FTIR Isotopic and DFT Studies of Transition MetalCarbon

FTIR Isotopic and DFT Studies of Transition Metal-Carbon Clusters Condensed in Solid Argon: Cr. C 3 S. A. Bates, C. M. L. Rittby, and W. R. M. Graham Department of Physics and Astronomy Texas Christian University Fort Worth, TX 76129 61 st Meeting of the International Symposium on Molecular Spectroscopy The Ohio State University June 19 -23, 2006

Motivation • Astrophysical – Metals observed in small molecules found in circumstellar shells and in the ISM • Cr. O in M stars (Davis, Ap. J, 1947) • Al. NC, Na. Cl in IRC+10216 (Cernicharo, A&A, 1987; Ziurys, Ap. J, 2002) • Na. CN, Mg. NC in CRL 2688 (Highberger, Ap. J, 2001) – Pure carbon chains observed in circumstellar shells (e. g. C 3, C 5 ) (Hinkle, Science, 1988; Cernicharo, Ap. J, 2000; Bernath, Science, 1989) – Silicon-bearing species observed in IRC+10216 include Si. CN and Si. C 3 (Apponi, Ap. J, 1999; Guélin, A&A, 2000) – See WH 10 on Ti. C 3 (Kinzer, Astronomical Species and Processes)

Motivation • Metallocarbohedrenes – Small metal carbon clusters important in understanding the formation of metcars (Guo, Science, 1992; Guo & Castleman, Advances in Metal and Semiconductor Clusters, 1994; Castleman, Nano Lett, 2001) – Ti. C 2, VC 2 as “building blocks” for larger metcars (Castleman, JPC, 1992; Tono, JCP, 2002) • Previous photoelectron spectroscopy (PES) and density functional theory (DFT) studies on MC 2 and MC 3 clusters (M=Sc, V, Cr, Mn, Fe, Co, and Ni) (Wang & Li, JCP, 1999; Wang & Li, JCP, 2000)

Motivation Uncertain ground state for Cr. C 3 (Zhai, JCP, 2004) DFT predictions for the lowest energy isomers of Cr. C 3ˉ and Cr. C 3 anion neutral Electronic configuration Geometry Energy (e. V) 4 B fanlike (C 2 v) 0. 0 a 4Σˉ linear +0. 05 3 B fanlike (C 2 v) 0. 0 a linear +0. 30 1 1 5Π a. Denotes nearly isoenergetic ground state energy PES spectra • Exhibit features consistent with both isomers. • Abundance of C 2 v isomer increased with hotter source conditions indicating to the authors the linear isomer may be more stable.

Research Objectives • To measure the vibrational fundamentals and isotopic shifts of metal carbon (MCn) species produced by Nd: YAG laser ablation and trapped in solid Ar at ~10 K. • To identify and determine the structures of the MCn species created by comparing Fourier transform Infrared (FTIR) measurements with DFT predictions.

