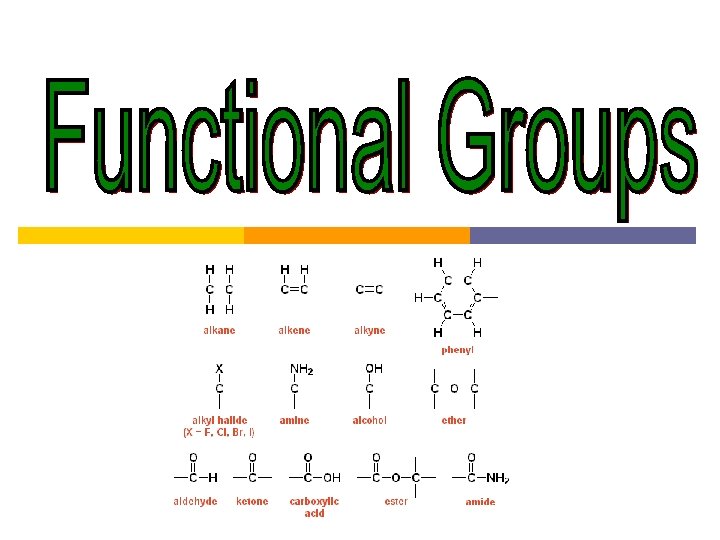
Functional Groups Functional groups special groups of atoms



























- Slides: 31


Functional Groups Functional groups: special groups of atoms attached to a hydrocarbon skeleton; the most common sites of chemical reactivity.

Organic halides: a hydrogen is replaced by a halogen fluoro-, chloro-, bromo-, iodo- 2 -iodobutane

Organic halides: a hydrogen is replaced by a halogen fluoro-, chloro-, bromo-, iodo- 2, 4 -dibromo-1 -hexene

Organic halides: a hydrogen is replaced by a halogen fluoro-, chloro-, bromo-, iodo- 1 -bromo-2 -chlorobenzene

Alcohols & phenols: contain the hydroxyl group (-OH) alcohols: at least 1 H on a hydrocarbon is replaced by OH phenols: at least 1 H on an aromatic ring is replaced by OH 2 -propanol

Alcohols & phenols: contain the hydroxyl group (-OH) alcohols: at least 1 H on a hydrocarbon is replaced by OH phenols: at least 1 H on an aromatic ring is replaced by OH 3 -methyl-1 -butanol

Alcohols & phenols: contain the hydroxyl group (-OH) alcohols: at least 1 H on a hydrocarbon is replaced by OH phenols: at least 1 H on an aromatic ring is replaced by OH 1, 2 -butanediol

ethers: compounds in which an O atom is bonded to 2 organic groups: -C-O-C- methoxymethane (dimethyl ether)

ethers: compounds in which an O atom is bonded to 2 organic groups: -C-O-C- methoxypropane (methyl propyl ether)

ethers: compounds in which an O atom is bonded to 2 organic groups: -C-O-C- methoxybenzene (methyl phenyl ether)

Carboxylic acids: compounds that contain the carboxyl group (general formula is R-COOH) butanoic acid

Carboxylic acids: compounds that contain the carboxyl group (general formula is R-COOH) ethanoic acid

Carboxylic acids: compounds that contain the carboxyl group (general formula is R-COOH) 3 -methylpentanoic acid

Carboxylic acids: compounds that contain the carboxyl group (general formula is R-COOH) benzoic acid

amines: derivatives of ammonia (NH 3) in which 1 or more H atoms are replaced by organic groups (alkyl or aryl groups) ammonia

amines: derivatives of ammonia (NH 3) in which 1 or more H atoms are replaced by organic groups (alkyl or aryl groups) methylamine

amines: derivatives of ammonia (NH 3) in which 1 or more H atoms are replaced by organic groups (alkyl or aryl groups) trimethylamine

amines: derivatives of ammonia (NH 3) in which 1 or more H atoms are replaced by organic groups (alkyl or aryl groups) 2 -aminobutane

amines: derivatives of ammonia (NH 3) in which 1 or more H atoms are replaced by organic groups (alkyl or aryl groups) 1 -amino-3 -propylcyclohexane

*aniline: the simplest aromatic amine aniline

*aniline: the simplest aromatic amine 3, 5 -dichloroaniline Naming it “aniline” make this carbon #1 by definition

*aniline: the simplest aromatic amine N, N-dimethylaniline

Summary of IUPAC rules for naming organic compounds p IUPAC: International Union of Pure and Applied Chemistry p International, non-governmental organization that is best known for its system of nomenclature, which is now recognized as the world authority in this field.

Rule #1: Identify the longest chain of carbon atoms a) The longest chain of carbon atoms gives the stem/root of the name as shown in the table below: # of C-atoms in Stem in longest chain IUPAC name 1 meth 2 3 4 5 6 7 8 9 ethpropbutpenthexheptoctnon- Example (C 2 H 2 n+2 for alkanes) CH 4, methane C 2 H 6, ethane C 3 H 8, propane C 4 H 10, butane C 5 H 12, pentane C 6 H 14, hexane C 7 H 16, heptane C 8 H 18, octane C 9 H 20, nonane

