Full Starting Dose in a Single Vial Combating

Full Starting Dose in a Single Vial Combating Another Side of Smoke Cyanide Poisoning and How to Treat It Using the Single-Vial CYANOKIT® 5 g (hydroxocobalamin for injection)

Table of Contents Identifying Cyanide Poisoning 5 CYANOKIT® 5 g (hydroxocobalamin for injection) 11 How to Administer CYANOKIT® 17 Storing and Stocking CYANOKIT® 22 Grant Information 27 Full Prescribing Information 35 2 Please see Important Safety Information on slide 3 and full Prescribing Information for CYANOKIT® on slide 35.

Indication and Important Safety Information Indication CYANOKIT® (hydroxocobalamin for injection) 5 g for intravenous infusion is indicated for the treatment of known or suspected cyanide poisoning. If clinical suspicion of cyanide poisoning is high, CYANOKIT® should be administered without delay. Important Safety Information Cyanide poisoning may result from inhalation, ingestion, or dermal exposure. Prior to administration of CYANOKIT®, smoke-inhalation victims should be assessed for: exposure to fire or smoke in an enclosed area; presence of soot around the mouth, nose, or oropharynx; and altered mental status. In addition to CYANOKIT®, treatment of cyanide poisoning must include immediate attention to airway patency, adequacy of oxygenation and hydration, cardiovascular support, and management of any seizure activity. Use caution in the management of patients with known anaphylactic reactions to hydroxocobalamin or cyanocobalamin. Allergic reactions may include: anaphylaxis, chest tightness, edema, urticaria, pruritus, dyspnea, rash, and angioneurotic edema. Please see full Prescribing Information for CYANOKIT®. 3

Important Safety Information (cont’d) Substantial increases in blood pressure may occur following CYANOKIT® therapy. Usage may interfere with some clinical laboratory evaluations. Also, because of its deep red color, hydroxocobalamin may cause hemodialysis machines to shut down due to an erroneous detection of a “blood leak. ” This should be considered before hemodialysis is initiated in patients treated with hydroxocobalamin. Due to potential photosensitivity, patients should avoid direct sun until erythema resolves. CYANOKIT® is Pregnancy Category C and should be used during pregnancy only if the potential benefit justifies the potential risk. Safety and effectiveness of CYANOKIT® have not been established in pediatric patients. The most common adverse reactions (>5%) are transient and include chromaturia, erythema, rash (predominantly acneiform), increased blood pressure, nausea, headache, decreased lymphocyte percentage, and injection site reactions. Please see full Prescribing Information for CYANOKIT®. 4

Identifying Cyanide Poisoning A hidden threat in some closed-space fires Please see Important Safety Information on slide 3 and full Prescribing Information for CYANOKIT® on slide 35. ►Click for Table of Contents

Cyanide Is an Often Unrecognized Danger in Some Closed-Space Fires Common Building Materials Known to Release High Levels of Cyanide During Combustion*1 • Cyanide (CN) can be released by virtually any material containing carbon and nitrogen when burned under high temperature and low oxygen conditions 2 • There is potential for CN toxicity due to the increased use of synthetic materials that produce CN during combustion in closed-space fires 2, 3 Important Note Not all smoke inhalation victims will have cyanide poisoning and may present with burns, trauma, and exposure to other toxic substances making a diagnosis of cyanide poisoning particularly difficult. *Items pictured may not be accurate representations. 1. Hertzberg T, Blomqvist P, Dalene M, Skarping G. Particles and Isocyanates From Fires. Borås, Sweden: SP Swedish National Testing and Research Institute; 2005: 1 -51. 2. Agency for Toxic Substances and Disease Registry. Cyanide toxicity. Am Fam Physician. 1993; 48(1): 107 -114. 3. Alcorta R. Smoke inhalation & acute cyanide poisoning. In: Losavio K, ed. Smoke Inhalation & Hydrogen Cyanide Poisoning. San Diego, CA: JEMS Communications; 2004: 6 -17. 6 Please see Important Safety Information on slide 3 and full Prescribing Information for CYANOKIT® on slide 35.

