From Bench to Bedside Step 1 Firsttime application

From Bench to Bedside Step 1: First-time application of DESWT in a rodent model of ischemic HF (pilot trial); redesign of device Step 2: In-vitro validation Step 3: Reproduction of results of pilot trial in a rodent large scale trial (320 rodents); human fitting studies Step 4: Reproduction of the results of the rodent large scale trial in a large animal (pig) model; human fitting studies Step 5: Phase I clinical trial with external data monitoring

Pilot Trials show promising results, exceeding comparable stem cell trials. Main problem: size of the applicator

8 Weeks Later: Cardiac Shock Wave Applicator Cardio. Gold® CG 050 CRT – Cardiac Regeneration Technologies, Woodstock, USA (manufactured by MTS-Europe Gmb. H, Konstanz, Germany) Electro-hydraulic, focused, ECG-triggering facility EFD: 0. 38 m. J/mm 2 Frequency: 1 -5 Hz 35 mm

In-vitro Experiments distance bar adapter temp. sensor Shock Wave Devices Water bath – prototype appl. fixation Degassed water, temperature 37°C heater Derma. Gold® CP 155 (TRT, USA) 300 impulses at 0. 15 m. J/mm 2, 5 Hz
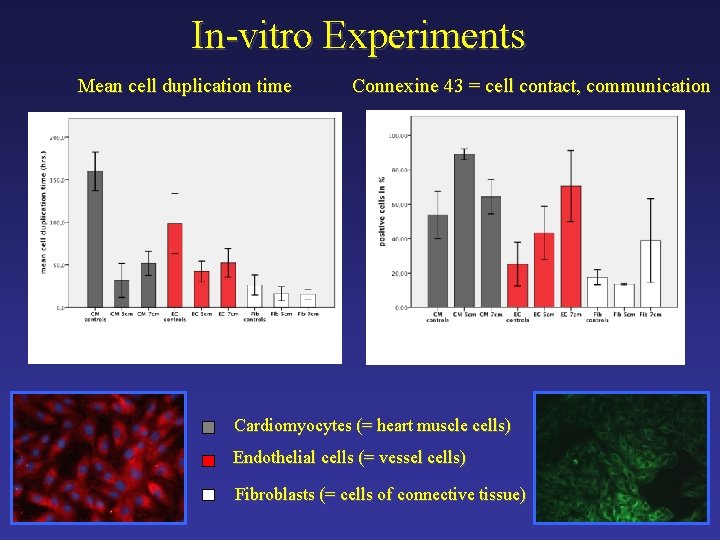
In-vitro Experiments Mean cell duplication time Connexine 43 = cell contact, communication Cardiomyocytes (= heart muscle cells) Endothelial cells (= vessel cells) Fibroblasts (= cells of connective tissue)
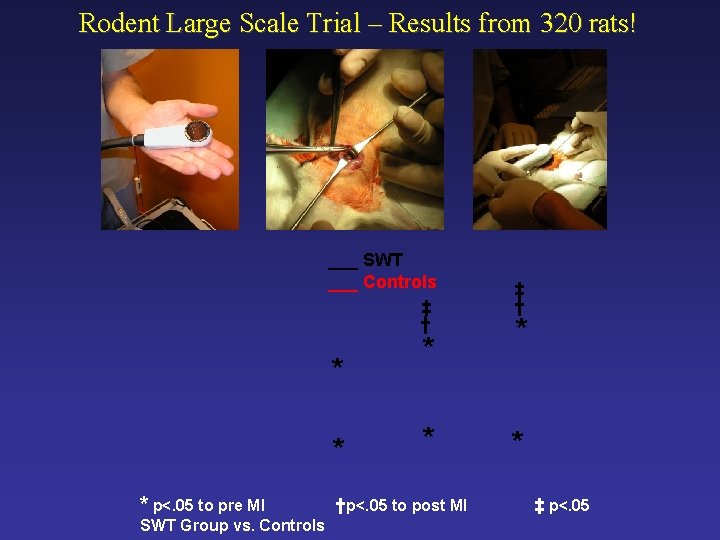
Rodent Large Scale Trial – Results from 320 rats! ___ SWT ___ Controls ‡ † * * * p<. 05 to pre MI SWT Group vs. Controls * * †p<. 05 to post MI ‡ † * * ‡ p<. 05
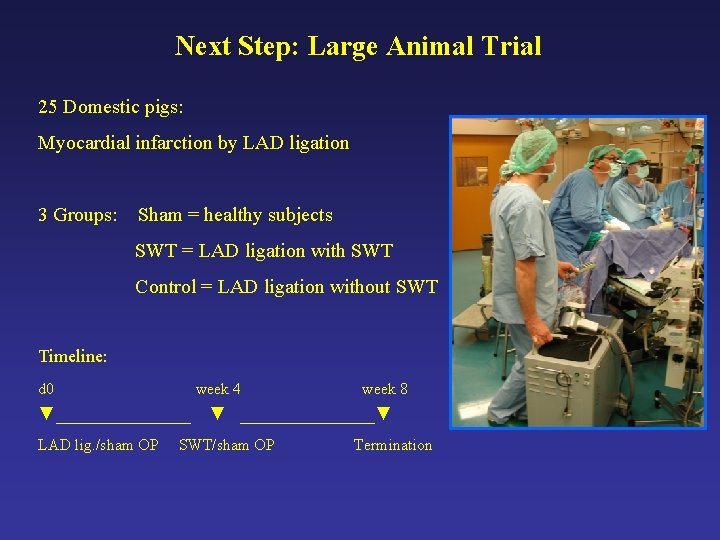
Next Step: Large Animal Trial 25 Domestic pigs: Myocardial infarction by LAD ligation 3 Groups: Sham = healthy subjects SWT = LAD ligation with SWT Control = LAD ligation without SWT Timeline: d 0 week 4 ▼________ LAD lig. /sham OP week 8 ▼ ________▼ SWT/sham OP Termination
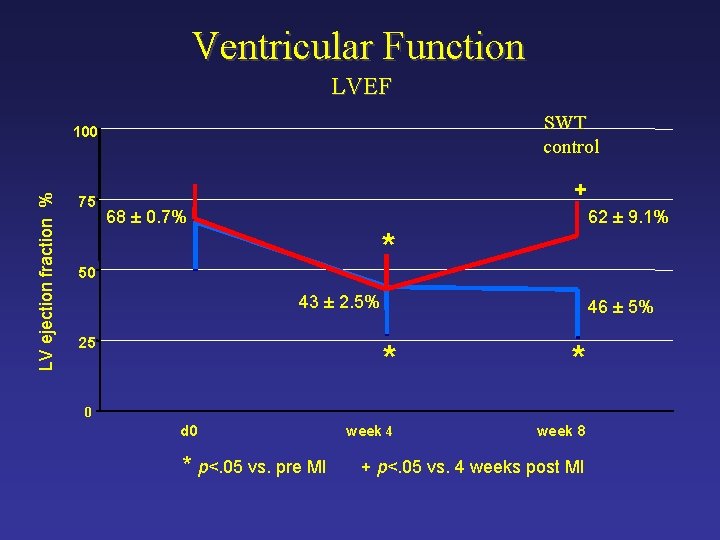
Ventricular Function LVEF SWT control LV ejection fraction % 100 75 + 68 ± 0. 7% 62 ± 9. 1% * 50 43 ± 2. 5% 25 46 ± 5% * * week 4 week 8 0 d 0 * p<. 05 vs. pre MI + p<. 05 vs. 4 weeks post MI