Strategy • 13 C isotopic shifts necessary for species and structure determination. • Low 13 C enrichment (~10%) limits isotopic shifts to single 13 C isotopomers, which is useful for large Cn clusters (n>6). • But for small clusters (n<5), using ~50% 13 C enrichment produces all of the 13 C isotopomers.
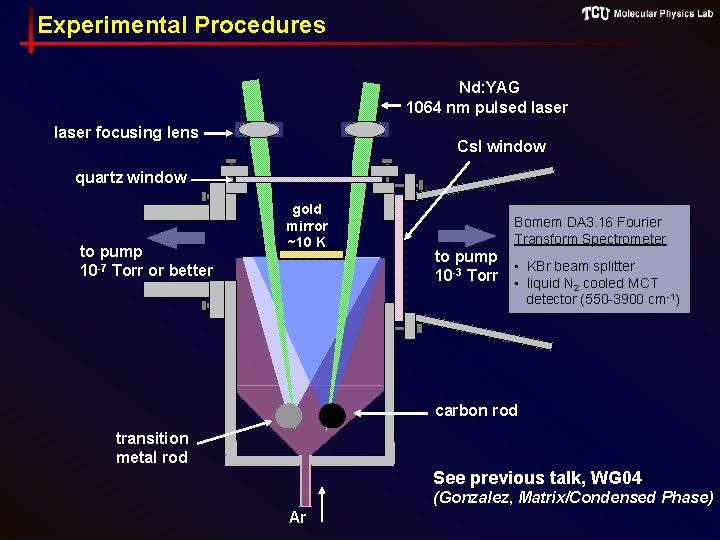
Experimental Procedures Nd: YAG 1064 nm pulsed laser focusing lens Cs. I window quartz window to pump 10 -7 Torr or better gold mirror ~10 K Bomem DA 3. 16 Fourier Transform Spectrometer to pump • KBr beam splitter 10 -3 Torr • liquid N cooled MCT 2 detector (550 -3900 cm-1) carbon rod transition metal rod See previous talk, WG 04 (Gonzalez, Matrix/Condensed Phase) Ar
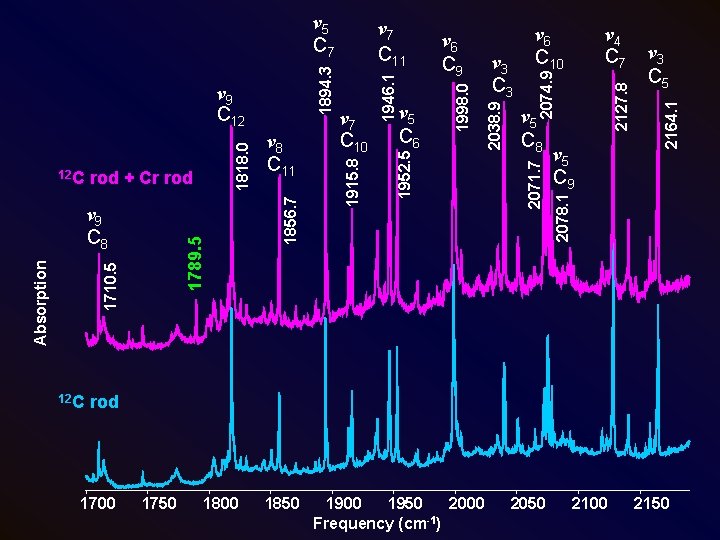
12 C 2127. 8 ν 5 C 9 ν 3 C 5 2164. 1 ν 4 C 7 2074. 9 ν 5 C 8 2071. 7 2038. 9 1998. 0 1946. 1 ν 5 C 6 ν 3 C 3 ν 6 C 10 2078. 1 1789. 5 1710. 5 Absorption ν 9 C 8 ν 7 C 10 ν 6 C 9 1952. 5 rod + Cr rod ν 8 C 11 1856. 7 12 C 1818. 0 ν 9 C 12 ν 7 C 11 1915. 8 1894. 3 ν 5 C 7 rod 1700 1750 1800 1850 1900 1950 2000 -1 Frequency (cm ) 2050 2100 2150
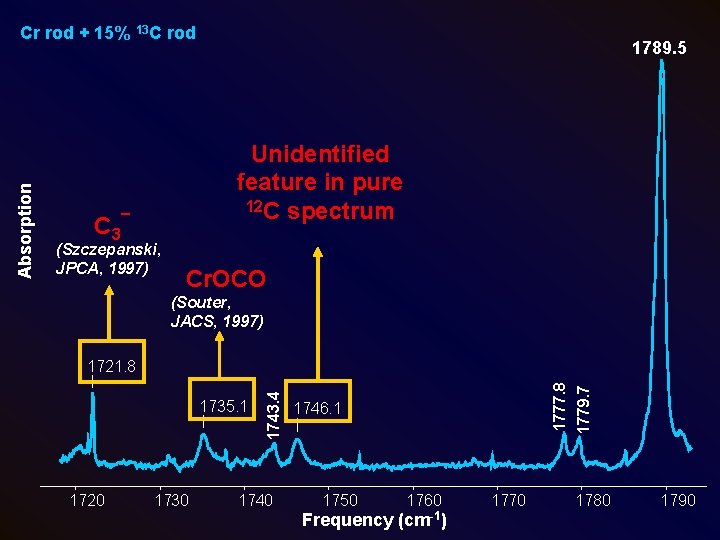
1789. 5 Unidentified feature in pure 12 C spectrum C 3¯ (Szczepanski, JPCA, 1997) Cr. OCO (Souter, JACS, 1997) 1720 1730 1746. 1 1750 1760 Frequency (cm-1) 1770 1779. 7 1735. 1 1777. 8 1721. 8 1743. 4 Absorption Cr rod + 15% 13 C rod 1780 1790
