FUEL CELLS Viktria Barbara KOVCS kovacsenergia bme hu

FUEL CELLS Viktória Barbara KOVÁCS kovacs@energia. bme. hu This semester not available: Build. D room 207 B 06 1 463 2592 Viktória B. Kovács| Fuel cells| © 2015 BMEGEENAG 51 | D 218 | 2015 -16 -1 |

QUESTIONS What is the working principle of fuel cells (PEMFC)? What are the advantages and disadvantages of fuel cells? Describe fuel cell system for power generation! What type of fuel cells do you know? Describe one of them in detail: type of electrolyte, fuel, operating temperature, efficiency! Viktória B. Kovács| Fuel cells| © 2015 BMEGEENAG 51 | D 218 | 2015 -16 -1 | 2
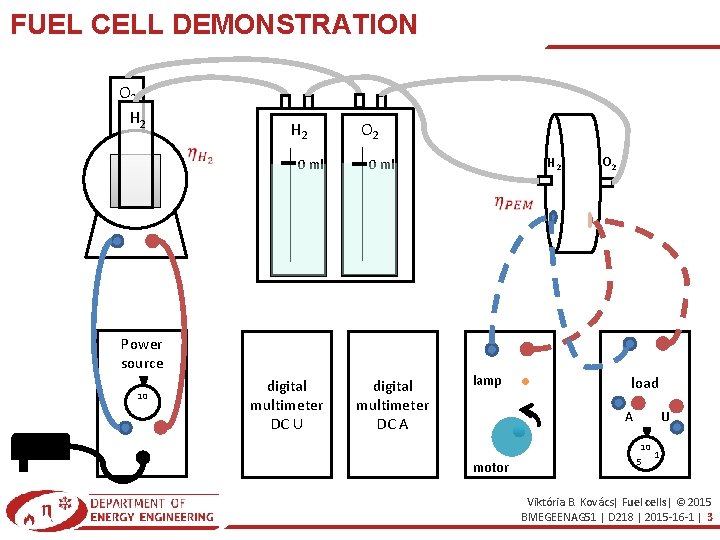
FUEL CELL DEMONSTRATION O 2 H 2 0 ml O 2 Power source 10 digital multimeter DC U H 2 0 ml digital multimeter DC A lamp O 2 load U A 10 motor 5 1 Viktória B. Kovács| Fuel cells| © 2015 BMEGEENAG 51 | D 218 | 2015 -16 -1 | 3
![MEASURED PROPERTIES H 2 [ml] time[s] I in [A] U in [V] I out MEASURED PROPERTIES H 2 [ml] time[s] I in [A] U in [V] I out](http://slidetodoc.com/presentation_image_h2/75874ebfc1d750694f52903ab4c6eb40/image-4.jpg)
MEASURED PROPERTIES H 2 [ml] time[s] I in [A] U in [V] I out [A] U out [V] R in [Ω] R out [Ω] P in [W] P out [W] ηH 2 [%] ηPEM [%] ηΣ [%] 1 Viktória B. Kovács| Fuel cells| © 2015 BMEGEENAG 51 | D 218 | 2015 -16 -1 | 4

INTRODUCTION Fuel cell is a device which use hydrogen or hydrogen rich fuel and oxygen to produce electricity through electrochemical process. Byproducts: water and heat Present use: vehicles, energy supply of buildings, PC. Viktória B. Kovács| Fuel cells| © 2015 BMEGEENAG 51 | D 218 | 2015 -16 -1 | 5

WHY FUEL CELLS? Decrease pollution Decrease fossil energy source dependence Slowing down global warming Prevent energy crisis Viktória B. Kovács| Fuel cells| © 2015 BMEGEENAG 51 | D 218 | 2015 -16 -1 | 6

FUEL CELLS Advantages High and size independent efficiency (40. . 70%). Heat (byproduct) can be used in cogeneration. Low specific mass: 1 kg/k. W. No moving parts → long lifetime, silent, reliable. Very low GHG emission. No toxic /harmful pollutants for health or for the environment In case of pure hydrogen operation only water and heat emission. Disadvantages New technology → averseness in the beginning. High costs in the beginning of market introduction → high risk Missing or undeveloped hydrogen infrastructure. Viktória B. Kovács| Fuel cells| © 2015 BMEGEENAG 51 | D 218 | 2015 -16 -1 | 7

PRINCIPLE OF OPERATION The fuel cell produces electric power from hydrogen and oxygen through electrochemical process. An ordinary fuel cell consists of two fine and porous electrode (anode and cathode) and the electrolyte between the electrodes. cathode anode Fuel Cells part 1 _ University Of Surrey. mp 4 https: //www. youtube. com/watch? v=p. Ot. F-Wv. Rp. OM Fuel Cells part 2 _ University Of Surrey. mp 4 https: //www. youtube. com/watch? v=qng 8 NZ 7 iw. N 8 The hydrogen or hydrogen rich fuel dissociates to e- and p+ on the anode by the help of the catalyst. The oxygen with the electrons and protons (or other ions) form water (or something else) on the cathode. The electrons are not allowed to penetrate through the membrane, they are forced to the cathode through the current circuit. The drift of the electrons produces electrical direct current. Viktória B. Kovács| Fuel cells| © 2015 BMEGEENAG 51 | D 218 | 2015 -16 -1 | 8
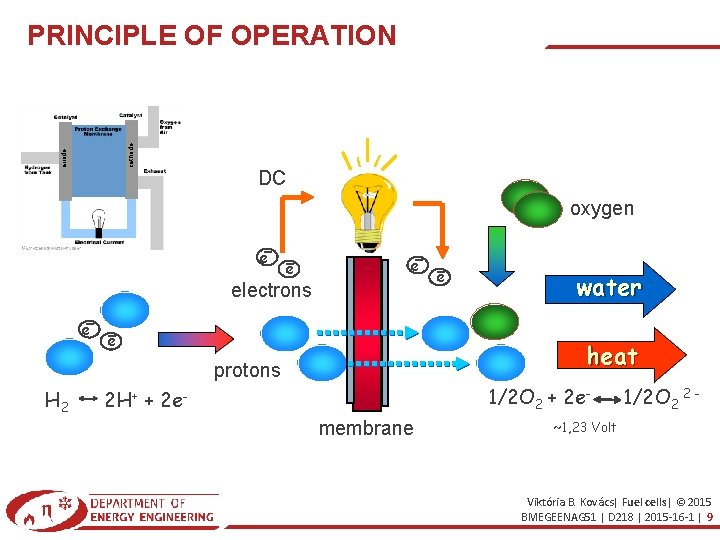
anode cathode PRINCIPLE OF OPERATION DC oxygen e electrons e e water heat protons H 2 e 1/2 O 2 + 2 e- 2 H+ + 2 emembrane 1/2 O 2 2 - ~1, 23 Volt Viktória B. Kovács| Fuel cells| © 2015 BMEGEENAG 51 | D 218 | 2015 -16 -1 | 9
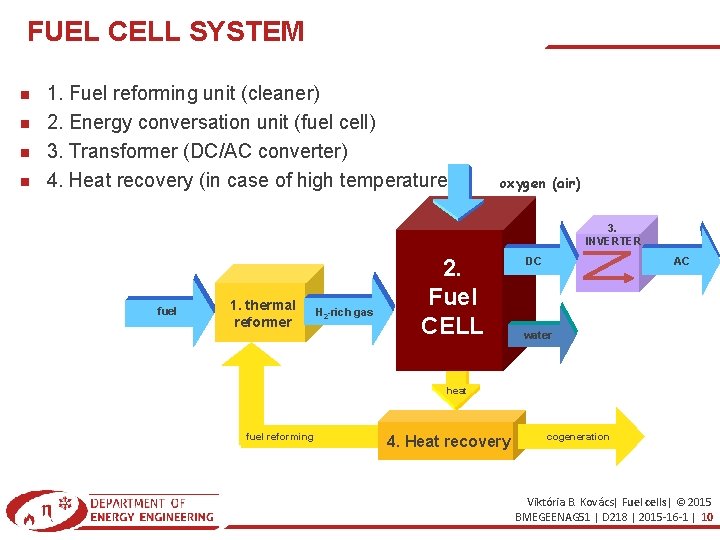
FUEL CELL SYSTEM 1. Fuel reforming unit (cleaner) 2. Energy conversation unit (fuel cell) 3. Transformer (DC/AC converter) 4. Heat recovery (in case of high temperature) oxygen (air) 3. INVERTER fuel 1. thermal reformer H 2 -rich gas 2. Fuel CELL AC DC water heat fuel reforming 4. Heat recovery cogeneration Viktória B. Kovács| Fuel cells| © 2015 BMEGEENAG 51 | D 218 | 2015 -16 -1 | 10