Difficult to Diagnose, Easy to Suspect Distinguishing between cyanide and carbon monoxide poisoning can be difficult Moderate to Severe Poisoning Signs/Symptoms 1, 2 BECAUSE… • Many symptoms of cyanide (CN) and carbon monoxide poisoning are common to both or very similar, 1 and • There is no diagnostic test to confirm or distinguish cyanide poisoning from carbon monoxide poisoning in the field 1 CYANIDE POISONING… • Should be suspected in any person exposed to smoke in a closed-space fire, 1, 3 and • When suspicion of cyanide poisoning is high, an antidote should be considered for empiric treatment 1, 3 1. O'Brien DJ, Augustine J, Walsh DW. Cyanide exposure, smoke inhalation, and pre-hospital treatment: recognizing the signs and symptoms and available treatment options. In: Smoke: Cyanide and Carbon Monoxide: The Toxic Twins of Smoke Inhalation. Cyanide Poisoning Treatment Coalition: Indianapolis, IN: 2009: 20 -24. 2. Lawson-Smith P, Jansen EC, Hyldegaard O. Cyanide intoxication as part of smoke inhalation—a review on diagnosis and treatment from the emergency perspective. Scand J Trauma Resusc Emerg Med. 2011; 19: 1 -5. 3. Eckstein M, Maniscalco PM. Focus on smoke inhalation—the most common cause of acute cyanide poisoning. Prehosp Disaster Med. 2006; 21(2): s 49 -s 55. 7 Please see Important Safety Information on slide 3 and full Prescribing Information for CYANOKIT® on slide 35.

When to Suspect Cyanide Poisoning*1, 2 Cyanide poisoning in smoke-inhalation victims should be suspected if the following manifestations are present*1, 2: R Exposure to fire or smoke in an enclosed area R Soot around mouth, nose, or back of mouth R Altered mental status (eg, confusion, disorientation) *List may not be comprehensive. Important Note In some settings, panic symptoms including tachypnea and vomiting may mimic early cyanide poisoning signs. The presence of altered mental status (eg, confusion and disorientation) and/or mydriasis is suggestive of true cyanide poisoning although these signs can occur with other toxic exposures as well. 1. Eckstein M, Maniscalco PM. Focus on smoke inhalation—the most common cause of acute cyanide poisoning. Prehosp Disaster Med. 2006; 21(2): s 49 -s 55. 2. Cyanokit (single 5 -g vial) [package insert]. Columbia, MD: Meridian Medical Technologies, Inc. ; 2011. 8 Please see Important Safety Information on slide 3 and full Prescribing Information for CYANOKIT® on slide 35.
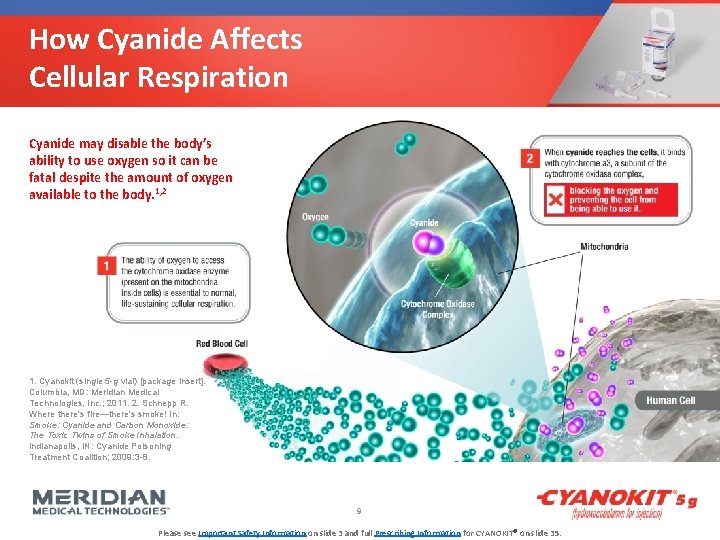
How Cyanide Affects Cellular Respiration Cyanide may disable the body’s ability to use oxygen so it can be fatal despite the amount of oxygen available to the body. 1, 2 1. Cyanokit (single 5 -g vial) [package insert]. Columbia, MD: Meridian Medical Technologies, Inc. ; 2011. 2. Schnepp R. Where there's fire—there's smoke! In: Smoke: Cyanide and Carbon Monoxide: The Toxic Twins of Smoke Inhalation. Indianapolis, IN: Cyanide Poisoning Treatment Coalition; 2009: 3 -8. 9 Please see Important Safety Information on slide 3 and full Prescribing Information for CYANOKIT® on slide 35.