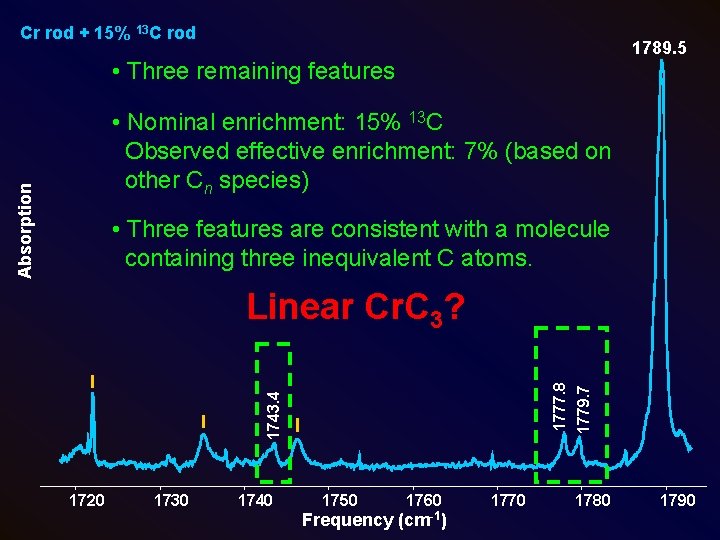
Cr rod + 15% 13 C rod 1789. 5 • Three remaining features Absorption • Nominal enrichment: 15% 13 C Observed effective enrichment: 7% (based on other Cn species) • Three features are consistent with a molecule containing three inequivalent C atoms. 1720 1730 1740 1750 1760 Frequency (cm-1) 1770 1779. 7 1743. 4 1777. 8 Linear Cr. C 3? 1780 1790
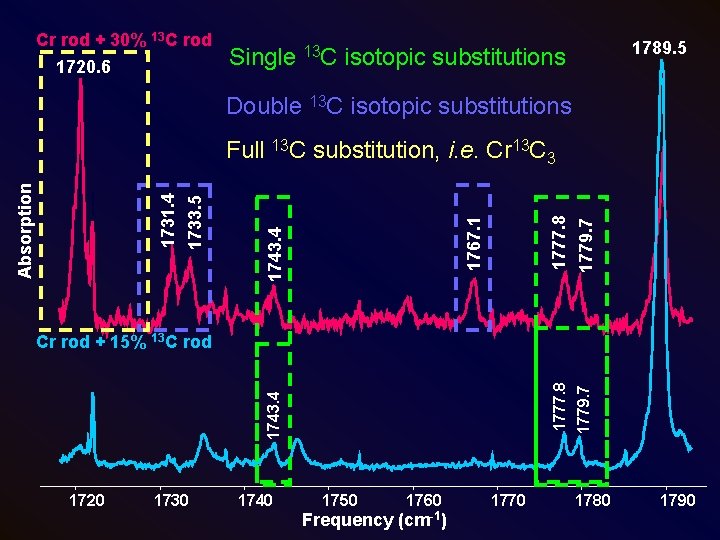
Cr rod + 30% 13 C rod 1720. 6 1789. 5 Single 13 C isotopic substitutions Double 13 C isotopic substitutions 1777. 8 1779. 7 1767. 1 1743. 4 1733. 5 1731. 4 Absorption Full 13 C substitution, i. e. Cr 13 C 3 1743. 4 Cr rod + 15% 13 C rod 1720 1730 1740 1750 1760 Frequency (cm-1) 1770 1780 1790
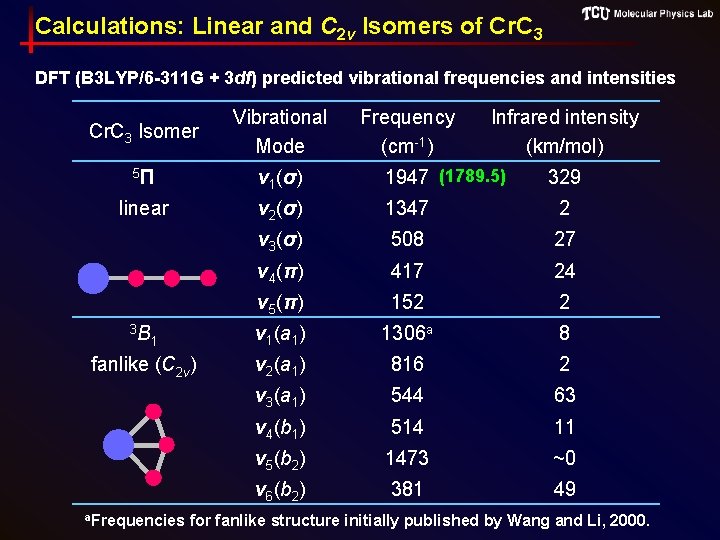
Calculations: Linear and C 2 v Isomers of Cr. C 3 DFT (B 3 LYP/6 -311 G + 3 df) predicted vibrational frequencies and intensities Cr. C 3 Isomer Vibrational Mode 5Π ν 1(σ) 1947 (1789. 5) linear ν 2(σ) 1347 2 ν 3(σ) 508 27 ν 4(π) 417 24 ν 5(π) 152 2 ν 1(a 1) 1306 a 8 ν 2(a 1) 816 2 ν 3(a 1) 544 63 ν 4(b 1) 514 11 ν 5(b 2) 1473 ~0 ν 6(b 2) 381 49 3 B 1 fanlike (C 2 v) a. Frequencies Frequency (cm-1) Infrared intensity (km/mol) 329 for fanlike structure initially published by Wang and Li, 2000.