1. FUEL REFORMING UNIT Fuel reforming and cleaning. If the fuel is hydrogen only cleaning is needed. Liquid fuel (methanol, gasoline. . . ) are changed by thermal reforming to gaseous hydrocarbons. 2. ENERGY CONVERSATION UNIT (FUEL CELL) Chemical energy → electrical power. DC is produced through chemical reaction. Viktória B. Kovács| Fuel cells| © 2015 BMEGEENAG 51 | D 218 | 2015 -16 -1 | 11

3. TRANSFORMER AND REGULATOR Keep regulated and constant electrical connection between the fuel cell and network (consumer). Transform DC to AC. Regulates the current intensity, potential, frequency, and other parameters according to the demands. 4. HEAT RECOVERY UNIT Not always added, because heat is not the main product. In case of high temperature: – steam production for combined power generation – direct use in steam turbine. The overall efficiency is higher with heat recovery. Viktória B. Kovács| Fuel cells| © 2015 BMEGEENAG 51 | D 218 | 2015 -16 -1 | 12

CLASSIFICATION OF FUEL CELLS By Electrolyte polymer electrolyte membrane (PEMFC) ~80 °C phosphoric acid (PAFC) ~200 °C alkaline (AFC) 80. . 100 °C molten carbonate (MCFC) ~650 °C solid oxide (SOFC) By Fuel Direct: hydrogen to anode Indirect: Hydrogen rich fuel reformatted to anode Regenerative: electricity is used to produce H 2 and O 2 – tubular (TSOFC) 800 °C – intermediate temperature (ITSOFC) 1000 °C Viktória B. Kovács| Fuel cells| © 2015 BMEGEENAG 51 | D 218 | 2015 -16 -1 | 13

POLYMER ELECTROLYTE MEMBRANEPEMFC PEM Fuel Cell_ How it works. mp 4 https: //www. youtube. com/watch? v=_Ms. G 9 REFN 3 s Other name: SPEFC (Solid Polymer Electrolyte Fuel Cells) Electrolyte: proton exchange membrane Low temperature (85. . 105 °C) Nafion® membrane (Du. Pont development) which is bed in poli-tetra-fluorethilen (PTFE, Teflon) based composition Anode: H 2 → 2 H+ + 2 e. Cathode: 1/2 O 2+ 2 H+ + 2 e- → H 2 O High power density (Power/mass) Fast stand-up Mainly in vehicles disadvantage: low CO tolerance (Pt poison) Viktória B. Kovács| Fuel cells| © 2015 BMEGEENAG 51 | D 218 | 2015 -16 -1 | 14
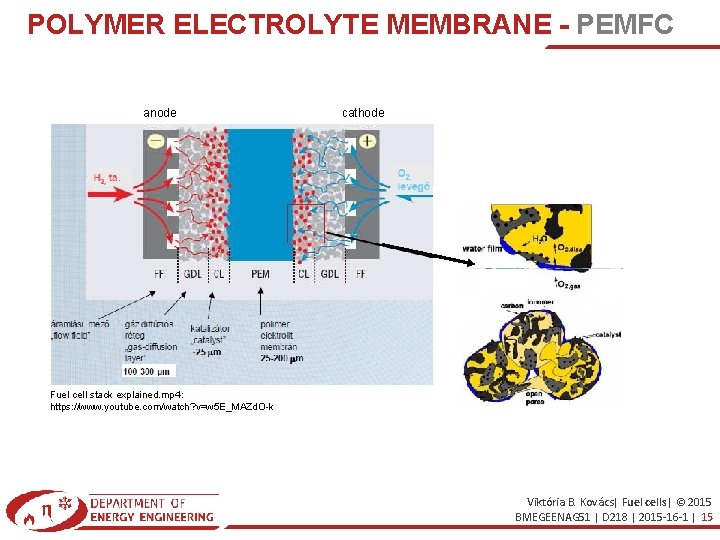
POLYMER ELECTROLYTE MEMBRANE - PEMFC anode cathode Fuel cell stack explained. mp 4: https: //www. youtube. com/watch? v=w 5 E_MAZd. O-k Viktória B. Kovács| Fuel cells| © 2015 BMEGEENAG 51 | D 218 | 2015 -16 -1 | 15

PHOSPHORIC ACID ELECTROLYTE - PAFC 100% concentration of H 3 PO 4 in Si. C matrix with Pt catalyst Anode: H 2 → 2 H+ + 2 e. Cathode: 1/2 O 2+ 2 H+ + 2 e- → H 2 O High temperature is needed, because H 3 PO 4 is bad conductor CO<3. . 5 vol% or Pt is poisoned Viktória B. Kovács| Fuel cells| © 2015 BMEGEENAG 51 | D 218 | 2015 -16 -1 | 16