How Hydroxocobalamin Interacts With Cyanide CYANOKIT® 5 g (hydroxocobalamin for injection) is the only cyanide antidote that contains hydroxocobalamin, which binds to cyanide, allowing the body to use oxygen again. 1, 2 1. Cyanokit (single 5 -g vial) [package insert]. Columbia, MD: Meridian Medical Technologies, Inc. ; 2011. 2. Schnepp R. Where there's fire—there's smoke! In: Smoke: Cyanide and Carbon Monoxide: The Toxic Twins of Smoke Inhalation. Indianapolis, IN: Cyanide Poisoning Treatment Coalition; 2009: 3 -8. 10 Please see Important Safety Information on slide 3 and full Prescribing Information for CYANOKIT® on slide 35.
CYANOKIT® 5 g (hydroxocobalamin for injection) FDA-approved for the treatment of known or suspected cyanide poisoning. Please see Important Safety Information on slide 3 and full Prescribing Information for CYANOKIT® on slide 35. ►Click for Table of Contents

Indication 1 • CYANOKIT® 5 g (hydroxocobalamin for injection) is indicated for the treatment of known or suspected cyanide poisoning • If clinical suspicion of cyanide poisoning is high, CYANOKIT® should be administered without delay Important Safety Information In addition to CYANOKIT®, treatment of cyanide poisoning must include immediate attention to airway patency, adequacy of oxygenation and hydration, cardiovascular support, and management of any seizure activity. Consideration should be given to decontamination measures based on the route of exposure. 1. Cyanokit (single 5 -g vial) [package insert]. Columbia, MD: Meridian Medical Technologies, Inc. ; 2011. 12 Please see Important Safety Information on slide 3 and full Prescribing Information for CYANOKIT® on slide 35.

