From ODMSDM to SDTM Prepared by Jozef Aerts

From ODM-SDM to SDTM Prepared by Jozef Aerts - XML 4 Pharma Presented by Kurt Hellstern, Hands-on Gmb. H

The ODM-SDM Extension • • SDM = Study Design Model Draft v. 1. 0 published June 2011 Final expected October 2011 treats: – Study Parameters – Eligibility Criteria (inclusion-exclusion) – Study Structure: arms, epochs, cells, segments, activities – Workflows – Timings • SDM is an extension to ODM 1. 3. 1 => XML !

Elements in ODM and ODM-SDM • In ODM: <Study. Event. Def …. …> …. . </ Study. Event. Def > • In ODM-SDM: <sdm: Summary> …. </sdm: Summary>

Study Parameters Maps 1: 1 to SDTM Domain TS (Trial Summary)
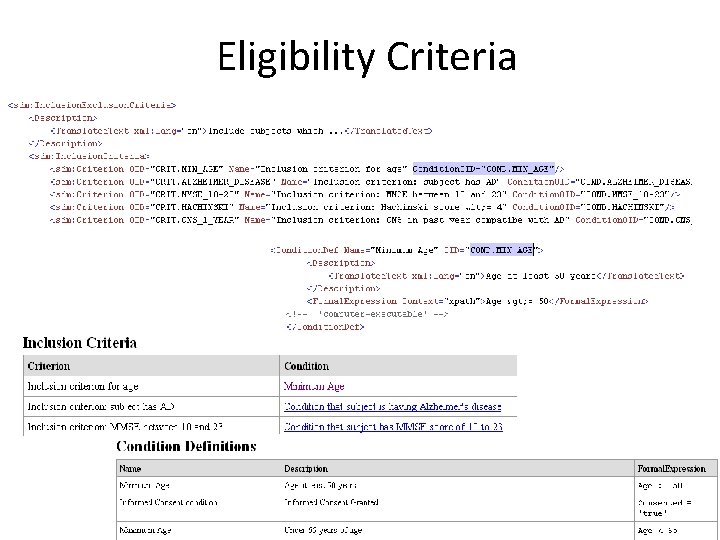
Eligibility Criteria

Eligibility Criteria • list of inclusion and exclusion criteria • criterium itself (Condition. Def) – human-readable (Description) – machine-readable (Formal. Expression) • 1: 1 mapping to SDTM domain TI (Trial Inclusion/exclusion Criteria)

Study Structure • • Arms Epochs Cells Segments • Activities (new)
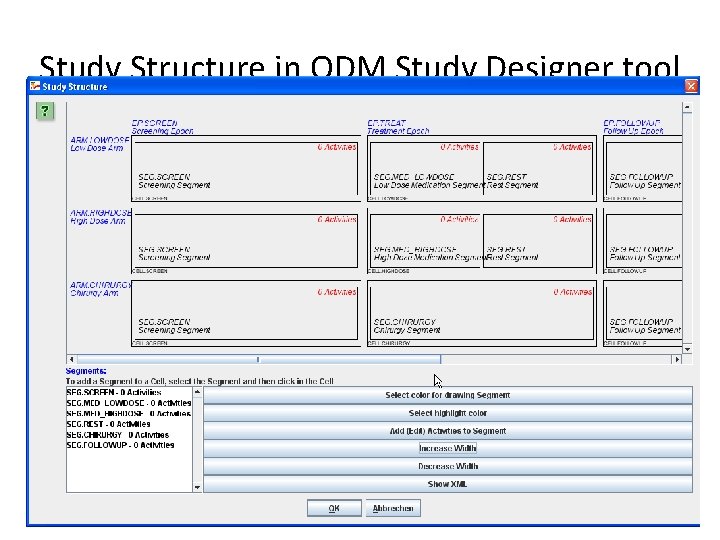
Study Structure in ODM Study Designer tool

Study Structure • Content (XML) can be used to automatically populate SDTM datasets – TA (Trial Arms) – TE (Trial Elements) • Workflow Section (see further) enables to add: – entry / exit criteria for epoch, cell, segment. . . (SDTM TESTRL, TEENRL) • Timing Section (see further) enables to add: – estimated duration (SDTM TEDUR)