ALKALINE ELECTROLYTE - AFC high concentration KOH (35. . 85 m%) in asbestos matrix Anode: H 2 + 2 OH- → 2 H 2 O + e. Cathode: 1/2 O 2 + H 2 O + 2 e- → 2 OHCO 2 is poison: CO 2 + KOH → K 2 CO 3 because the electrolyte changes High efficiency (~60%) Disadvantage: expensive Viktória B. Kovács| Fuel cells| © 2015 BMEGEENAG 51 | D 218 | 2015 -16 -1 | 17
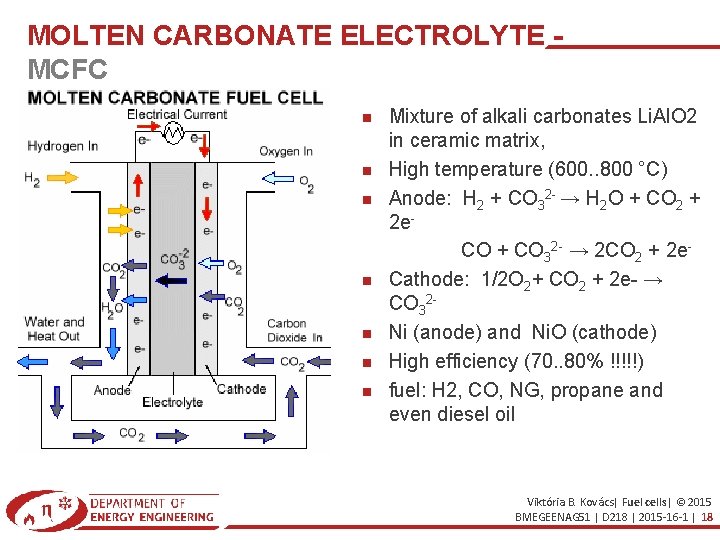
MOLTEN CARBONATE ELECTROLYTE MCFC Mixture of alkali carbonates Li. Al. O 2 in ceramic matrix, High temperature (600. . 800 °C) Anode: H 2 + CO 32 - → H 2 O + CO 2 + 2 e. CO + CO 32 - → 2 CO 2 + 2 e. Cathode: 1/2 O 2+ CO 2 + 2 e- → CO 32 Ni (anode) and Ni. O (cathode) High efficiency (70. . 80% !!!!!) fuel: H 2, CO, NG, propane and even diesel oil Viktória B. Kovács| Fuel cells| © 2015 BMEGEENAG 51 | D 218 | 2015 -16 -1 | 18
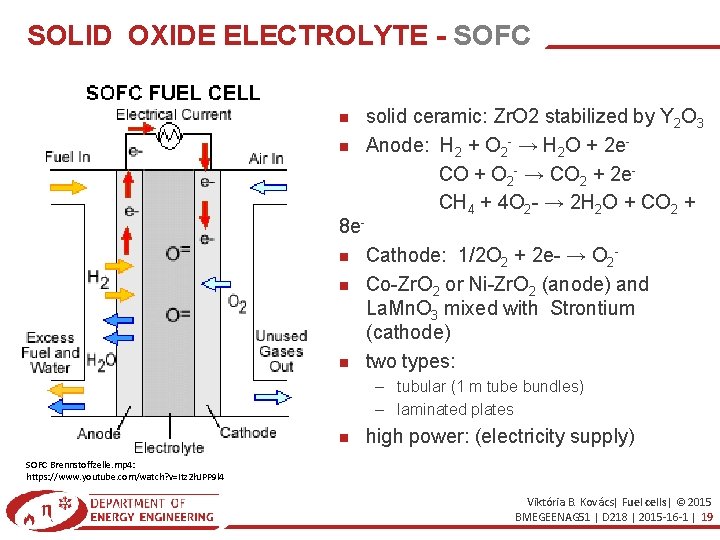
SOLID OXIDE ELECTROLYTE - SOFC 8 e solid ceramic: Zr. O 2 stabilized by Y 2 O 3 Anode: H 2 + O 2 - → H 2 O + 2 e. CO + O 2 - → CO 2 + 2 e. CH 4 + 4 O 2 - → 2 H 2 O + CO 2 + Cathode: 1/2 O 2 + 2 e- → O 2 Co-Zr. O 2 or Ni-Zr. O 2 (anode) and La. Mn. O 3 mixed with Strontium (cathode) two types: – tubular (1 m tube bundles) – laminated plates high power: (electricity supply) SOFC Brennstoffzelle. mp 4: https: //www. youtube. com/watch? v=Itz 2 h. JPP 9 l 4 Viktória B. Kovács| Fuel cells| © 2015 BMEGEENAG 51 | D 218 | 2015 -16 -1 | 19
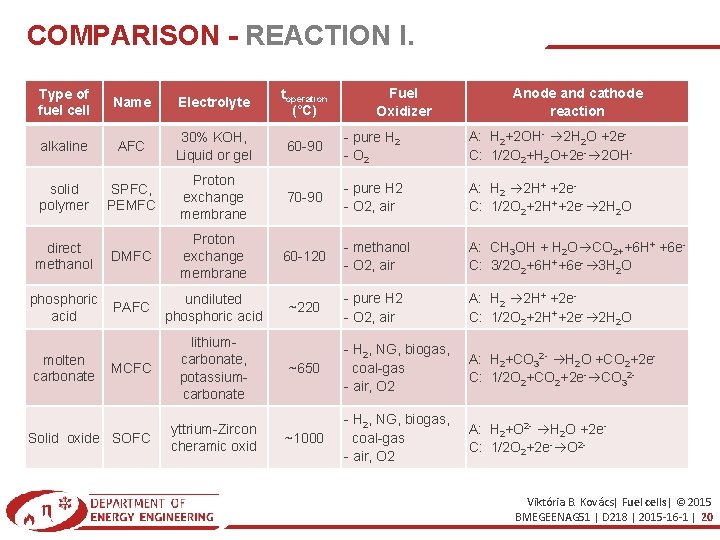
COMPARISON - REACTION I. Fuel Oxidizer Anode and cathode reaction Type of fuel cell Name Electrolyte toperation (°C) alkaline AFC 30% KOH, Liquid or gel 60 -90 - pure H 2 - O 2 A: H 2+2 OH- 2 H 2 O +2 e. C: 1/2 O 2+H 2 O+2 e- 2 OH- solid polymer SPFC, PEMFC Proton exchange membrane 70 -90 - pure H 2 - O 2, air A: H 2 2 H+ +2 e. C: 1/2 O 2+2 H++2 e- 2 H 2 O DMFC Proton exchange membrane 60 -120 - methanol - O 2, air A: CH 3 OH + H 2 O CO 2++6 H+ +6 e. C: 3/2 O 2+6 H++6 e- 3 H 2 O PAFC undiluted phosphoric acid ~220 - pure H 2 - O 2, air A: H 2 2 H+ +2 e. C: 1/2 O 2+2 H++2 e- 2 H 2 O MCFC lithiumcarbonate, potassiumcarbonate ~650 - H 2, NG, biogas, coal-gas - air, O 2 A: H 2+CO 32 - H 2 O +CO 2+2 e. C: 1/2 O 2+CO 2+2 e- CO 32 - ~1000 - H 2, NG, biogas, coal-gas - air, O 2 A: H 2+O 2 - H 2 O +2 e. C: 1/2 O 2+2 e- O 2 - direct methanol phosphoric acid molten carbonate Solid oxide SOFC yttrium-Zircon cheramic oxid Viktória B. Kovács| Fuel cells| © 2015 BMEGEENAG 51 | D 218 | 2015 -16 -1 | 20