For Known or Suspected Cyanide Poisoning 1 • It has been shown that the concentration of blood lactate increases proportionally with the severity of cyanide (CN) poisoning and that hydroxocobalamin treatment results in rapid resolution of CN-induced lactic acidemia 13 • Studies have shown CYANOKIT® 5 g (hydroxocobalamin for injection) to be generally well-tolerated even in the absence of cyanide poisoning 4 -7 • Has been used in France since 1996 and in the U. S. since 2007 to treat suspected or known cyanide poisoning 8 Important Safety Information Substantial increases in blood pressure may occur following CYANOKIT® therapy. Usage may interfere with some clinical laboratory evaluations. Also, because of its deep red color, hydroxocobalamin may cause hemodialysis machines to shut down due to an erroneous detection of a “blood leak. ” This should be considered before hemodialysis is initiated in patients treated with hydroxocobalamin. 1. Lawson-Smith P, Jansen EC, Hyldegaard O. Cyanide intoxication as part of smoke inhalation—a review on diagnosis and treatment from the emergency perspective. Scand J Trauma Resusc Emerg Med. 2011; 19: 1 -5. 10. 2. Baud FJ, Borron SW, Mégarbane B, et al. Value of lactic acidosis in the assessment of the severity of acute cyanide poisoning. Crit Care Med. 2002; 30(9): 2044 -2050. 3. Torres EC, Bustamante RS, Granizo EG, et al. Hydroxocobalamin and lactate concentration in patients suspected of having cyanuric acid poisoning related to smoke inhalation syndrome. Emergencias. 2010; 22: 9 -14. 4. Cyanokit (single 5 -g vial) [package insert]. Columbia, MD: Meridian Medical Technologies, Inc. ; 2011. 5. Borron SW, Baud FJ, Barriot P, Imbert M, Bismuth C. Prospective study of hydroxocobalamin for acute cyanide poisoning in smoke inhalation. Ann Emerg Med. 2007; 49(6): 794 -801. 6. Borron SW, Baud FJ, Mégarbane B, Bismuth C. Hydroxocobalamin for severe acute cyanide poisoning by ingestion or inhalation. Am J Emerg Med. 2007; 25(5): 551 -558. 7. Fortin JL, Giocanti JP, Ruttimann M, Kowalski JJ. Prehospital administration of hydroxocobalamin for smoke inhalation-associated cyanide poisoning: 8 years of experience in the Paris Fire Brigade. Clin Toxicol (Phila). 2006; 44(suppl 1): 37 -44. 8. Borron SW. Recognition and treatment of acute cyanide poisoning. J Emerg Nurs. 2006; 32(4 suppl): s 12 -s 18. 13 Please see Important Safety Information on slide 3 and full Prescribing Information for CYANOKIT® on slide 35.

Contents of Single-Vial CYANOKIT® 5 g (hydroxocobalamin for injection)1 1. Cyanokit (single 5 -g vial) [package insert]. Columbia, MD: Meridian Medical Technologies, Inc. ; 2011. 14 Please see Important Safety Information on slide 3 and full Prescribing Information for CYANOKIT® on slide 35.

Compared to the Two-Vial Version… The Single-Vial CYANOKIT® (hydroxocobalamin for injection) 5 g Features: • The full starting dose in a single vial • A longer shelf life from the date of formulation • More compact packaging 15 Please see Important Safety Information on slide 3 and full Prescribing Information for CYANOKIT® on slide 35.

Recommended Dosing 1 • The starting dose of CYANOKIT® 5 g (hydroxocobalamin for injection) for adults is 5 g – One 5 -g vial – Administer by intravenous (IV) infusion over 15 minutes • Depending upon the severity of the poisoning and the clinical response, a second dose of 5 g may be administered by IV infusion up to a total dose of 10 g – The rate of infusion for a potential second dose may range from 15 minutes (for patients in extremis) to 2 hours, as clinically indicated Important Safety Information Use caution in the management of patients with known anaphylactic reactions to hydroxocobalamin or cyanocobalamin. Allergic reactions may include anaphylaxis, chest tightness, edema, urticaria, pruritus, dyspnea, rash, and angioneurotic edema. Substantial increases in blood pressure may occur following treatment with CYANOKIT®. 1. Cyanokit (single 5 -g vial) [package insert]. Columbia, MD: Meridian Medical Technologies, Inc. ; 2011. 16 Please see Important Safety Information on slide 3 and full Prescribing Information for CYANOKIT® on slide 35.

How to Administer Single-Vial CYANOKIT® 5 g (hydroxocobalamin for injection) Prepare and administer in 3 steps. Please see Important Safety Information on slide 3 and full Prescribing Information for CYANOKIT® on slide 35. ►Click for Table of Contents

Step 1: RECONSTITUTE 1 • Place the vial in an upright position • Add 200 m. L of 0. 9% Sodium Chloride Injection to the vial using the sterile transfer spike. Fill to the line. – 0. 9% Sodium Chloride Injection is the recommended diluent – Diluent is not included in the kit – Lactated Ringer's Solution and 5% Dextrose Injection have also been found to be compatible with hydroxocobalamin 1. Cyanokit (single 5 -g vial) [package insert]. Columbia, MD: Meridian Medical Technologies, Inc. ; 2011. 18 Please see Important Safety Information on slide 3 and full Prescribing Information for CYANOKIT® on slide 35.