First Human Application – Study Design Title: DESWT - Safety of Myocardial Regeneration by Direct Epicardial Shock Wave Therapy in Combination with Coronary Artery Bypass Grafting Design: Prospective, Phase I Enrollment: Commenced September 2008, 10 Patients – last patient enrolled June 2009 Primary Endpoint: Safety Secondary Endpoint: Efficacy Study outline: CABG+DESWT 8 -week FUP 6 -month FUP ▼________▼_______▼

First Human Application (Sep. 23, 2008)

First Human Application (Sep. 23, 2008)
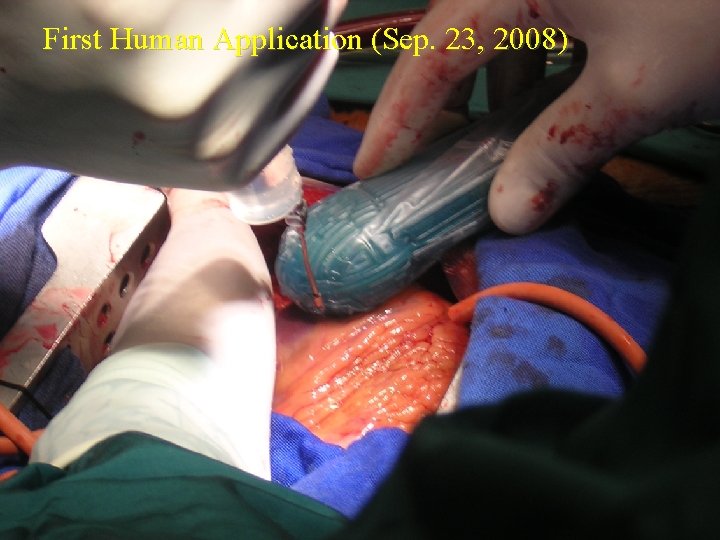
First Human Application (Sep. 23, 2008)