low temperatire high temperature COMPARISON - REACTION II. Not used fuel not used O 2 / air solid oxide SOFC H 2/CO O 2 - O 2 >800°C molten carbonate MCFC H 2/CO CO 32 - O 2 650°C phosphoric acid PAFC H 2 H+ O 2 200°C direct methano l polymer electrolyte DMFC CH 3 OH H+ O 2 60 – 120°C PEMFC H 2 H+ O 2 < 90°C AFC H 2 OH- O 2 < 80°C alkaline fuel O 2 / air Viktória B. Kovács| Fuel cells| © 2015 BMEGEENAG 51 | D 218 | 2015 -16 -1 | 21
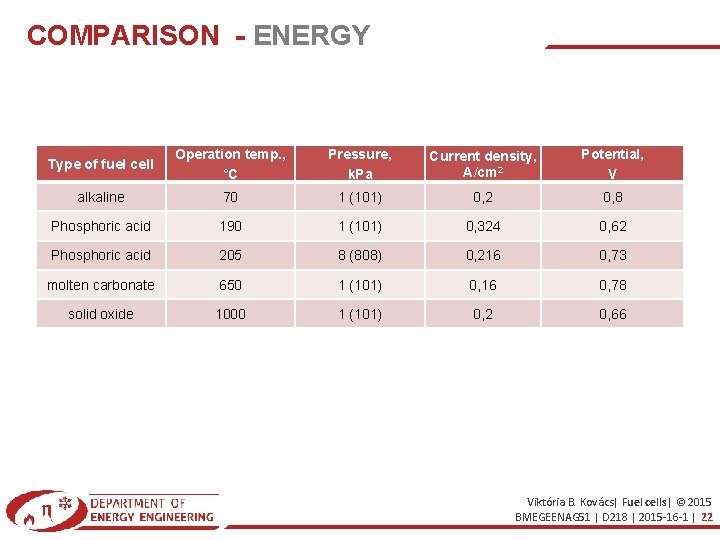
COMPARISON - ENERGY Type of fuel cell Operation temp. , °C Pressure, k. Pa Current density, A/cm 2 Potential, V alkaline 70 1 (101) 0, 2 0, 8 Phosphoric acid 190 1 (101) 0, 324 0, 62 Phosphoric acid 205 8 (808) 0, 216 0, 73 molten carbonate 650 1 (101) 0, 16 0, 78 solid oxide 1000 1 (101) 0, 2 0, 66 Viktória B. Kovács| Fuel cells| © 2015 BMEGEENAG 51 | D 218 | 2015 -16 -1 | 22
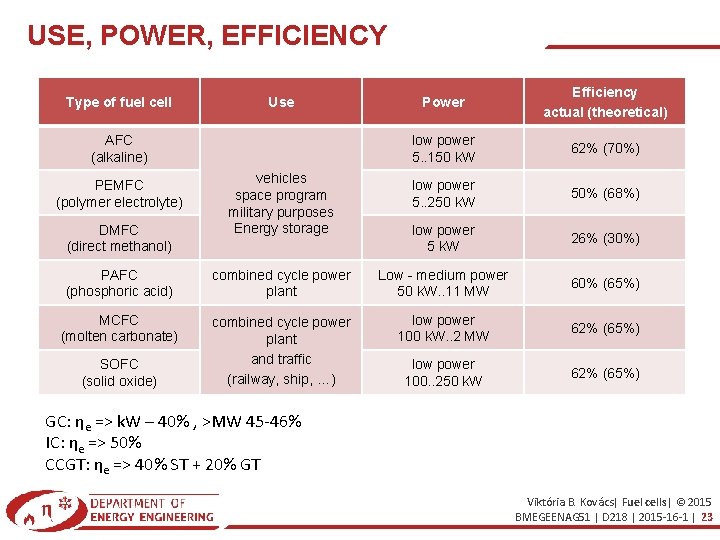
USE, POWER, EFFICIENCY Power Efficiency actual (theoretical) low power 5. . 150 k. W 62% (70%) vehicles space program military purposes Energy storage low power 5. . 250 k. W 50% (68%) low power 5 k. W 26% (30%) PAFC (phosphoric acid) combined cycle power plant Low - medium power 50 k. W. . 11 MW 60% (65%) MCFC (molten carbonate) combined cycle power plant and traffic (railway, ship, …) low power 100 k. W. . 2 MW 62% (65%) low power 100. . 250 k. W 62% (65%) Type of fuel cell Use AFC (alkaline) PEMFC (polymer electrolyte) DMFC (direct methanol) SOFC (solid oxide) GC: ηe => k. W – 40% , >MW 45 -46% IC: ηe => 50% CCGT: ηe => 40% ST + 20% GT Viktória B. Kovács| Fuel cells| © 2015 BMEGEENAG 51 | D 218 | 2015 -16 -1 | 23
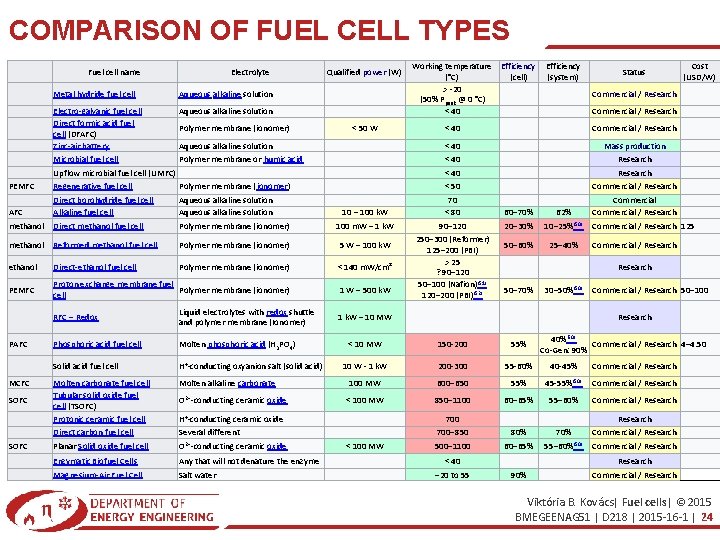
COMPARISON OF FUEL CELL TYPES Fuel cell name Electrolyte Metal hydride fuel cell Aqueous alkaline solution Electro-galvanic fuel cell Direct formic acid fuel cell (DFAFC) Zinc-air battery Microbial fuel cell Aqueous alkaline solution Polymer membrane (ionomer) Qualified power (W) < 50 W Aqueous alkaline solution Polymer membrane or humic acid Upflow microbial fuel cell (UMFC) Working temperature Efficiency (°C) (cell) > -20 (50% Ppeak @ 0 °C) < 40 Mass production Research < 40 Research Commercial / Research Polymer membrane (ionomer) < 50 AFC methanol Aqueous alkaline solution Polymer membrane (ionomer) 10 – 100 k. W 100 m. W – 1 k. W methanol Reformed methanol fuel cell Polymer membrane (ionomer) 5 W – 100 k. W ethanol Direct-ethanol fuel cell Polymer membrane (ionomer) < 140 m. W/cm² PEMFC Proton exchange membrane fuel Polymer membrane (ionomer) cell 1 W – 500 k. W 70 < 80 90– 120 250– 300 (Reformer) 125– 200 (PBI) > 25 ? 90– 120 50– 100 (Nafion)[51] 120– 200 (PBI)[52] PAFC MCFC SOFC Phosphoric acid fuel cell Molten phosphoric acid (H 3 PO 4) Solid acid fuel cell H+-conducting oxyanion salt (solid acid) Molten carbonate fuel cell Tubular solid oxide fuel cell (TSOFC) Protonic ceramic fuel cell Direct carbon fuel cell Planar Solid oxide fuel cell Molten alkaline carbonate Enzymatic Biofuel Cells Magnesium-Air Fuel Cell Any that will not denature the enzyme Salt water O 2−-conducting ceramic oxide H+-conducting ceramic oxide Several different O 2−-conducting ceramic oxide Commercial / Research < 40 Direct borohydride fuel cell Alkaline fuel cell Direct methanol fuel cell Cost (USD/W) Commercial / Research Regenerative fuel cell Liquid electrolytes with redox shuttle and polymer membrane (Ionomer) Status < 40 PEMFC RFC – Redox Efficiency (system) 60– 70% 20– 30% 62% 10– 25%[50] 50– 60% 25– 40% Commercial / Research 125 Commercial / Research 50– 70% 30– 50%[50] 1 k. W – 10 MW Commercial / Research 50– 100 Research 40%[50] Commercial / Research 4– 4. 50 Co-Gen: 90% < 10 MW 150 -200 55% 10 W - 1 k. W 200 -300 55 -60% 40 -45% Commercial / Research 100 MW 600– 650 55% 45 -55%[50] < 100 MW 850– 1100 60– 65% 55– 60% Commercial / Research < 100 MW 700– 850 500– 1100 80% 60– 65% 70% 55– 60%[50] Research Commercial / Research < 40 − 20 to 55 90% Research Commercial / Research Viktória B. Kovács| Fuel cells| © 2015 BMEGEENAG 51 | D 218 | 2015 -16 -1 | 24
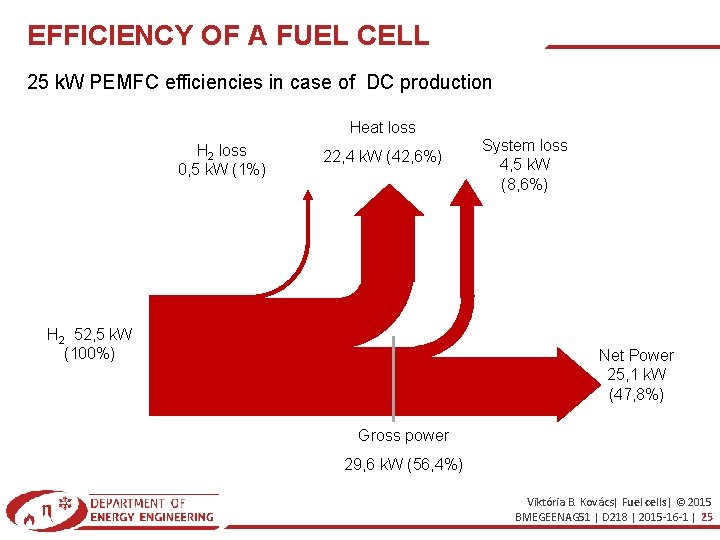
EFFICIENCY OF A FUEL CELL 25 k. W PEMFC efficiencies in case of DC production Heat loss H 2 loss 0, 5 k. W (1%) 22, 4 k. W (42, 6%) H 2 52, 5 k. W (100%) System loss 4, 5 k. W (8, 6%) Net Power 25, 1 k. W (47, 8%) Gross power 29, 6 k. W (56, 4%) Viktória B. Kovács| Fuel cells| © 2015 BMEGEENAG 51 | D 218 | 2015 -16 -1 | 25
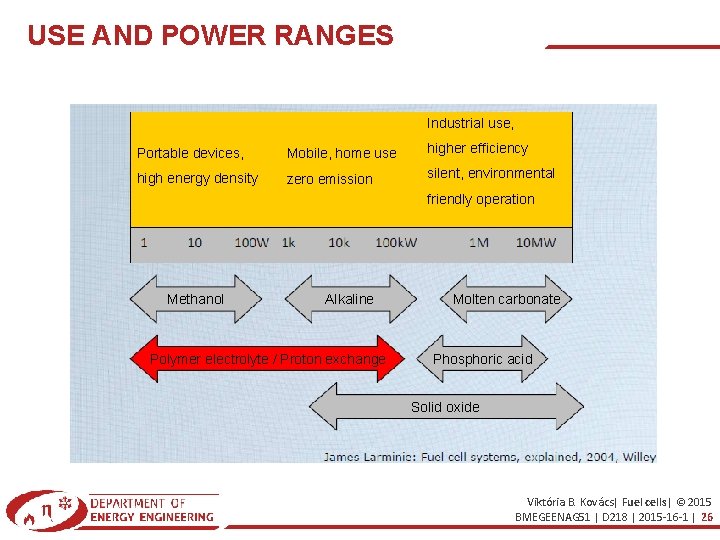
USE AND POWER RANGES Industrial use, Portable devices, Mobile, home use higher efficiency high energy density zero emission silent, environmental friendly operation Methanol Alkaline Polymer electrolyte / Proton exchange Molten carbonate Phosphoric acid Solid oxide Viktória B. Kovács| Fuel cells| © 2015 BMEGEENAG 51 | D 218 | 2015 -16 -1 | 26