Theoretical Calculations • Used Gaussian 03 • Used density functional theory (DFT) with B 3 LYP functional and 6 -311+G(3 df) basis set • Calculations performed for linear and C 2 v (fan) structures • Calculations for C 2 v structure vibrational frequencies in good agreement with previous (Wang & Li, JCP, 2000) • 13 C isotopic shift frequencies were also calculated for the linear isomer
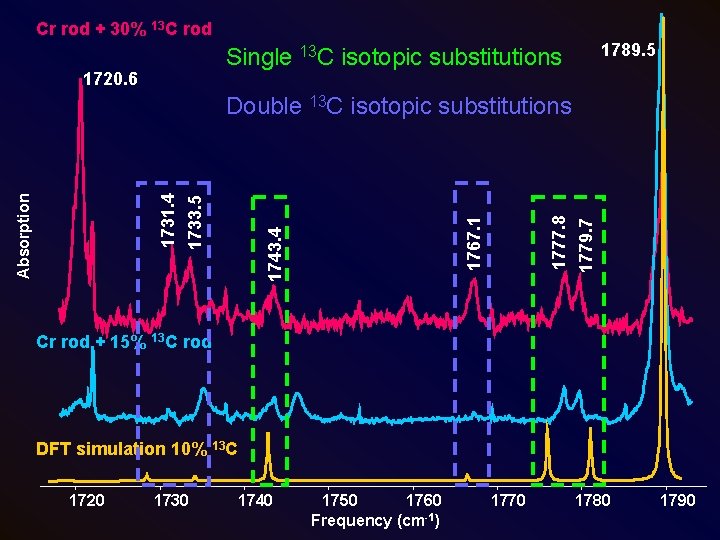
Cr rod + 30% 13 C rod 1789. 5 Single 13 C isotopic substitutions 1720. 6 1779. 7 1777. 8 1767. 1 1743. 4 1733. 5 Absorption 1731. 4 Double 13 C isotopic substitutions Cr rod + 15% 13 C rod DFT simulation 10% 13 C 1720 1730 1740 1750 1760 Frequency (cm-1) 1770 1780 1790

