Frontier Molecular Orbitals FMOs From Theory to Application

Frontier Molecular Orbitals (FMOs): From Theory to Application ┄Take Diels-Alder Cycloaddition for Example Ming-Liang Lou 4/9/2016

一、Introduction to FMOs 1. Comparison between molecular orbital and atomic orbital 1) Similar to an atomic orbital (AO), a molecular orbital (MO) describes the probability of finding an electron in a given region. The difference is that a molecular orbital can be spread across multiple atoms within the same molecule; 2) Atomic orbitals are labelled 1 s, 2 p, etc. where the letter (s, p, d or f) indicates the orbital shape. Molecular orbitals are labelled 1 σ, 2 σ, 1 π, 2 π, etc. where the Greek letter (σ or π) indicates the orbital symmetry; 3) Molecular orbitals are generated by combining atomic orbitals.
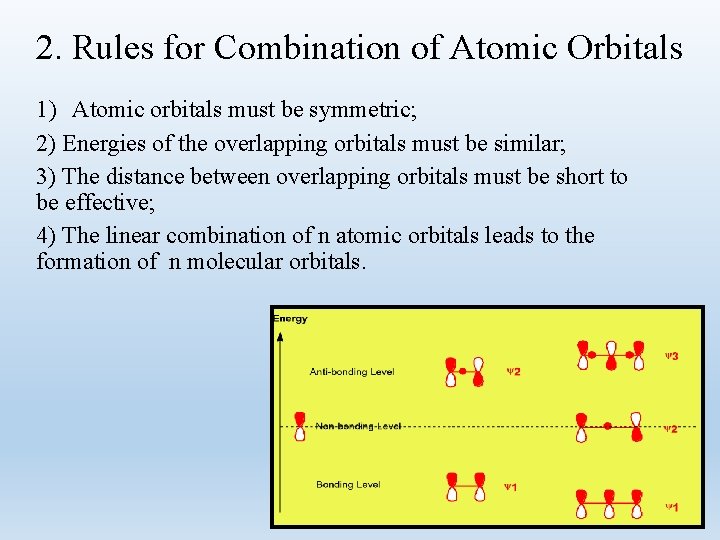
2. Rules for Combination of Atomic Orbitals 1) Atomic orbitals must be symmetric; 2) Energies of the overlapping orbitals must be similar; 3) The distance between overlapping orbitals must be short to be effective; 4) The linear combination of n atomic orbitals leads to the formation of n molecular orbitals.
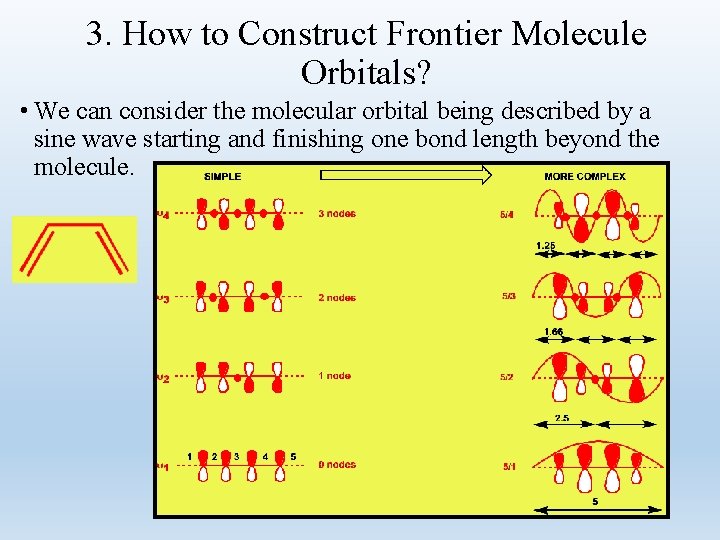
3. How to Construct Frontier Molecule Orbitals? • We can consider the molecular orbital being described by a sine wave starting and finishing one bond length beyond the molecule.