USE Road transport Energy storage Small scale power plants Analytics Viktória B. Kovács| Fuel cells| © 2015 BMEGEENAG 51 | D 218 | 2015 -16 -1 | 27

PEMFC IN VEHICLES https: //en. wikipedia. org/wiki/List_of_fuel_cell_vehicles Viktória B. Kovács| Fuel cells| © 2015 BMEGEENAG 51 | D 218 | 2015 -16 -1 | 28

USE OF HYDROGEN reducing agent ammonia production (Haber-Bosch) hydrogenation of vegetable oils production of synthetic gasoline welding HCl production metal hydrides, complex metal hydrides prduction fuel Viktória B. Kovács| Fuel cells| © 2015 BMEGEENAG 51 | D 218 | 2015 -16 -1 | 29
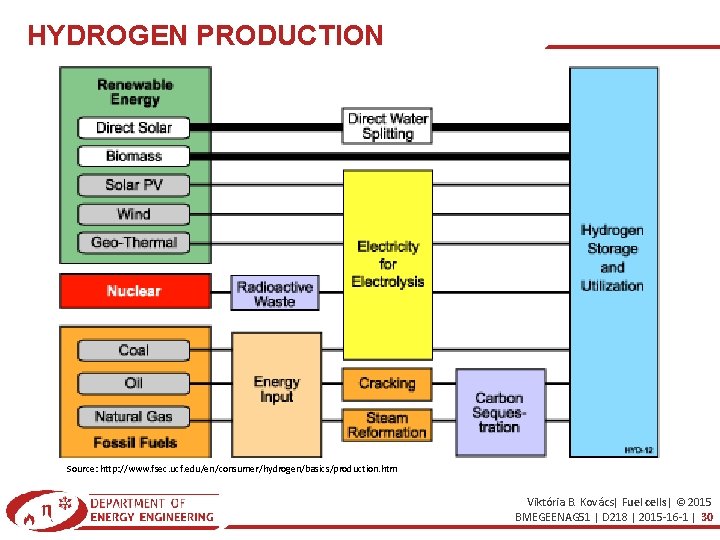
HYDROGEN PRODUCTION Source: http: //www. fsec. ucf. edu/en/consumer/hydrogen/basics/production. htm Viktória B. Kovács| Fuel cells| © 2015 BMEGEENAG 51 | D 218 | 2015 -16 -1 | 30
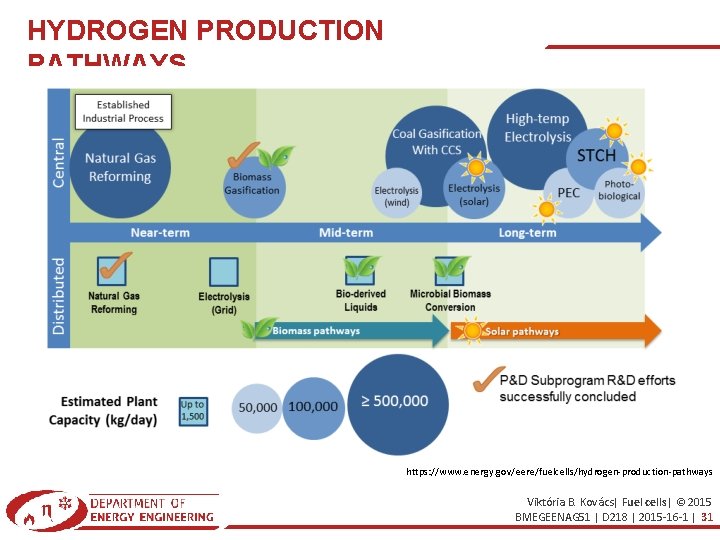
HYDROGEN PRODUCTION PATHWAYS https: //www. energy. gov/eere/fuelcells/hydrogen-production-pathways Viktória B. Kovács| Fuel cells| © 2015 BMEGEENAG 51 | D 218 | 2015 -16 -1 | 31

HYDROGEN PRIDUCTION PROCESSES Thermochemical Processes Some thermal processes use the energy in various resources, such as natural gas, coal, or biomass, to release hydrogen from their molecular structure. In other processes, heat, in combination with closed-chemical cycles, produces hydrogen from feedstocks such as water. Learn more about the following thermochemical processes: – Natural gas reforming (also called steam methane reforming or SMR) > 95% USA – Coal gasification – Biomass-derived liquid reforming – Solar thermochemical hydrogen (STCH). Electrolytic Processes Electrolyzers use electricity to split water into hydrogen and oxygen. This technology is well developed and available commercially, and systems that can efficiently use intermittent renewable power are being developed. Learn more about electrolysis. https: //www. energy. gov/eere/fuelcells/hydrogen-production-processes Viktória B. Kovács| Fuel cells| © 2015 BMEGEENAG 51 | D 218 | 2015 -16 -1 | 32