Workflow • workflow between activities • visits (Study. Event) can have several activities, e. g. : – visit start activity (for scheduling) – fill-a-form activity – do-a-test activity – give medication activity • >90% of all clinical study workflows can be described
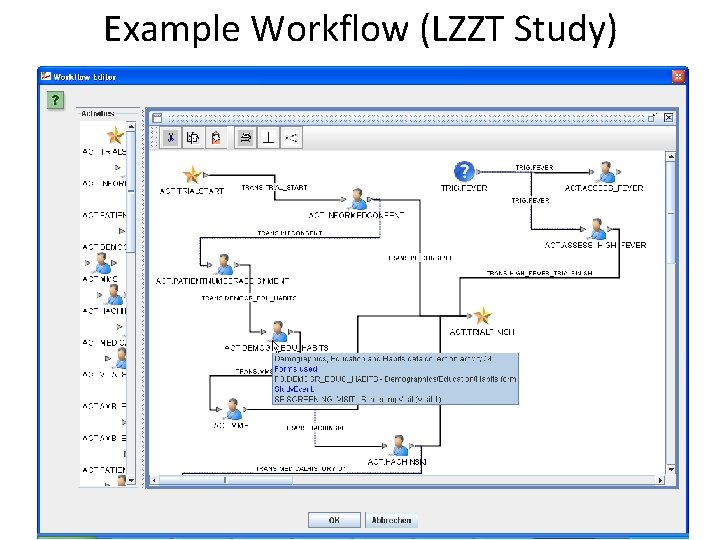
Example Workflow (LZZT Study)

ODM-SDM Workflows can be transformed • There are many "standards" for describing workflows, e. g. – MS Workflow Foundation – BPEL – BPMN 2. 0 – XPDL. . . • ODM-SDM workflows can easily be transformed into these, e. g. for use in Hospital Planning Systems
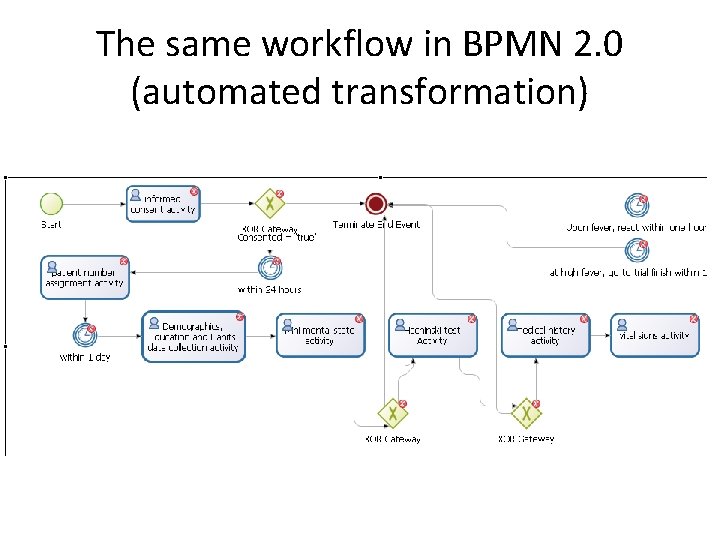
The same workflow in BPMN 2. 0 (automated transformation)

Timings • Timings and timing windows can be defined on: – activities (e. g. "informed consent between January 1 and February 28") – transitions between activities (e. g. "activity 2 must follow activity 1 within 24 hours with a time window of -2 and +4 hours") – activity durations (e. g. "activity is planned to be take not more than 2 hours")

Conclusions • ODM-SDM is a first step into a full machinereadable Protocol • SDTM Trial Design datasets can be automatically generated from ODM-SDM study designs • ODM-SDM can contain much more detailed information than the corresponding SDTM datasets • ODM-SDM is extremely helpfull for integration with hospital information systems • Design tools are already available, other vendors will quickly follow

ODM-SDM can contain much more detailed information of a study design than the corresponding SDTM datasets • If the FDA would like to be able to fully reconstruct the design of a study. . . • They could better accept ODM-SDM instead of Trial Design SDTM datasets
- Slides: 16