Calculations: Isotopic Shift Frequencies for the ν 1(σ) Mode of Linear Cr. C 3 Comparison of observed vibrational frequencies (cm-1) of the ν 1(σ) mode for 13 C-substituted isotopomers of linear Cr. C 3 with the predictions of B 3 LYP/6 -311 G + (3 df) calculations Isotopomer Cr-C-C-C Observed B 3 LYP/ 6 -311 G+(3 df) Scaleda Difference νobs νDFT νsc Δν=νobs-νsc 52 -12 -12 -12 (A) 1789. 5 1947. 4 … … 52 -13 -12 -12 (B) 1777. 8 1932. 5 1775. 8 2. 0 52 -12 -13 -12 (C) 1743. 4 1896. 8 1743. 0 0. 4 52 -12 -12 -13 (D) 1779. 7 1938. 0 1780. 9 -1. 2 52 -13 -13 -13 (A') 1720. 6 1872. 4 1720. 6 0. 0 52 -12 -13 -13 (B') 1733. 5 1888. 6 1735. 5 -2. 0 52 -13 -12 -13 (C') 1767. 1 1923. 8 1767. 8 -0. 7 52 -13 -13 -12 (D') 1731. 4 1883. 3 1730. 6 0. 8 a. DFT calculations scaled by a factor of 1789. 5/1947. 4=0. 91892.
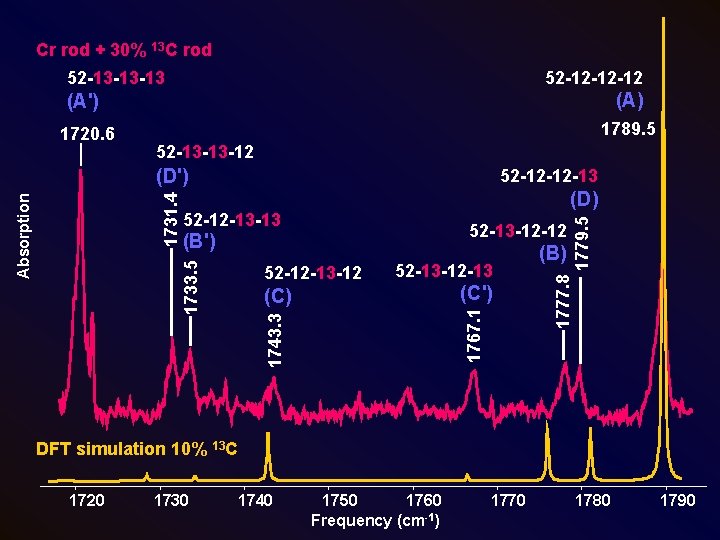
Cr rod + 30% 13 C rod 52 -12 -12 -12 52 -13 -13 -13 (A') (A) 1720. 6 1789. 5 52 -13 -13 -12 52 -12 -12 -13 52 -13 -12 -12 52 -12 -13 -12 52 -13 -12 -13 (C') 1743. 3 1767. 1 (C) (B) 1777. 8 (B') 1779. 5 (D) 52 -12 -13 -13 1733. 5 Absorption 1731. 4 (D') DFT simulation 10% 13 C 1720 1730 1740 1750 1760 Frequency (cm-1) 1770 1780 1790