HYDROGEN PRIDUCTION PROCESSES Direct Solar Water Splitting Processes Direct solar water splitting, or photolytic processes use light energy to split water into hydrogen and oxygen. These processes are currently in the very early stages of research but offer long-term potential for sustainable hydrogen production with low environmental impact. Learn more about the following solar water splitting processes: – Photoelectrochemical (PEC) – Photobiological. Biological Processes Microbes such as bacteria and microalgae can produce hydrogen through biological reactions, using sunlight or organic matter. These technology pathways are at an early stage of research, but in the long term have the potential for sustainable, low-carbon hydrogen production. Learn more about the following biological processes: – Microbial biomass conversion – Photobiological. https: //www. energy. gov/eere/fuelcells/hydrogen-production-processes Viktória B. Kovács| Fuel cells| © 2015 BMEGEENAG 51 | D 218 | 2015 -16 -1 | 33

EFFICIENCIES Electrolysis: – Power plant efficiency: 45% – Transformation: 80% Σ production: 29% – Electrolysis: 80% + Fuel cell: 70% ~ 12% ↔ ICE 25% + vehicle mechanical efficiency: 80% Viktória B. Kovács| Fuel cells| © 2015 BMEGEENAG 51 | D 218 | 2015 -16 -1 | 34
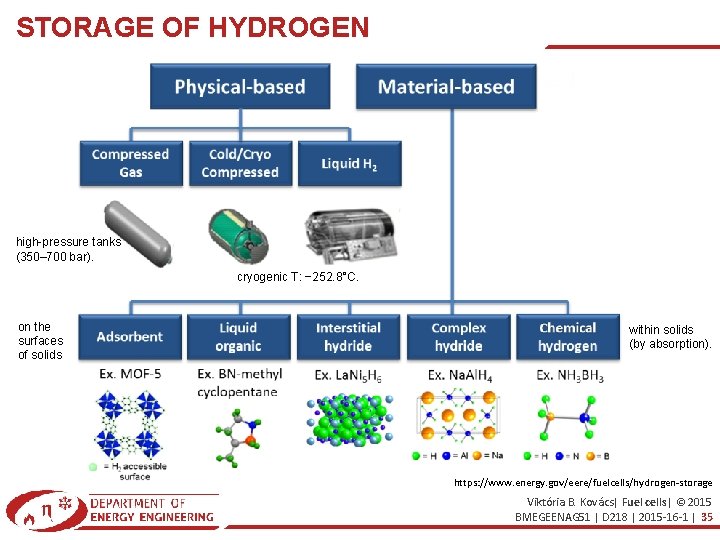
STORAGE OF HYDROGEN high-pressure tanks (350– 700 bar). cryogenic T: − 252. 8°C. on the surfaces of solids within solids (by absorption). https: //www. energy. gov/eere/fuelcells/hydrogen-storage Viktória B. Kovács| Fuel cells| © 2015 BMEGEENAG 51 | D 218 | 2015 -16 -1 | 35
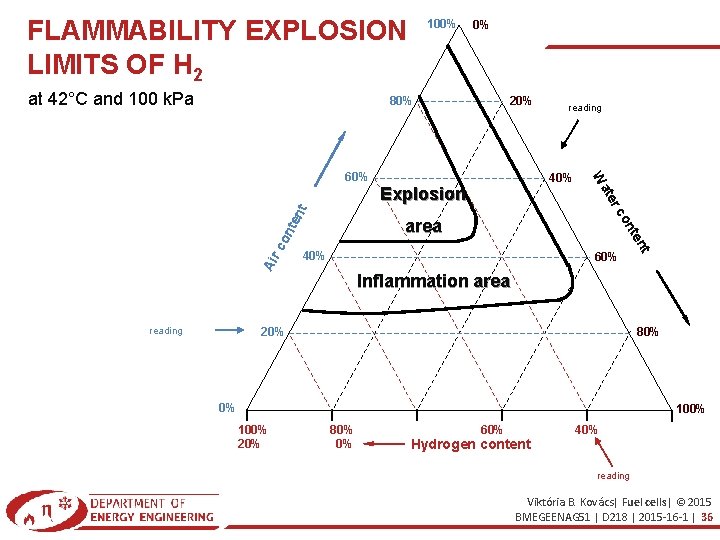
FLAMMABILITY EXPLOSION LIMITS OF H 2 at 42°C and 100 k. Pa 100% 0% 80% 20% 42°C és 100 k. Pa mellett 40% nt co 40% 60% t ten nte area on Air rc ate Explosion W 60% reading Inflammation area 20% reading 80% 0% 100% 20% 80% 0% 60% Hydrogen content 40% reading Viktória B. Kovács| Fuel cells| © 2015 BMEGEENAG 51 | D 218 | 2015 -16 -1 | 36

HYDROGEN SAFETY Security management for hydrogen (legislation, risk assessments, practical regulations) https: //www. energy. gov/eere/fuelcells/safety-codes-and-standards Hydrogen Technology Regulations (USA, EU): – ATEX 95 (94/9 / EC) product specif. : „ Equipment Directive” ATEX directive 2014/34/EU (April 2016) – ATEX 137 (1999/92 / EC) user specification: „ Workplace Directive” – ASME, CGA, NFPA, SAE, IFC, . . . Safe fuelling: – ISO/TS 19880 -1: 2016 Gaseous hydrogen -- Fuelling stations -- Part 1: General requirements – Society of Automotive Engineers: SAE J 2601 Fueling Protocols for Light Duty Gaseous Hydrogen Surface Vehicles https: //h 2 tools. org/lessons – long maintained a user-populated database of hydrogen incidents s Viktória B. Kovács| Fuel cells| © 2015 BMEGEENAG 51 | D 218 | 2015 -16 -1 | 37
HYDROGEN SAFETY Safety distance Ensure ventilation Electric and Hydrogen systems Installing sensors Viktória B. Kovács| Fuel cells| © 2015 BMEGEENAG 51 | D 218 | 2015 -16 -1 | 38

