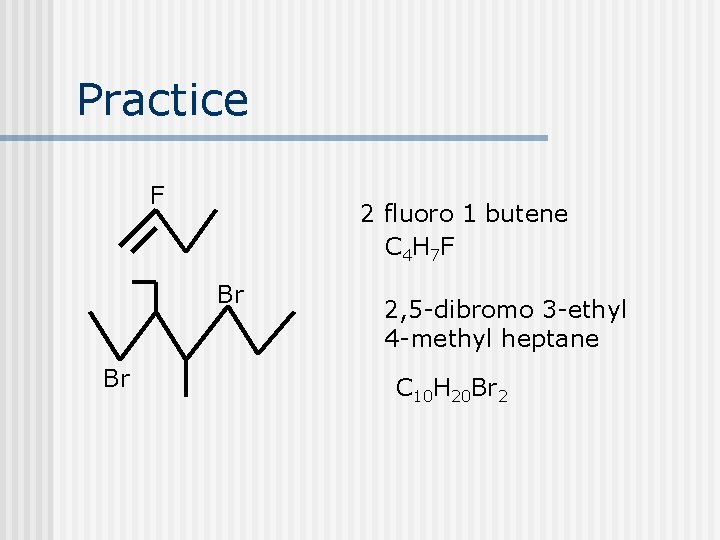
Practice F 2 fluoro 1 butene C 4 H 7 F Br Br 2, 5 -dibromo 3 -ethyl 4 -methyl heptane C 10 H 20 Br 2
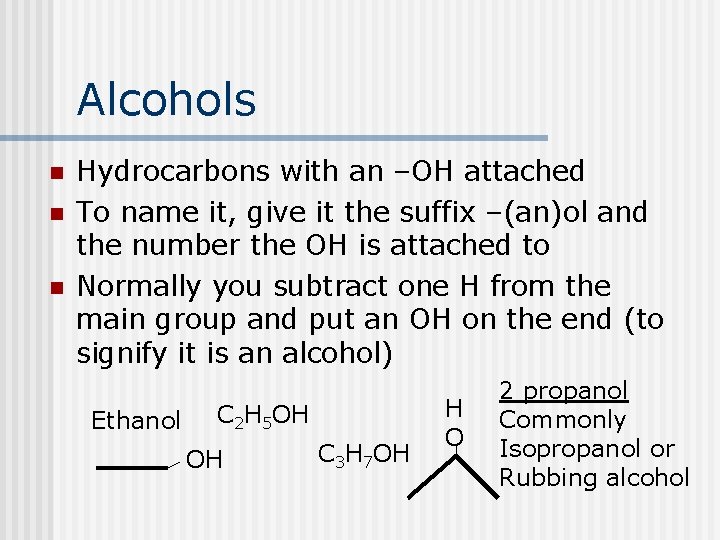
Alcohols n n n Hydrocarbons with an –OH attached To name it, give it the suffix –(an)ol and the number the OH is attached to Normally you subtract one H from the main group and put an OH on the end (to signify it is an alcohol) Ethanol C 2 H 5 OH OH C 3 H 7 OH H O 2 propanol Commonly Isopropanol or Rubbing alcohol
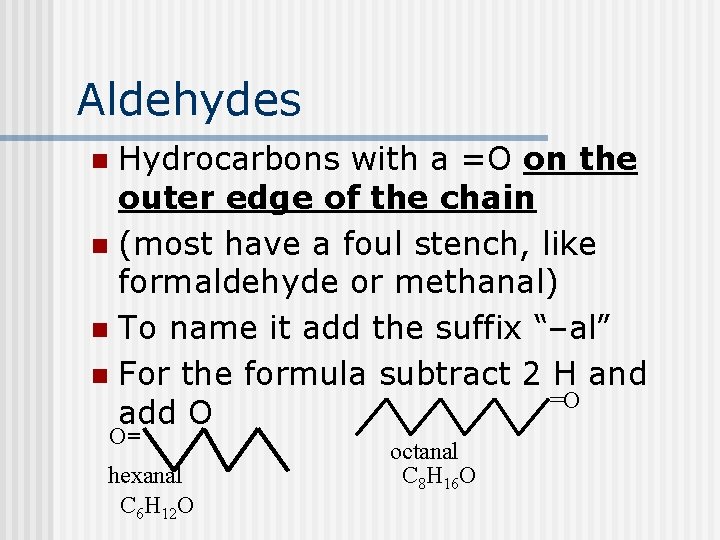
Aldehydes Hydrocarbons with a =O on the outer edge of the chain n (most have a foul stench, like formaldehyde or methanal) n To name it add the suffix “–al” n For the formula subtract 2 H and =O add O n O= hexanal C 6 H 12 O octanal C 8 H 16 O

Ketones Hydrocarbons with a =O not on the edge of the compound n To name it add the suffix “–one” n For the formula subtract 2 H and add O O= cyclopropanone C 3 H 4 O O= n 3 -nonanone C 9 H 18 O