Step 2: MIX 1 • The vial should be repeatedly inverted or rocked, not shaken, for at least 60 seconds prior to infusion 1. Cyanokit (single 5 -g vial) [package insert]. Columbia, MD: Meridian Medical Technologies, Inc. ; 2011. 19 Please see Important Safety Information on slide 3 and full Prescribing Information for CYANOKIT® on slide 35.

Step 3: INFUSE VIAL 1 • Use vented intravenous tubing • Hang and infuse over 15 minutes One 5 -g vial is a complete starting dose. 1. Cyanokit (single 5 -g vial) [package insert]. Columbia, MD: Meridian Medical Technologies, Inc. ; 2011. 20 Please see Important Safety Information on slide 3 and full Prescribing Information for CYANOKIT® on slide 35.

Considerations During Administration 1 • Inspect CYANOKIT® 5 g (hydroxocobalamin for injection) solution for particulate matter and color prior to administration – Discard if particulate matter is present – Discard if solution is not dark red • Use a separate IV line for infusion – A separate line is required as a number of drugs used in resuscitation efforts are incompatible with CYANOKIT® • Use caution when administering other cyanide antidotes simultaneously with CYANOKIT® – If the decision is made to administer another antidote, do not use the same IV line Important Safety Information In addition to CYANOKIT®, treatment of cyanide poisoning must include immediate attention to airway patency, adequacy of oxygenation and hydration, cardiovascular support, and management of any seizure activity. Consideration should be given to decontamination measures based on the route of exposure. 1. Cyanokit (single 5 -g vial) [package insert]. Columbia, MD: Meridian Medical Technologies, Inc. ; 2011. 21 Please see Important Safety Information on slide 3 and full Prescribing Information for CYANOKIT® on slide 35.

Storing and Stocking CYANOKIT® 5 g (hydroxocobalamin for injection) Please see Important Safety Information on slide 3 and full Prescribing Information for CYANOKIT® on slide 35. ►Click for Table of Contents

Storage of Lyophilized Form 1 • CYANOKIT® 5 g (hydroxocobalamin for injection) should be stored at 25°C (77°F) – Excursions are permitted to 15° to 30°C (59° to 86°F) • CYANOKIT® may be exposed, during short periods, to temperature variations of usual transport, transport in the desert, and freezing/defrosting cycles – Usual transport defined as 15 days subjected to temperatures ranging from 5° to 40°C (41° to 104°F) – Transport in the desert defined as 4 days subjected to temperatures ranging from 5° to 60°C (41° to 140°F) – Freezing/defrosting cycles defined as 15 days subjected to temperatures ranging from – 20° to 40°C (– 4° to 104°F) 1. Cyanokit (single 5 -g vial) [package insert]. Columbia, MD: Meridian Medical Technologies, Inc. ; 2011. 23 Please see Important Safety Information on slide 3 and full Prescribing Information for CYANOKIT® on slide 35.

Storage of Reconstituted Drug Product 1 • CYANOKIT® 5 g (hydroxocobalamin for injection) may be kept up to 6 hours at a temperature not exceeding 40°C (104°F) • Do not freeze • Product not used by 6 hours should be discarded 1. Cyanokit (single 5 -g vial) [package insert]. Columbia, MD: Meridian Medical Technologies, Inc. ; 2011. 24 Please see Important Safety Information on slide 3 and full Prescribing Information for CYANOKIT® on slide 35.