HYDROGEN STATIONS IN GERMANY Viktória B. Kovács| Fuel cells| © 2015 BMEGEENAG 51 | D 218 | 2015 -16 -1 | 39
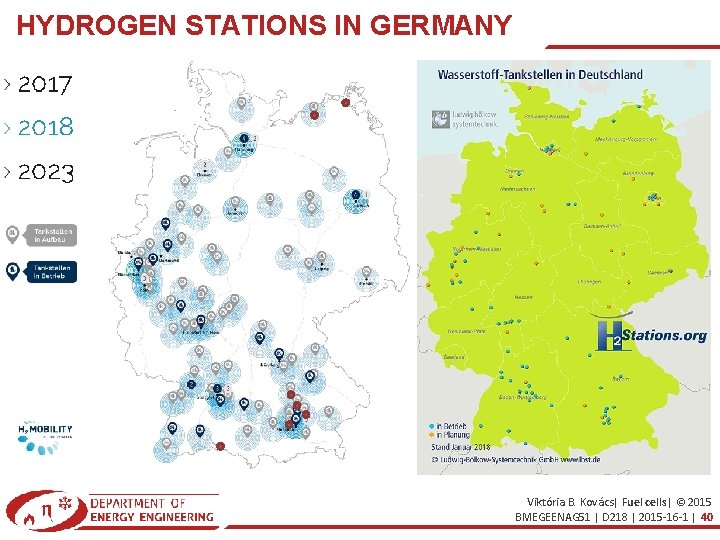
HYDROGEN STATIONS IN GERMANY Viktória B. Kovács| Fuel cells| © 2015 BMEGEENAG 51 | D 218 | 2015 -16 -1 | 40
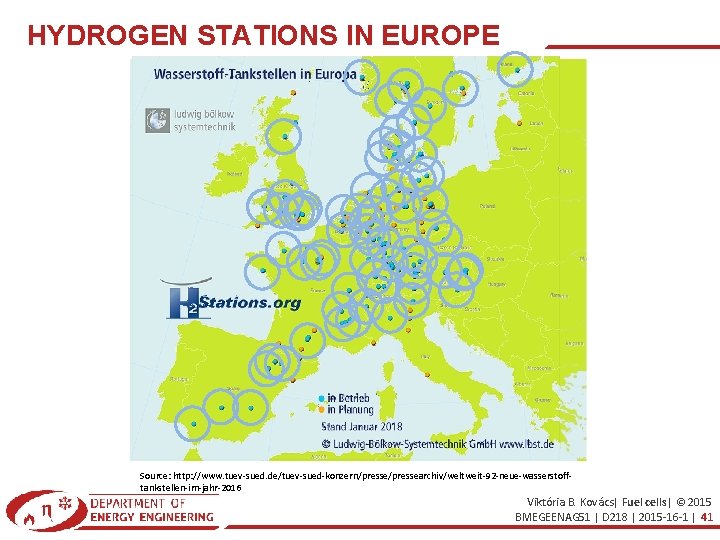
HYDROGEN STATIONS IN EUROPE Source: http: //www. tuev-sued. de/tuev-sued-konzern/pressearchiv/weltweit-92 -neue-wasserstofftankstellen-im-jahr-2016 Viktória B. Kovács| Fuel cells| © 2015 BMEGEENAG 51 | D 218 | 2015 -16 -1 | 41
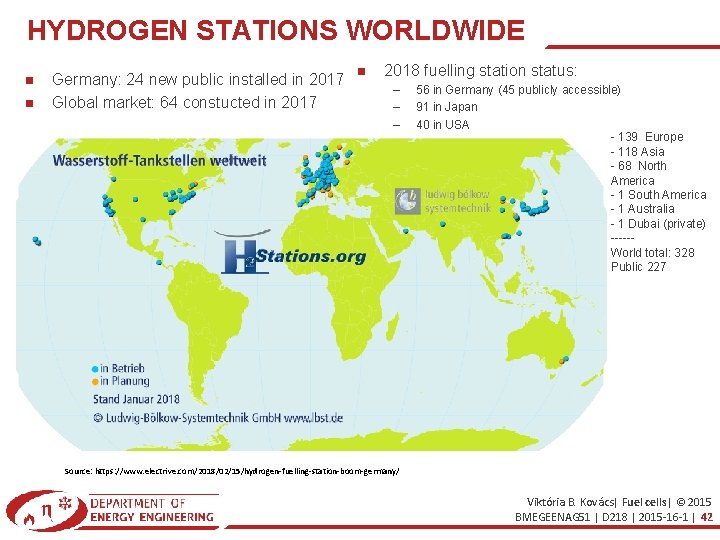
HYDROGEN STATIONS WORLDWIDE Germany: 24 new public installed in 2017 Global market: 64 constucted in 2017 2018 fuelling station status: – – – 56 in Germany (45 publicly accessible) 91 in Japan 40 in USA - 139 Europe - 118 Asia - 68 North America - 1 South America - 1 Australia - 1 Dubai (private) -----World total: 328 Public 227 Source: https: //www. electrive. com/2018/02/15/hydrogen-fuelling-station-boom-germany/ Viktória B. Kovács| Fuel cells| © 2015 BMEGEENAG 51 | D 218 | 2015 -16 -1 | 42
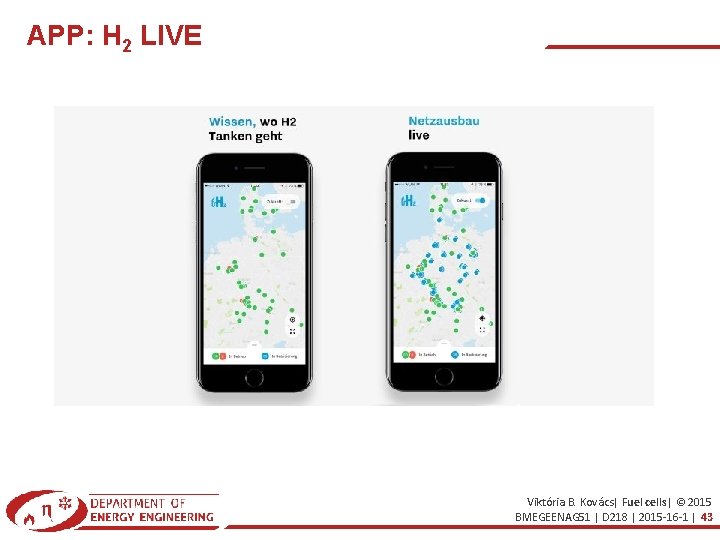
APP: H 2 LIVE Viktória B. Kovács| Fuel cells| © 2015 BMEGEENAG 51 | D 218 | 2015 -16 -1 | 43

REFERENCES Stróbl Alajos: Hidrogén az energiagazdálkodásban, ENERGOexpo Útkeresés, Debrecen, 2008. szeptember 24. Dőry Zsófia: Hidrogén – a jövő energiahordozója, Energo Expo, Debrecen 2008. http: //www. foek. hu/korkep/enhat/uzemanyagcella. html Kriston Ákos, Inzelt György: Protoncserélő membrános hidrogén – levegő tüzelőanyag-cellák működési elve, szabályozása és alkalmazása, MET Hidrogén Tagozat, Tüzelőanyag-cella workshop, 2009. Március 3. presentation: A hidrogén presentation: Tüzelőanyag cellák és hidrogén technológia, Viktória B. Kovács| Fuel cells| © 2015 BMEGEENAG 51 | D 218 | 2015 -16 -1 | 44