Conclusions • The linear isomer of Cr. C 3 has been observed. • The ν 1(σ) mode is assigned to 1789. 5 cm-1. • No evidence of the C 2 v (“fan”) structure is observed. Four modes are predicted to lie within detector range. The strongest mode at 544 cm-1 is predicted to be ~20% of the intensity of the ν 1(σ) mode of the linear structure and should be observable. • Observation of the linear ground state structure is consistent with thermal behavior in PES experiments.

Acknowledgments • Our group would like to acknowledge funding from – Welch Foundation – TCU Research and Creative Activities Fund (TCURCAF) – W. M. Keck Foundation • Personal funding acknowledgments – Barnett Scholarship – Texas Space Grant Consortium Fellowship (TSGC)

References 1. 2. D. N. Davis, Astrophys. J. 106, 28 (1947). J. Cernicharo and M. Guélin, Astron. and Astrophys. 183, L 10 (1987). 3. L. M. Ziurys, C. Savage, J. L. Highberger, A. J. Apponi, M. Guélin, and J. Cernicharo, Astrophys. J. 564, L 45 (2002). 4. J. L. Highberger, C. S. Savage, J. H. Bieging, and L. M. Ziurys, Astrophys. J. 562, 790 (2001). 5. A. J. Apponi, M. C. Mc. Carthy, C. A. Gottlieb, and P. Thaddeus, Astrophys. J. 516, L 103 (1999). 6. M. Guélin, S. Muller, J. Cernicharo, A. J. Apponi, M. C. Mc. Carthy, C. A. Gottlieb, and P. Thaddeus, Astron. and Astrophys. 363, L 9 (2000). 7. K. H. Hinkle, J. J. Keady, and P. F. Bernath, Science 241, 1319 (1988). 8. J. Cernicharo, J. R. Goicoechea, and E. Caux, Astrophys. J. 534, L 199 (2000). 9. P. F. Bernath, K. H. Hinkle, and J. J. Keady, Science 244, 562 (1989). 10. B. C. Guo, K. P. Kerns, and A. W. Castleman, Jr. , Science 255, 1411 (1992).

References 11. B. C. Guo and A. W. Castleman, Jr. , in Advances in Metal and Semiconductor Clusters, ed. M. A. Duncan (Jai Press, London, 1994), Vol. 2, 137. 12. S. E. Kooi, B. D. Leskiw, and A. W. Castleman, Jr. , Nano Letters 1, 113 (2001). 13. S. Wei, B. C. Guo, J. Purnell, S. Buzza, and A. W. Castleman, Jr. , J. Phys. Chem. 96, 4166 (1992). 14. K. Tono, A. Terasaki, T. Ohta, and T. Kondow, J. Chem. Phys. 117, 7010 (2002). 15. S. -L. Wang and X. Li, J. Chem. Phys. 112, 3602 (2000). 16. H. -J. Zhai, L. -S. Wang, P. Jena, G. L. Gustev, and C. W. Bauschlicher, Jr. , J. Chem. Phys. 120, 8996 (2004). 17. M. E. Jacox, NIST Vibrational and Electronic Energy Levels Database (http: //webbook. nist. gov/chemistry) 18. J. Szczepanski, S. Eckern, C. Chapo, and M. Vala, Chem. Phys. 211, 359 (1996). 19. P. F. Souter and L. Andrews, J. Am. Chem. Soc. 119, 7350 (1997). 20. J. Szczepanski, C. Wehlburg, M. Vala, J. Phys. Chem. A 101, 7039 (1997).
- Slides: 20