Prehospital Stocking • Reasons to consider stocking CYANOKIT® 5 g (hydroxocobalamin for injection) directly on first responder vehicles: – Existence of cyanide in fire smoke from closed-space fires 1 – Toxic threat cyanide presents to fire victims as well as firefighters 1, 2 § Death can occur within minutes of exposure to moderate to high concentrations of cyanide 1 – Apparent low risk of causing harm by administering hydroxocobalamin for prehospital empiric treatment of smoke inhalation-associated cyanide poisoning 1 Important Safety Information Use caution in the management of patients with known anaphylactic reactions to hydroxocobalamin or cyanocobalamin. Allergic reactions may include anaphylaxis, chest tightness, edema, urticaria, pruritus, dyspnea, rash, and angioneurotic edema. Substantial increases in blood pressure may occur following treatment with CYANOKIT®. 1. Eckstein M, Maniscalco PM. Focus on smoke inhalation—the most common cause of acute cyanide poisoning. Prehosp Disaster Med. 2006; 21(2): s 49 -s 55. 2. Walsh DW, Eckstein M. Hydrogen cyanide in fire smoke: an underappreciated threat. Emerg Med Serv. 2004; 33(10): 160 -163. 25 Please see Important Safety Information on slide 3 and full Prescribing Information for CYANOKIT® on slide 35.

Hospital Stocking • It has been recommended by an expert panel of physicians for consensus guidelines for stocking antidotes that hydroxocobalamin be stocked in the emergency department 1 – It must be available immediately because the administration of a cyanide antidote can resuscitate a patient only of it is given before irreversible injury develops 1 Important Safety Information Usage may interfere with some clinical laboratory evaluations. Also, because of its deep red color, hydroxocobalamin may cause hemodialysis machines to shut down due to an erroneous detection of a “blood leak. ” This should be considered before hemodialysis is initiated in patients treated with hydroxocobalamin. Due to potential photosensitivity, patients should avoid direct sun until erythema resolves. 1. Dart RC, Borron SW, Caravati EM, et al. Expert consensus guidelines for stocking of antidotes in hospitals that provide emergency care. Ann Emerg Med. 2009; 54: 386 -394. 26 Please see Important Safety Information on slide 3 and full Prescribing Information for CYANOKIT® on slide 35.

Grant Information Please see Important Safety Information on slide 3 and full Prescribing Information for CYANOKIT® on slide 35. ►Click for Table of Contents

Grant Eligibility • CYANOKIT® 5 g (hydroxocobalamin for injection) is approved on the Department of Homeland Security's Authorized Equipment List (AEL), making it eligible for the following grant programs 1: • – State Homeland Security Program (SHSP) – Urban Area Security Initiative (UASI) – Metropolitan Medical Response System (MMRS) – Transit Security Grant Program (TSGP) Additional grant information and resources can be found at: – emsgrantshelp. com – firegrantshelp. com – usfa. dhs. gov/grants – fema. gov/firegrants/ – usfa. dhs. gov/fireservice/grants/afgp These Web sites are neither owned nor controlled by Meridian is not responsible for the content or services of these sites. 1. Department of Homeland Security. Authorized equipment list. https: //www. rkb. us/mel. cfm? subtypeid=549. Accessed September 2, 2011. 28 Please see Important Safety Information on slide 3 and full Prescribing Information for CYANOKIT® on slide 35.

Questions? For more information about CYANOKIT® 5 g (hydroxocobalamin for injection): Call 1 -800 -638 -8093 or Visit CYANOKIT. com For medical questions concerning CYANOKIT® call 1 -800 -438 -1985, Monday through Friday, 8: 00 am to 8: 00 pm EST. To report an adverse event or speak to a member of Pfizer U. S. Medical Information, call 1 -800 -438 -1985. 29 Please see Important Safety Information on slide 3 and full Prescribing Information for CYANOKIT® on slide 35.

