Functional Groups Functional Groups A group that substitutes

Functional Groups

Functional Groups A group that substitutes a hydrogen in hydrocarbons, changing the chemical properties of the compound. R = some chain of hydrocarbon
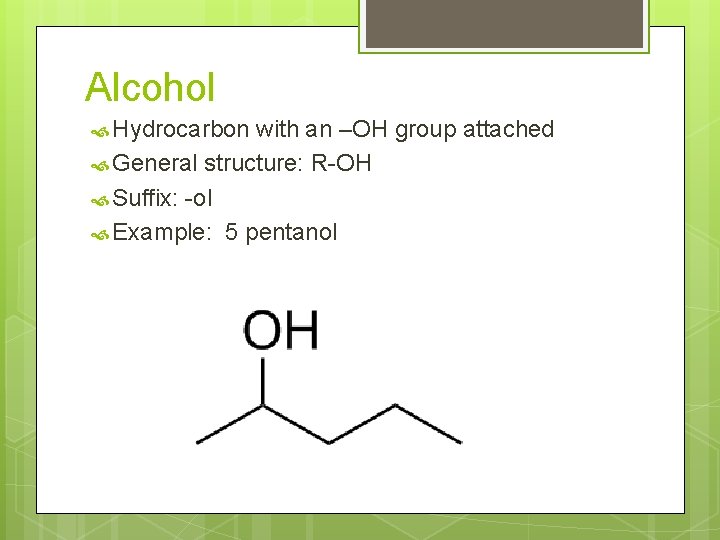
Alcohol Hydrocarbon with an –OH group attached General structure: R-OH Suffix: -ol Example: 5 pentanol