Carboxylic Acid Hydrocarbons with a –COOH group attached n To name it give it the suffix “–oic acid”, the C in the group does count n Subtract one C one H and add COOH n This group looks like… n -C=O O H Pentanoic acid =O O H C 4 H 9 COOH

Everything so far… Alkanes, alkenes, and alkynes n Isomers, halogenated and cyclic n -OH *R means any carbon chain Alcohols Carboxylic Acids R-OH R-C=O -ol -oic acid -al Ketones R-C-R =O Aldehydes on the end R=O -one
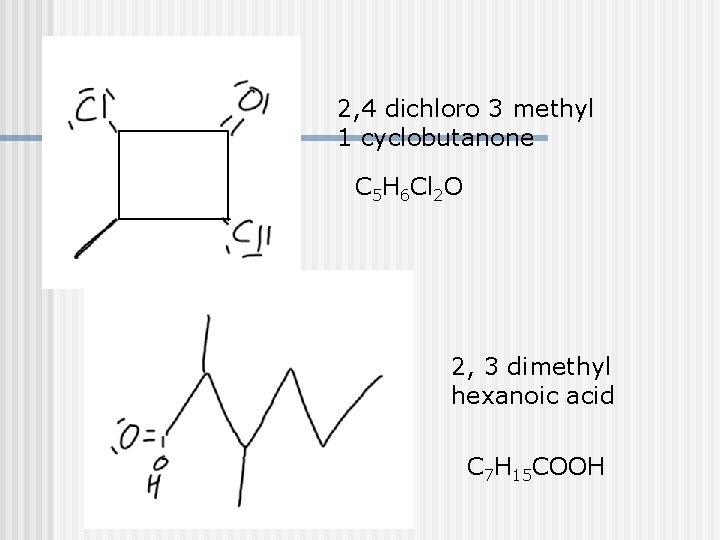
2, 4 dichloro 3 methyl 1 cyclobutanone C 5 H 6 Cl 2 O 2, 3 dimethyl hexanoic acid C 7 H 15 COOH
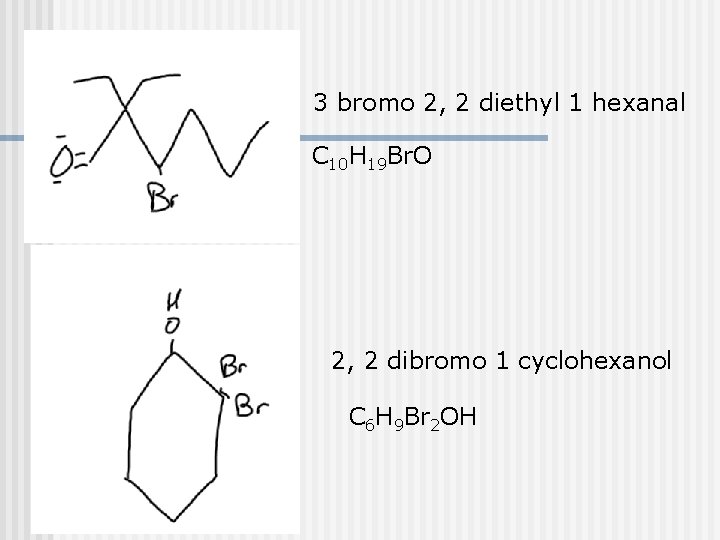
3 bromo 2, 2 diethyl 1 hexanal C 10 H 19 Br. O 2, 2 dibromo 1 cyclohexanol C 6 H 9 Br 2 OH

n 5, 6 dibromo 1 iodo, 3, 4, 5 trimethyl 2 nonanol n 2, 4 difluoro 3 methyl 5, 6, 7 triethyl 1 cyclooctanone
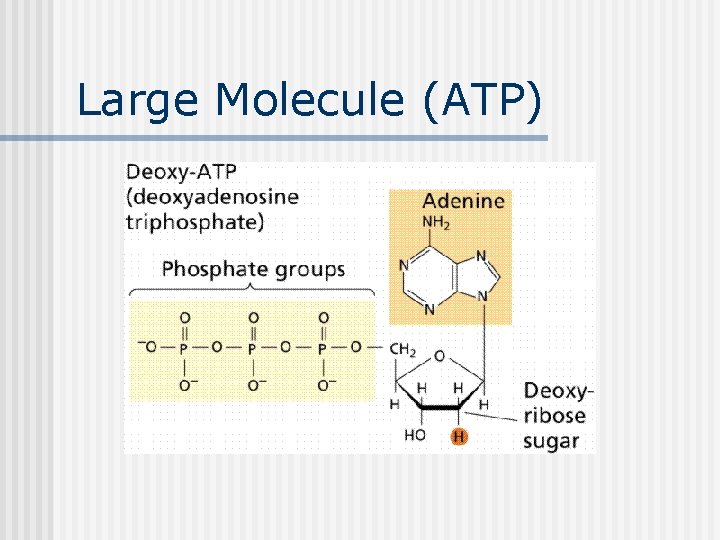
Large Molecule (ATP)
- Slides: 14