First Human Application (Sep. 23, 2008)

Primary Endpoint - Safety Ø Death Ø Myocardial infarction Ø Bleeding, including pericardial tamponade Ø Ventricular arrhythmias Ø Bacteriaemia / sepsis Ø Respiratory failure Ø CPR Ø Lesions related to DESWT (cardiac, intrathoracic) Ø Renal failure Ø Liver failure Ø Low cardiac output syndrome Ø CVA (Stroke/Bleeding/TIA) Ø Re-hospitalization Ø Other non-anticipated events fulfilling the definition for AEs/SAEs

Secondary Endpoint - Efficacy Ø Myocardial contractility Ø Cardiac MRI Ø Symptoms of heart failure Ø Minnesota Heart Failure Questionnaire Ø Six-minute walk test Ø Serum levels of pro. BNP Ø Device failures and malfunctions

Primary Endpoint AE/SAE´s Perioperative 1 Pat. ventricular arrhytmia 1 st. to 3 rd p. o. day 1 Pat. prolonged ICU stay, mediastinitis 1 Pat. post-op bleeding (no surgical revision) Follow-Up 3 Pat. rehospitalization (embolism, pneumonia, pacemaker implant) None of the complications device related!
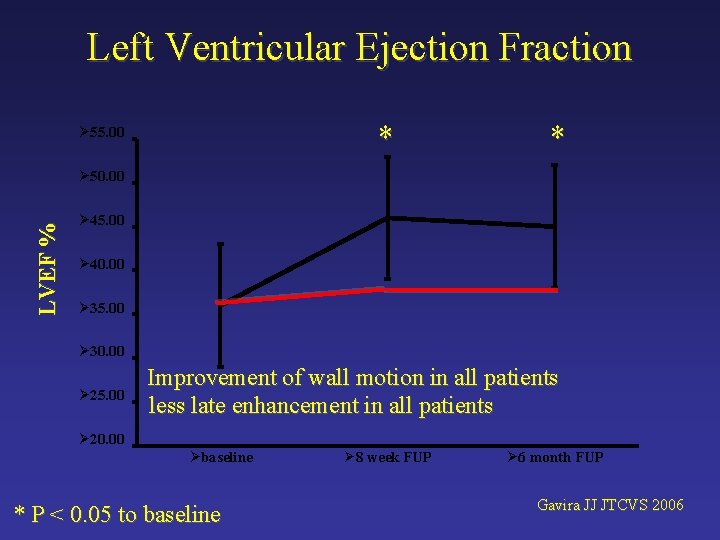
Left Ventricular Ejection Fraction * Ø 55. 00 * LVEF % Ø 50. 00 Ø 45. 00 Ø 40. 00 Ø 35. 00 Ø 30. 00 Ø 25. 00 Improvement of wall motion in all patients less late enhancement in all patients Ø 20. 00 Øbaseline * P < 0. 05 to baseline Ø 8 week FUP Ø 6 month FUP Gavira JJ JTCVS 2006