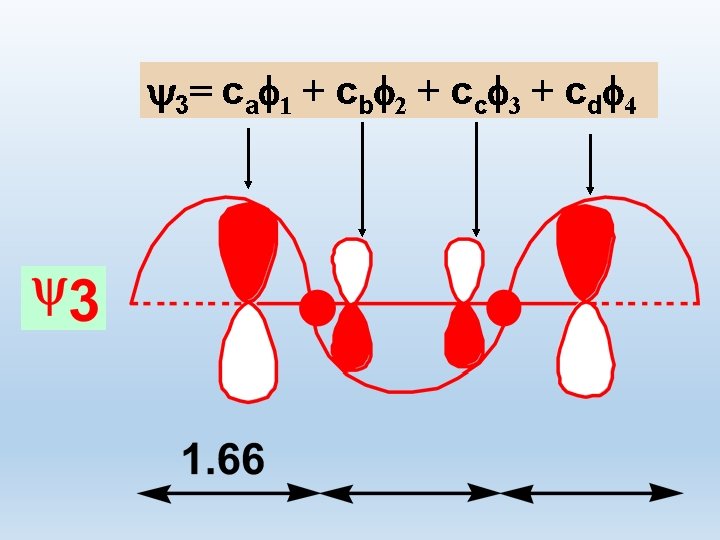
3 = c a f 1 + c b f 2 + c c f 3 + c d f 4

二、Application of Frontier Molecule Orbitals • 1. Introduction to Diels-Alder Cycloaddition • The discovers of the reaction (in 1928), Otto Diels and Kurt Alder, won a Nobel prize for their work in 1950. However, until the mid 1960 s there was no good explanation for how the reaction worked the way it did (a simple, concerted step). • The explanation of the Diels Alder reaction (and other related