THANK YOU FOR YOUR ATTENTION! Viktória Barbara KOVÁCS kovacs@energia. bme. hu Build. D room 207 B 06 1 463 2592 Viktória B. Kovács| Fuel cells| © 2015 BMEGEENAG 51 | D 218 | 2015 -16 -1 |
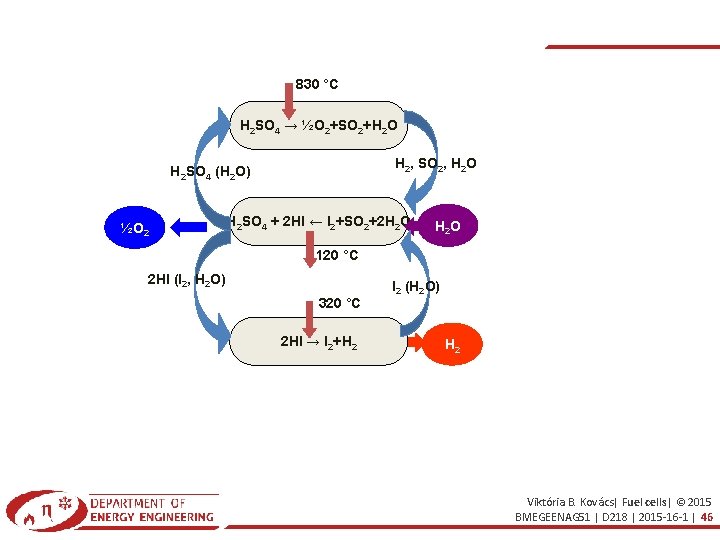
830 °C H 2 SO 4 → ½O 2+SO 2+H 2 O H 2, SO 2, H 2 O H 2 SO 4 (H 2 O) ½O 2 H 2 SO 4 + 2 HI ← I 2+SO 2+2 H 2 O 120 °C 2 HI (I 2, H 2 O) 320 °C 2 HI → I 2+H 2 I 2 (H 2 O) H 2 Viktória B. Kovács| Fuel cells| © 2015 BMEGEENAG 51 | D 218 | 2015 -16 -1 | 46
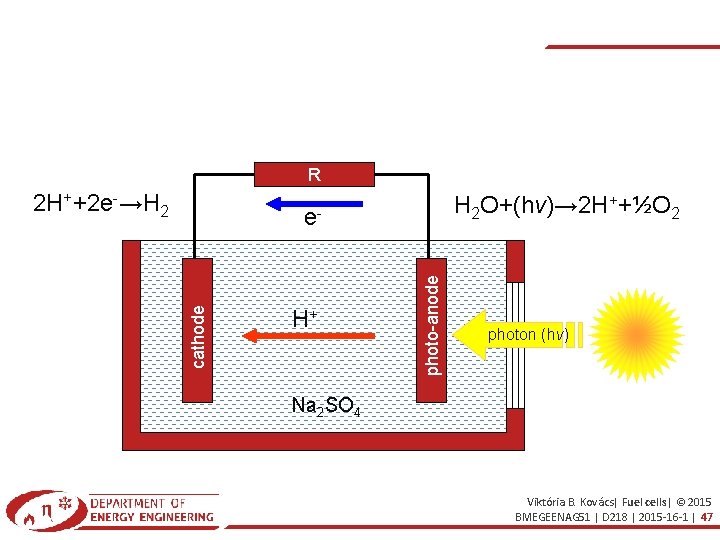
R 2 H++2 e-→H 2 H 2 O+(hν)→ 2 H++½O 2 H+ photo-anode cathode e- photon (hν) Na 2 SO 4 Viktória B. Kovács| Fuel cells| © 2015 BMEGEENAG 51 | D 218 | 2015 -16 -1 | 47
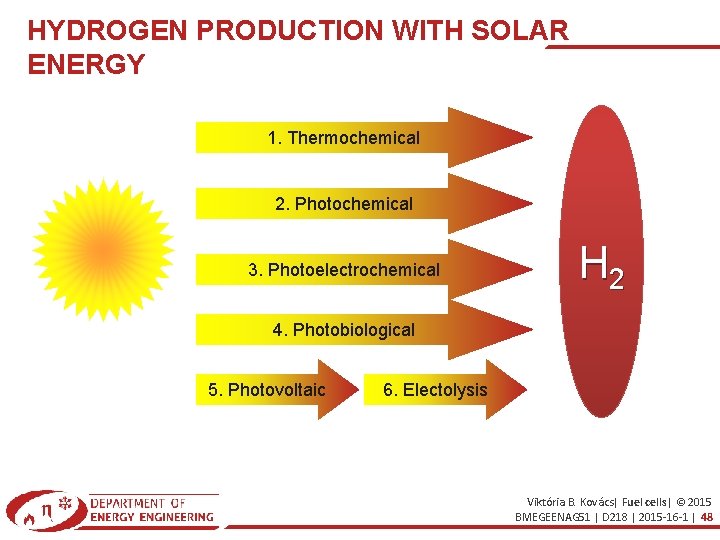
HYDROGEN PRODUCTION WITH SOLAR ENERGY 1. Thermochemical 2. Photochemical 3. Photoelectrochemical H 2 4. Photobiological 5. Photovoltaic 6. Electolysis Viktória B. Kovács| Fuel cells| © 2015 BMEGEENAG 51 | D 218 | 2015 -16 -1 | 48
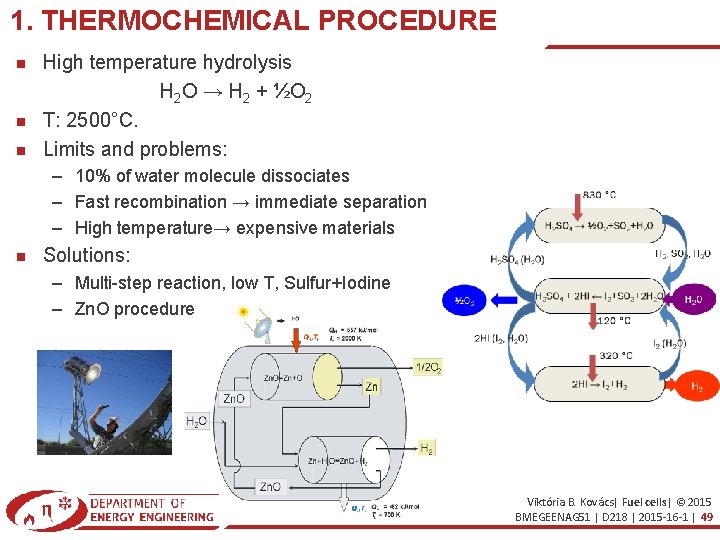
1. THERMOCHEMICAL PROCEDURE High temperature hydrolysis H 2 O → H 2 + ½O 2 T: 2500°C. Limits and problems: – 10% of water molecule dissociates – Fast recombination → immediate separation – High temperature→ expensive materials Solutions: – Multi-step reaction, low T, Sulfur+Iodine – Zn. O procedure Viktória B. Kovács| Fuel cells| © 2015 BMEGEENAG 51 | D 218 | 2015 -16 -1 | 49
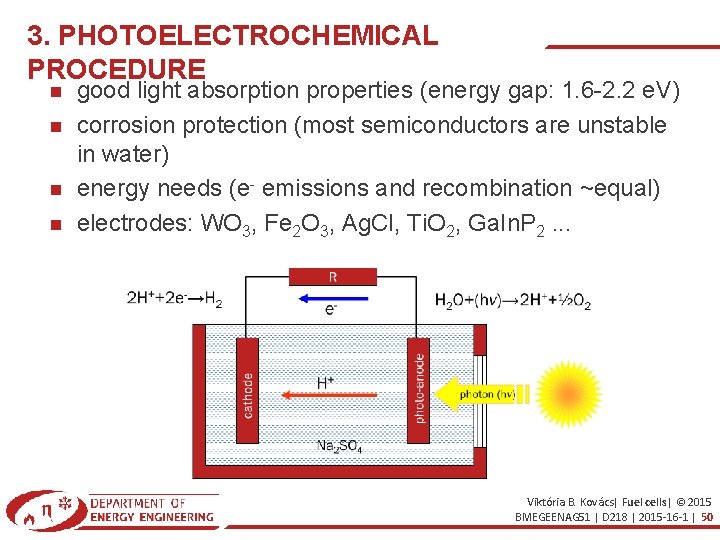
3. PHOTOELECTROCHEMICAL PROCEDURE good light absorption properties (energy gap: 1. 6 -2. 2 e. V) corrosion protection (most semiconductors are unstable in water) energy needs (e- emissions and recombination ~equal) electrodes: WO 3, Fe 2 O 3, Ag. Cl, Ti. O 2, Ga. In. P 2. . . Viktória B. Kovács| Fuel cells| © 2015 BMEGEENAG 51 | D 218 | 2015 -16 -1 | 50

STORAGE OF HYDROGEN Viktória B. Kovács| Fuel cells| © 2015 BMEGEENAG 51 | D 218 | 2015 -16 -1 | 51
- Slides: 51