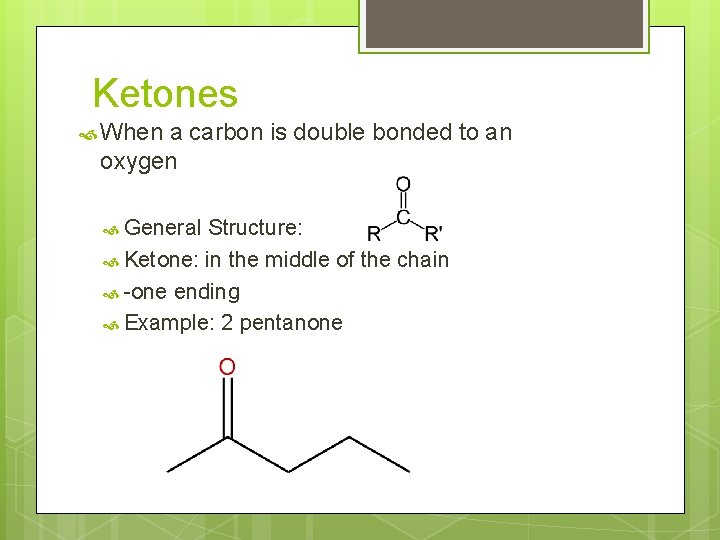
Ketones When a carbon is double bonded to an oxygen General Structure: Ketone: in the middle of the chain -one ending Example: 2 pentanone
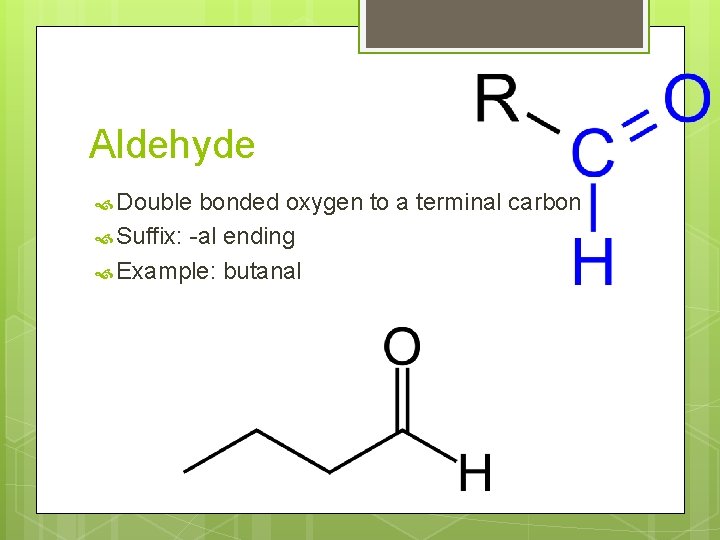
Aldehyde Double bonded oxygen to a terminal carbon Suffix: -al ending Example: butanal
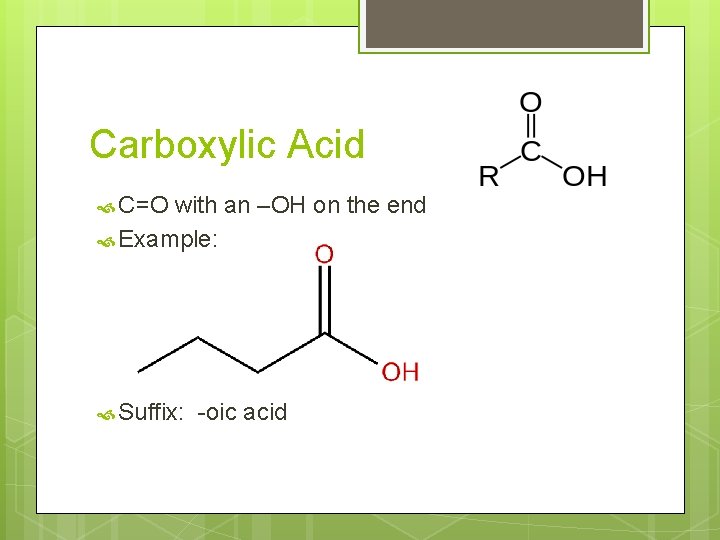