Cyanide Poisoning 1, 2 SUSPECT IT. * Cyanide poisoning in smoke-inhalation victims should be suspected if the following manifestations are present*1, 2: R Exposure to fire or smoke in an enclosed area R Soot around mouth, nose, or back of mouth R Altered mental status (eg, confusion, disorientation) Cyanide poisoning should be suspected. *List may not be comprehensive. Important Safety Information Cyanide poisoning may result from inhalation, ingestion, or dermal exposure. Prior to administration of CYANOKIT®, smoke-inhalation victims should be assessed for: exposure to fire or smoke in an enclosed area; presence of soot around the mouth, nose, or oropharynx; and altered mental status. In addition to CYANOKIT®, treatment of cyanide poisoning must include immediate attention to airway patency, adequacy of oxygenation and hydration, cardiovascular support, and management of any seizure activity. 1. Eckstein M, Maniscalco PM. Focus on smoke inhalation—the most common cause of acute cyanide poisoning. Prehosp Disaster Med. 2006; 21(2): s 49 -s 55. 2. Cyanokit (single 5 -g vial) [package insert]. Columbia, MD: Meridian Medical Technologies, Inc. ; 2011. 30 Please see Important Safety Information on slide 3 and full Prescribing Information for CYANOKIT® on slide 35.

Cyanide Poisoning 1 TREAT IT. Prepare and administer CYANOKIT® 5 g (hydroxocobalamin for injection) in 3 steps 1: 1. Reconstitute Place the vial in an upright position. Add 200 m. L of 0. 9% Sodium Chloride Injection to the vial using the transfer spike. Fill to the line. 2. Mix 3. Infuse vial The vial should be repeatedly inverted or rocked, not shaken, for at least 60 seconds prior to infusion. Use vented intravenous tubing, hang and infuse over 15 minutes. 1. Cyanokit (single 5 -g vial) [package insert]. Columbia, MD: Meridian Medical Technologies, Inc. ; 2011. 31 Please see Important Safety Information on slide 3 and full Prescribing Information for CYANOKIT® on slide 35.

Indication and Important Safety Information Indication CYANOKIT® (hydroxocobalamin for injection) 5 g for intravenous infusion is indicated for the treatment of known or suspected cyanide poisoning. If clinical suspicion of cyanide poisoning is high, CYANOKIT® should be administered without delay. Important Safety Information Cyanide poisoning may result from inhalation, ingestion, or dermal exposure. Prior to administration of CYANOKIT®, smoke-inhalation victims should be assessed for: exposure to fire or smoke in an enclosed area; presence of soot around the mouth, nose, or oropharynx; and altered mental status. In addition to CYANOKIT®, treatment of cyanide poisoning must include immediate attention to airway patency, adequacy of oxygenation and hydration, cardiovascular support, and management of any seizure activity. Use caution in the management of patients with known anaphylactic reactions to hydroxocobalamin or cyanocobalamin. Allergic reactions may include: anaphylaxis, chest tightness, edema, urticaria, pruritus, dyspnea, rash, and angioneurotic edema. Please see full Prescribing Information for CYANOKIT®. ►Click for Table of Contents 32

Important Safety Information (cont’d) Substantial increases in blood pressure may occur following CYANOKIT® therapy. Usage may interfere with some clinical laboratory evaluations. Also, because of its deep red color, hydroxocobalamin may cause hemodialysis machines to shut down due to an erroneous detection of a “blood leak. ” This should be considered before hemodialysis is initiated in patients treated with hydroxocobalamin. Due to potential photosensitivity, patients should avoid direct sun until erythema resolves. CYANOKIT® is Pregnancy Category C and should be used during pregnancy only if the potential benefit justifies the potential risk. Safety and effectiveness of CYANOKIT® have not been established in pediatric patients. The most common adverse reactions (>5%) are transient and include chromaturia, erythema, rash (predominantly acneiform), increased blood pressure, nausea, headache, decreased lymphocyte percentage, and injection site reactions. Please see full Prescribing Information for CYANOKIT® is a registered trademark of Merck Santé s. a. s. , licensed by Meridian Medical Technologies™, Inc. , a Pfizer company. Copyright © 2011 Meridian Medical Technologies™, Inc. , a Pfizer company. All rights reserved. CYK 404616 -01 10/2011 33

Prescribing Information for CYANOKIT® 5 g (hydroxocobalamin for injection) ►Click for Table of Contents






- Slides: 40