reactions) was ultimately found in molecular orbital theory and won its own Nobel prize in 1981 (Roald Hoffman and Kenichi Fukui).
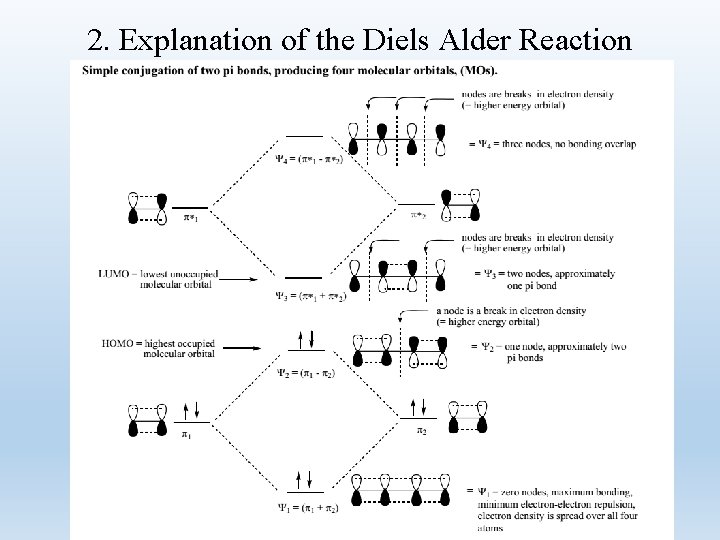
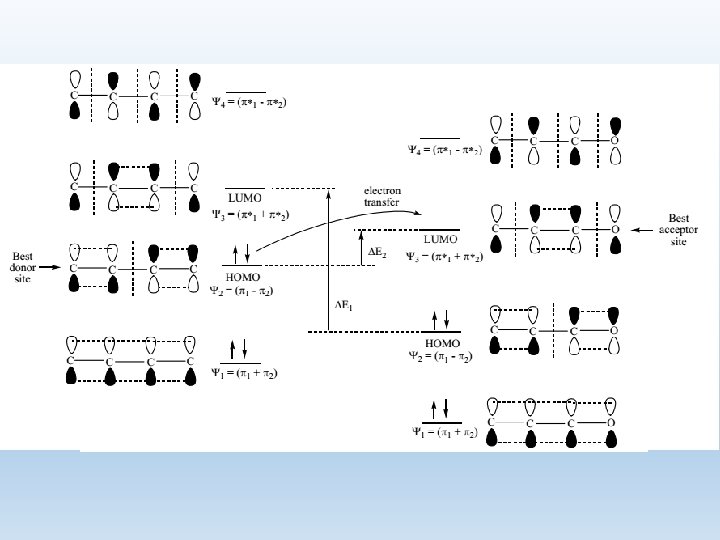
2. Explanation of the Diels Alder Reaction
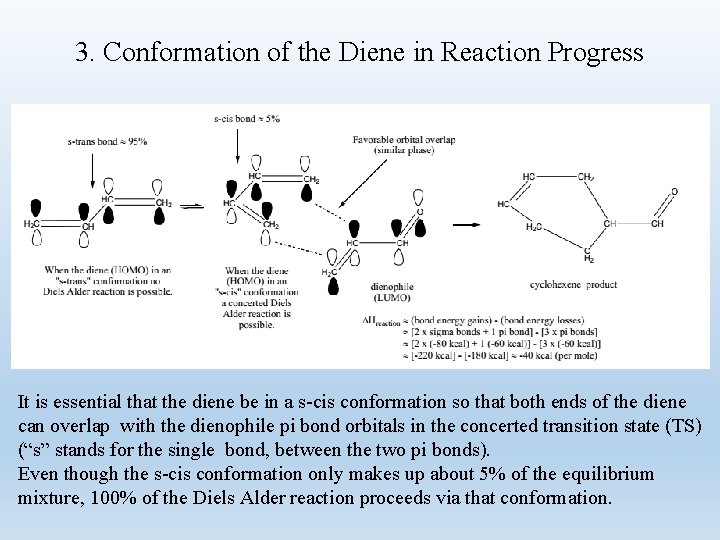
3. Conformation of the Diene in Reaction Progress It is essential that the diene be in a s-cis conformation so that both ends of the diene can overlap with the dienophile pi bond orbitals in the