Carboxylic Acid C=O with an –OH on the end Example: Suffix: -oic acid
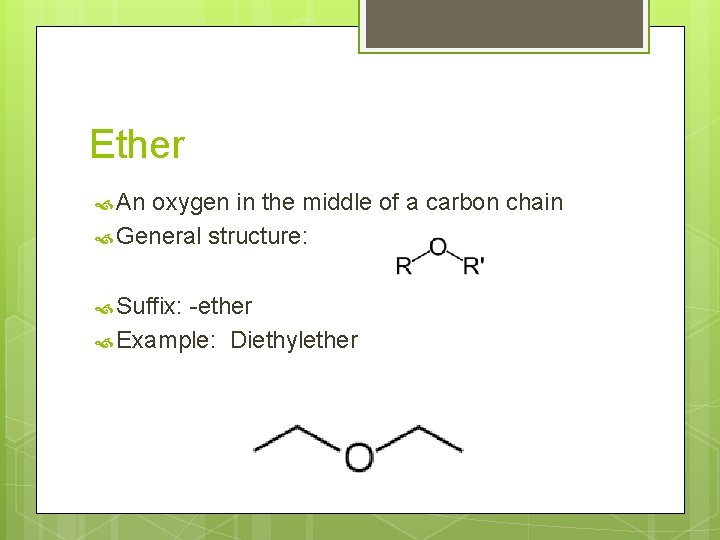
Ether An oxygen in the middle of a carbon chain General structure: Suffix: -ether Example: Diethylether
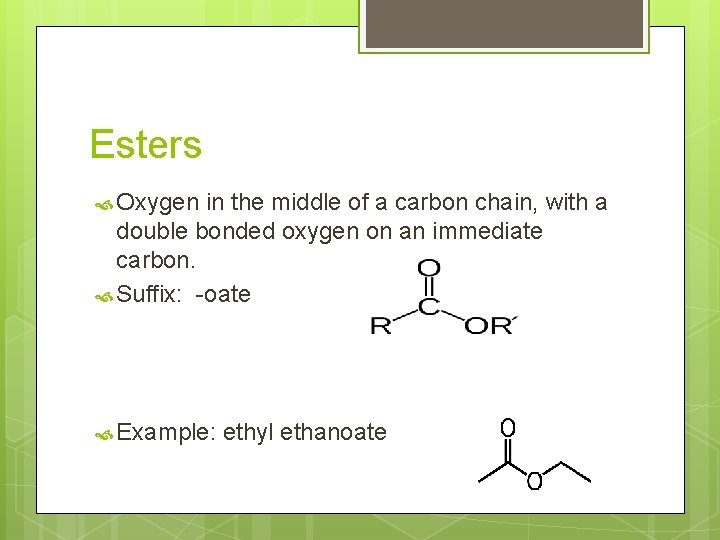
Esters Oxygen in the middle of a carbon chain, with a double bonded oxygen on an immediate carbon. Suffix: -oate Example: ethyl ethanoate