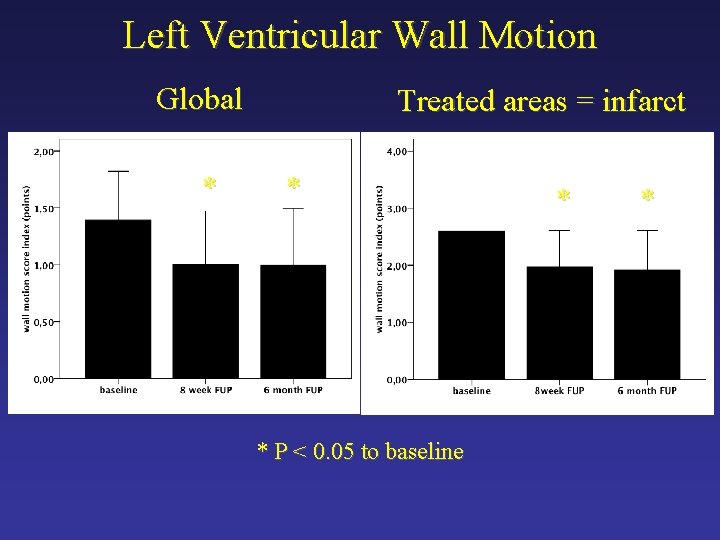
Left Ventricular Wall Motion Global * Treated areas = infarct * * P < 0. 05 to baseline * *
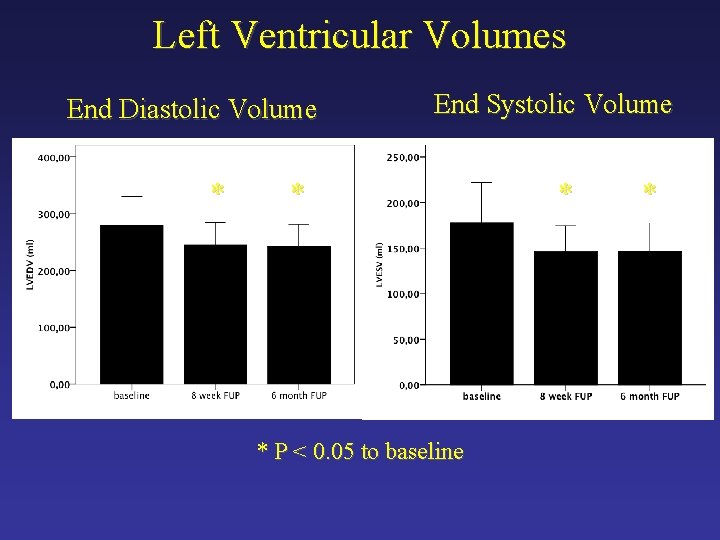
Left Ventricular Volumes End Diastolic Volume * End Systolic Volume * * P < 0. 05 to baseline * *
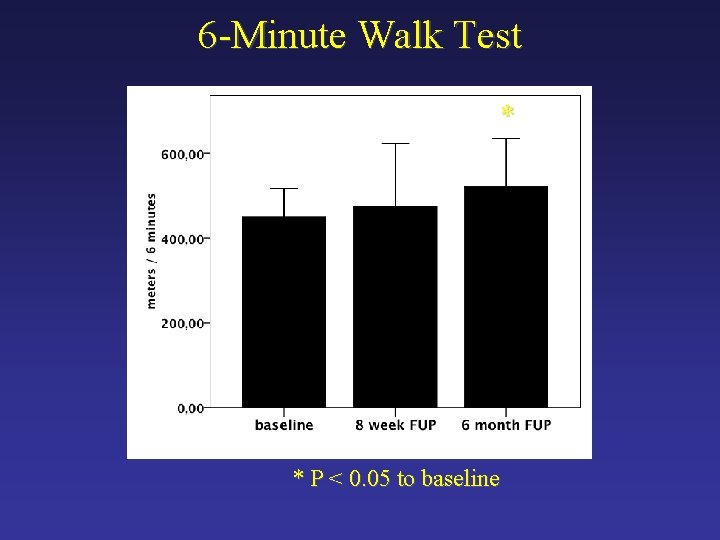
6 -Minute Walk Test * * P < 0. 05 to baseline
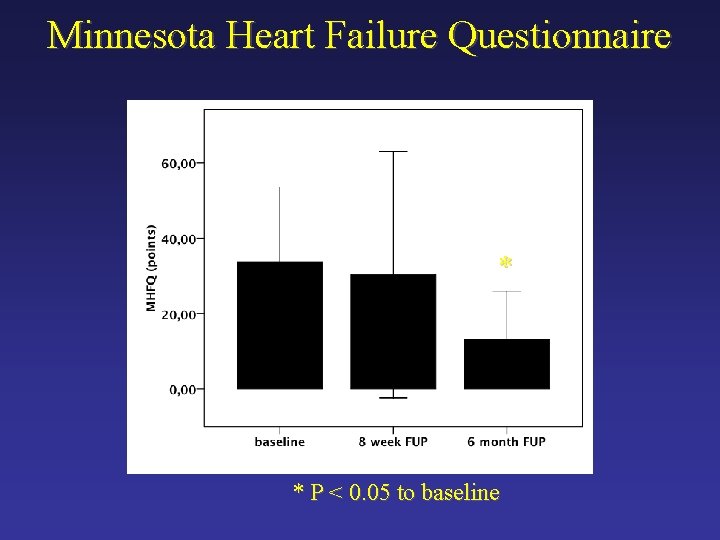
Minnesota Heart Failure Questionnaire * * P < 0. 05 to baseline

Conclusion Ø DESWT is SAFE Ø Neo – Angiogenesis Ø VEGF, PIGF, Flt-1 Ø Endogenous angiogenetic pathway activation ? Ø Stem cell homing ? Ø Improvement of LV function Ø Global and regional LV function Ø pro. BNP surrogate for heart failure Ø Improvement of Symptoms Ø 6 -MWT gold standard of symtom assessment in heart failure Ø Patients feel the difference – Minesota Heart Failure Questionnaire

Conclusion Ø Safe and simple method for myocardial regeneration Ø Bench to bedside completed Ø Next logical step – prospective randomized trial Ø Continuation of basic research (working mechanism) Ø Continuation of human trials (histology, stem cells) Ø Continuation of animal trials (dose-reaction, working mechanism) Ø Customized device, disposable therapy head Ø Cell cultivation, cell seeding on scaffolds ………
- Slides: 23