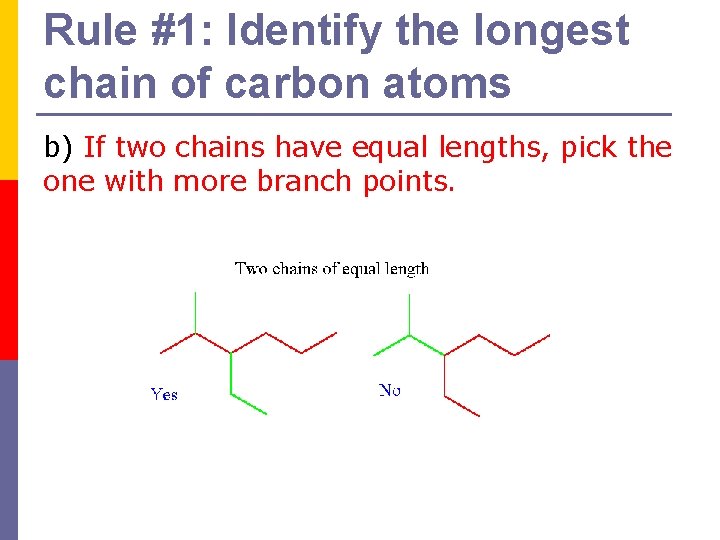
Rule #1: Identify the longest chain of carbon atoms b) If two chains have equal lengths, pick the one with more branch points.

Rule #2: Number the carbons in the main chain Number chain to minimize the position/number of the following in order of priority: a) thing you’re naming the compound after (double bond if alkene; -OH group if alcohol, etc) a) b) c) d) note: for multiple double bonds -diene, -triene, -tetraene first branch/substituent group If both ends have the same first branching number, then number chain to minimize position of second branch (and then third and so on). if still in need of a tie breaker, minimize # of substituent group that comes first alphabetically Note: in cyclic and aromatic (benzene derivatives) compounds, no number needed if only one substituent.

Rule #3: Identify the functional group and attach appropriate suffix p Note: the name for the stem/root is derived from the longest carbon chain, which may include the carbon of the functional group. Indicating position of the functional group: shown by a number inserted before the functional group ending. The number refers to the carbon atom to which the functional group is attached when the chain is numbered starting at the end that will give the smallest number to the group.

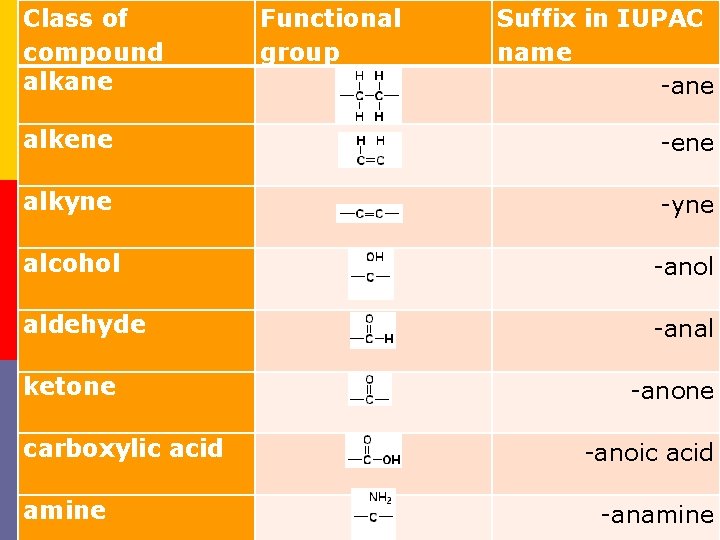
Class of compound alkane Functional group alkene alkyne alcohol aldehyde ketone carboxylic acid amine Suffix in IUPAC name -ane -ene -yne -anol -anal -anone -anoic acid -anamine

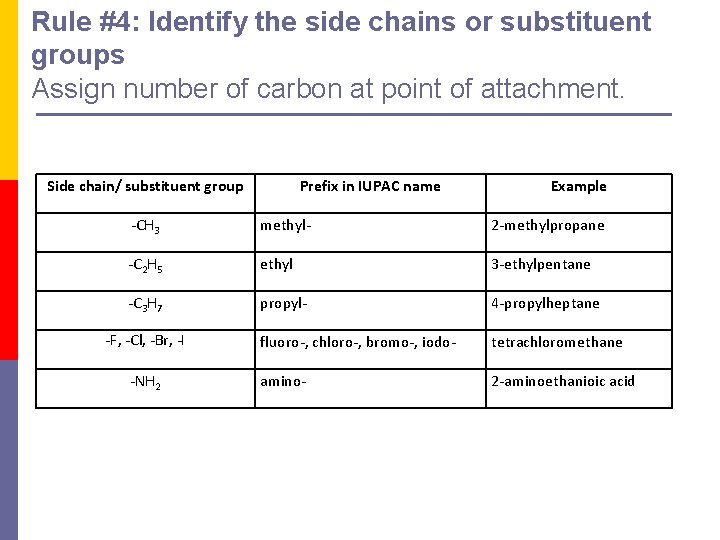
Rule #4: Identify the side chains or substituent groups Assign number of carbon at point of attachment. Side chain/ substituent group Prefix in IUPAC name Example -CH 3 methyl- 2 -methylpropane -C 2 H 5 ethyl 3 -ethylpentane -C 3 H 7 propyl- 4 -propylheptane fluoro-, chloro-, bromo-, iodo- tetrachloromethane amino- 2 -aminoethanioic acid -F, -Cl, -Br, -I -NH 2

Rule #5: Assemble name as a single word #, substituent, root, suffix a) List substituents alphabetically (i. e. butyl- before methyl-) b) If multiples of one substituent are present: “di-, ” “tri-, ” “tetra, ” etc. n Note: “di-, ” “tri-, ” “tetra, ” etc. aren’t part of alphabetical name (triethyl- before dimethyl-) c) punctuation: commas between numbers; hyphens between numbers and letters; merged into one word (exception: acid = word #2 for carboxylic acids)