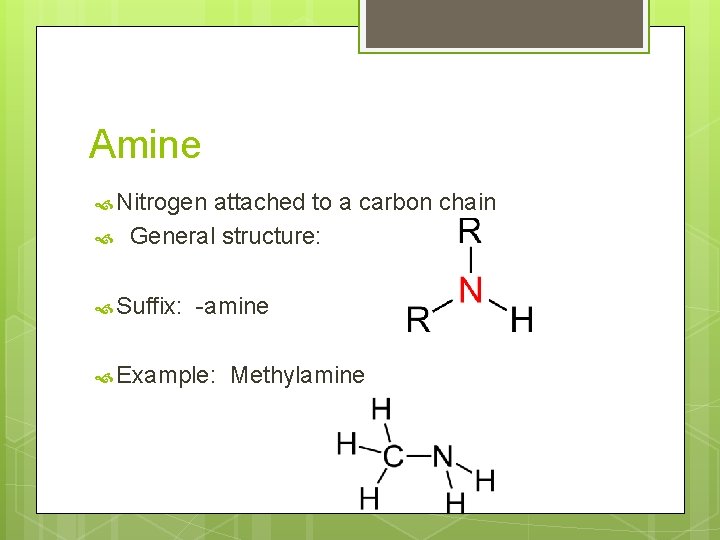
Amine Nitrogen attached to a carbon chain General structure: Suffix: -amine Example: Methylamine
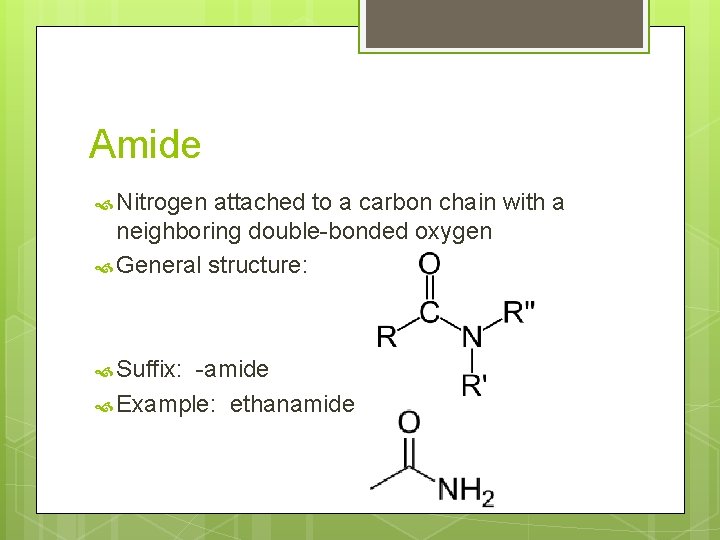
Amide Nitrogen attached to a carbon chain with a neighboring double-bonded oxygen General structure: Suffix: -amide Example: ethanamide