concerted transition state (TS) (“s” stands for the single bond, between the two pi bonds). Even though the s-cis conformation only makes up about 5% of the equilibrium mixture, 100% of the Diels Alder reaction proceeds via that conformation.
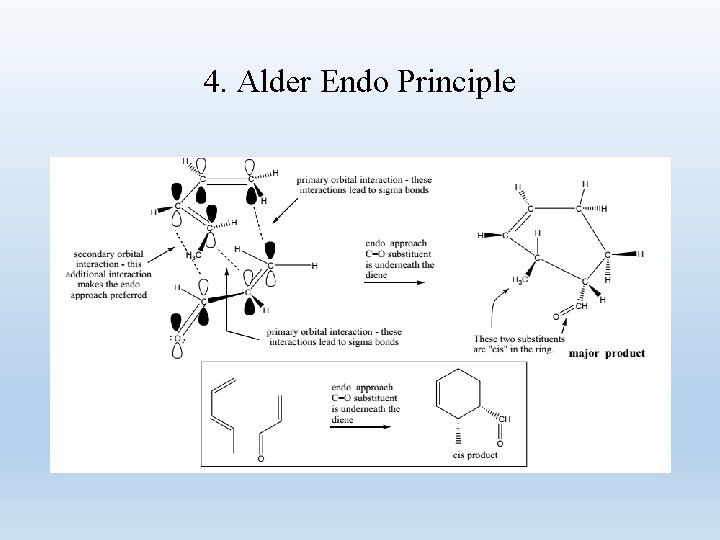
4. Alder Endo Principle
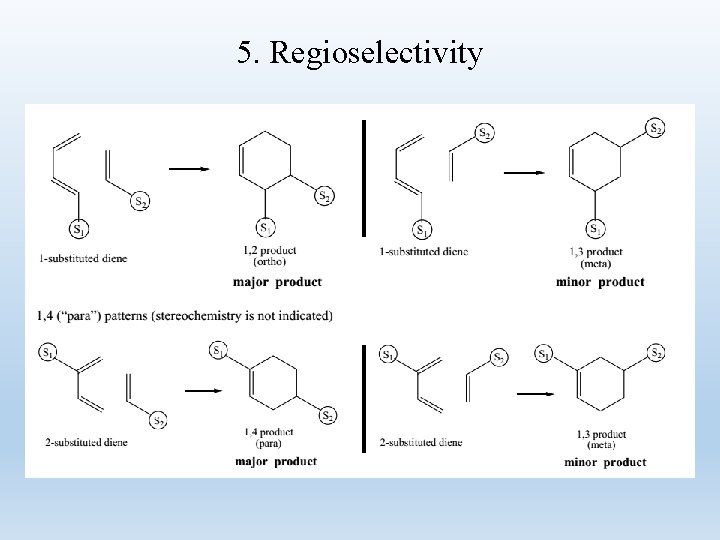
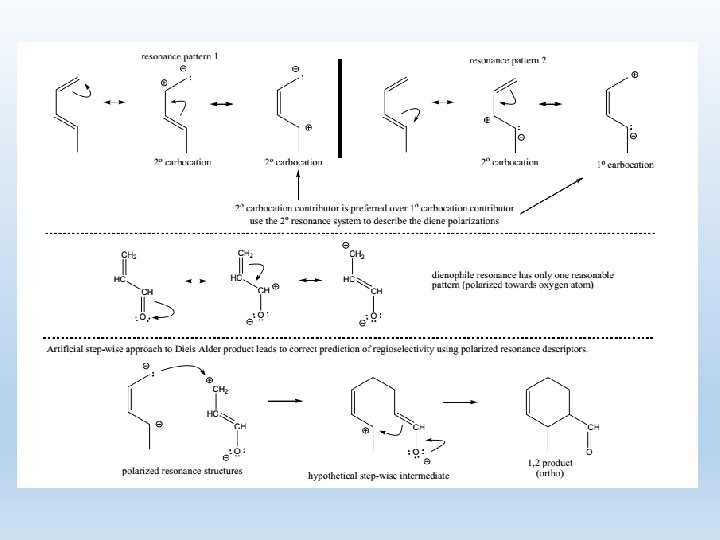
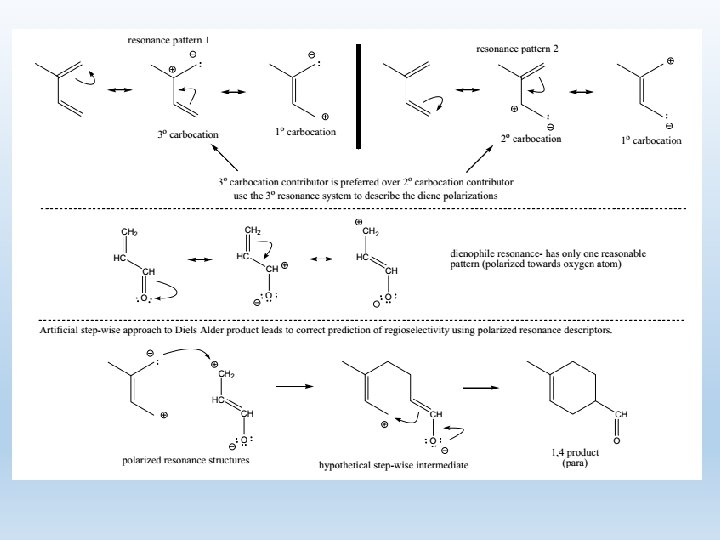
5. Regioselectivity

Thank you for your attention!
- Slides: 14