You should be able to…. Recognize all functional groups Name alcohols, ketones, aldehydes and carboxylic acids
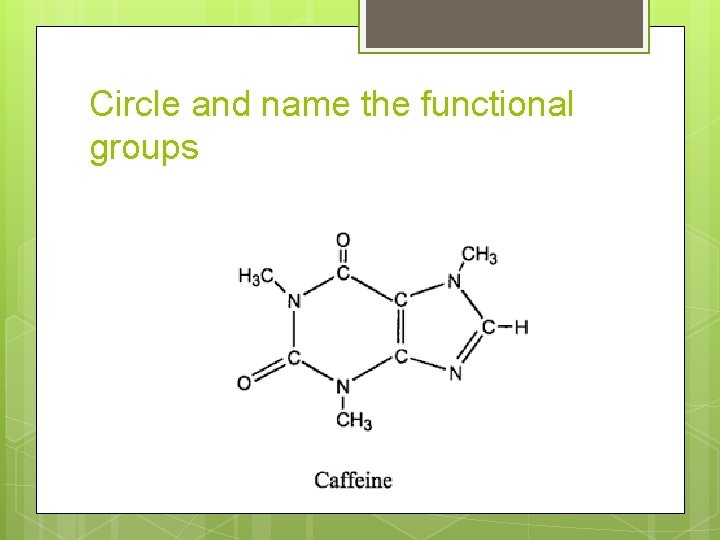
Circle and name the functional groups
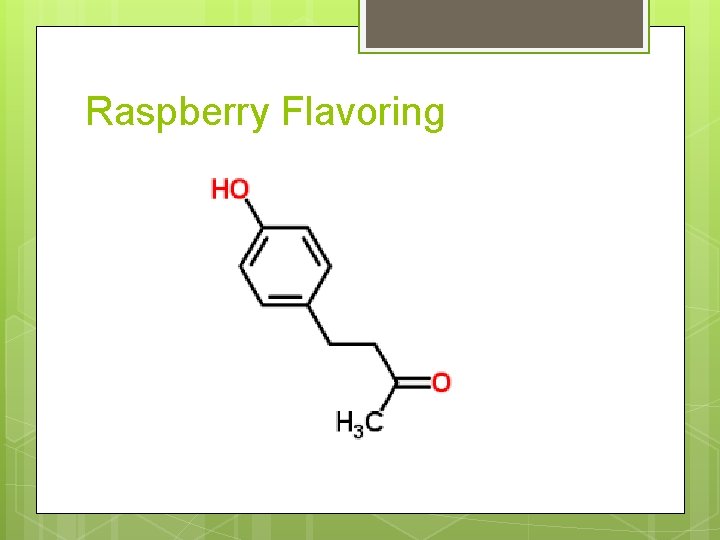
Raspberry Flavoring
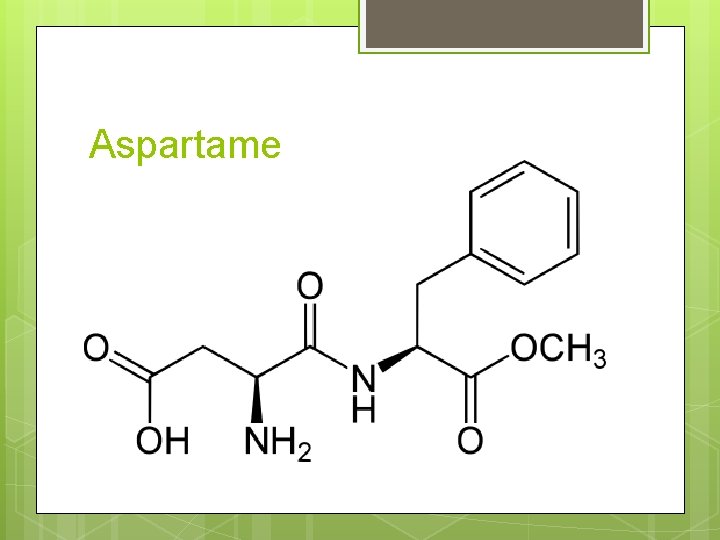
Aspartame
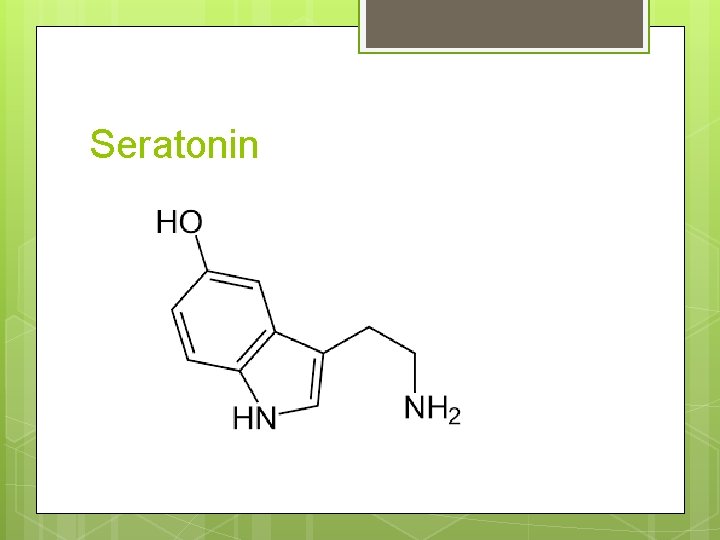
Seratonin
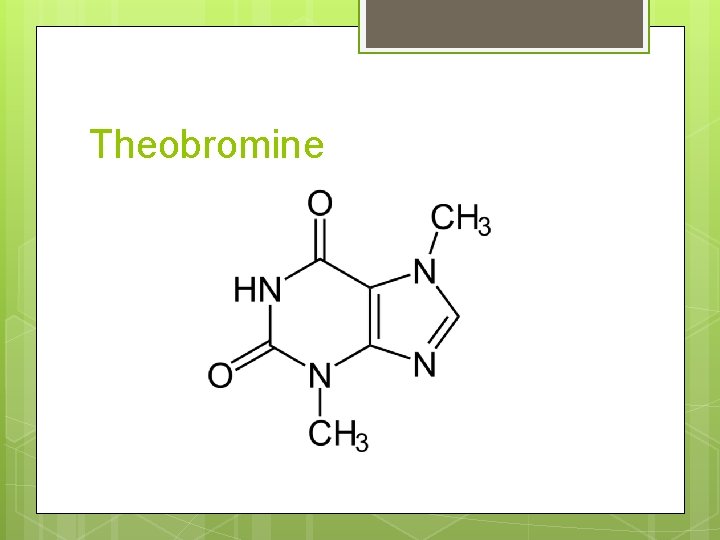
Theobromine
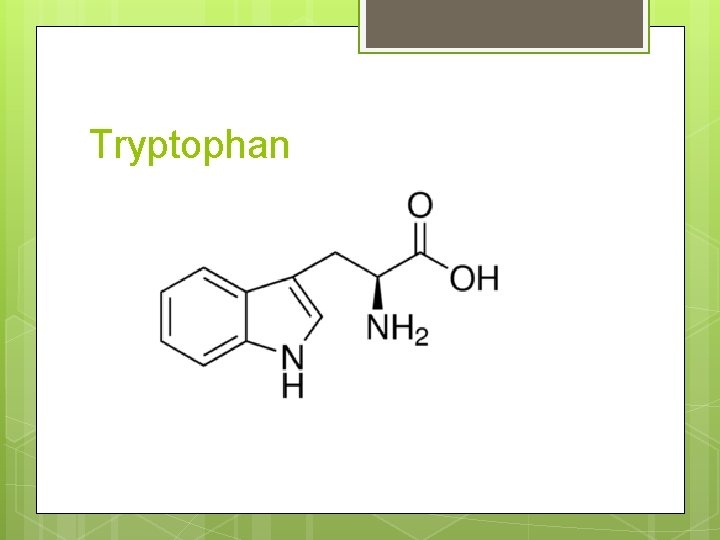
Tryptophan
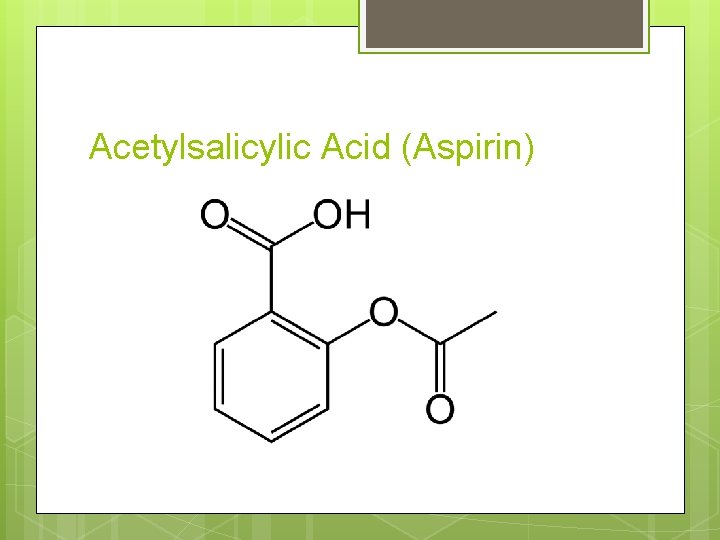
Acetylsalicylic Acid (Aspirin)
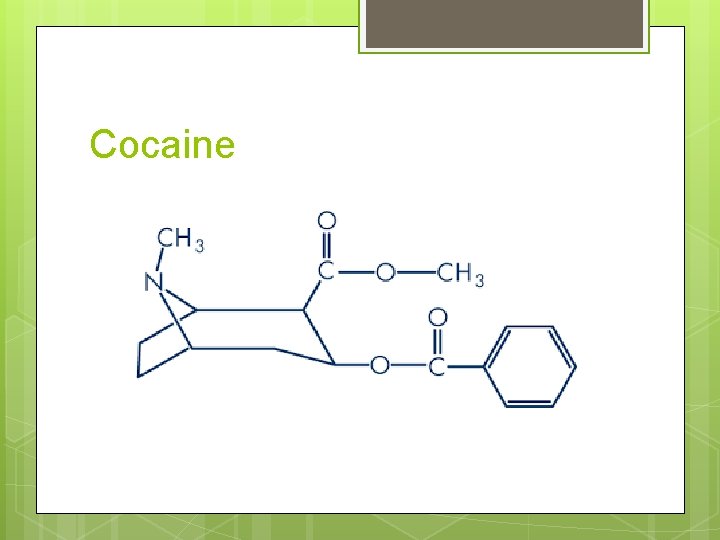
Cocaine
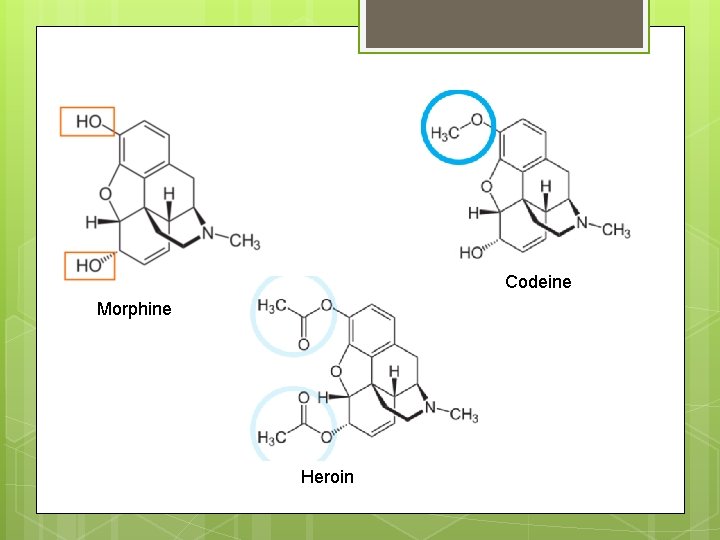
Codeine Morphine Heroin
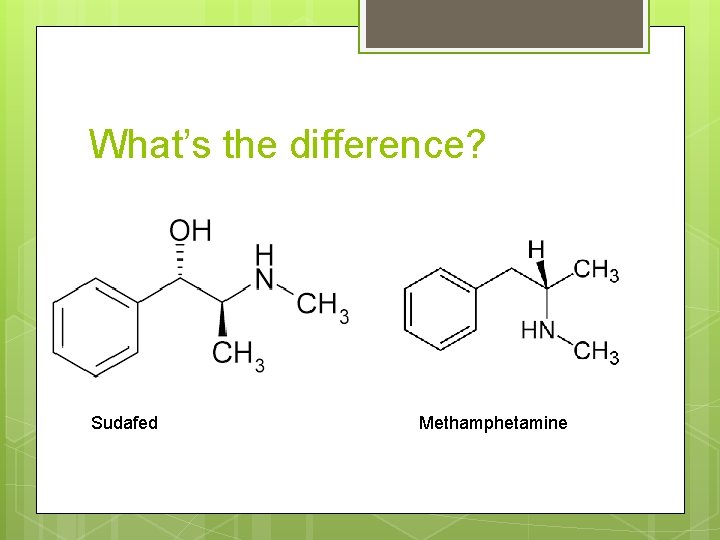
What’s the difference? Sudafed Methamphetamine
- Slides: 21