FreeFree Absorption from H I Much less than

Free-Free Absorption from H I • Much less than bf absorption • Kramers (1923) + Gaunt (1930) again • Absorption coefficient depends on the speed of the electron (slower electrons are more likely to absorb a photon because their encounters with H atoms take longer) • Adopt a Maxwell-Boltzman distribution for the speed of electrons • Again multiply by the number of neutral hydrogen atoms:

Opacity from Neutral Hydrogen • Neutral hydrogen (bf and ff) is the dominant source of opacity in stars of B, A, and F spectral type • Discussion Questions: – Why is neutral hydrogen not a dominant source of opacity in O stars: – Why not in G, K, and M stars?

Opacity from the H- Ion • Only one known bound state for bound-free absorption • 0. 754 e. V binding energy • So l < hc/hn = 16, 500 A • Requires a source of free electrons (ionized metals) • Major source of opacity in the Sun’s photosphere • Not a source of opacity at higher temperatures because H- becomes too ionized (average e- energy too high)

More H- Bound-Free Opacity • Per atom absorption coefficient for H- can be parameterized as a polynomial in l: • Peaks at 8500 A

H- Free-Free Absorption Coefficient • The free-free H- absorption coefficient depends on the speed of the electron • Possible because of the imperfect shielding of the hydrogen nucleus by one electron • Proportional to l 3 • Small at optical wavelengths • Comparable to H- bf at 1. 6 microns • Increases to the infrared

He absorption • Bound-free He- absorption is negligible (excitation potential if 19 e. V!) • Free-free He- can be important in cool stars in the IR • BF and FF absorption by He is important in the hottest stars (O and early B)

Electron Scattering • Thomson scattering: • Independent of wavelength • In hot stars (O and early B) where hydrogen dominates, then Pe~0. 5 Pg, and k(e) is independent of pressure • In cool stars, e- scattering is small compared to other absorbers for main sequence star but is more important for higher luminosity stars

Rayleigh Scattering • Generally can be neglected • But – since it depends on l-4 it is important as a UV opacity source in cool stars with molecules in their atmospheres. • H 2 can be an important scattering agent

Other Sources • Metals: C, Si, Al, Mg, Fe produce bound-free opacity in the UV • Line Opacity: Combined effect of millions of weak lines – Detailed tabulation of lines – Opacity distribution functions – Statistical sampling of the absorption • Molecules: CN-, C 2 -, H 20 - , CH 3, Ti. O are important in late and/or very late stars

Molecular Hydrogen Opacity • H 2 is more common than H in stars cooler than mid-M spectral type (think brown dwarfs!!) • H 2 does not absorb in the visible spectrum • H 2+ does, but is less than 10% of H- in the optical • H 2+ is a significant absorber in the UV • H 2 - ff absorption in the IR
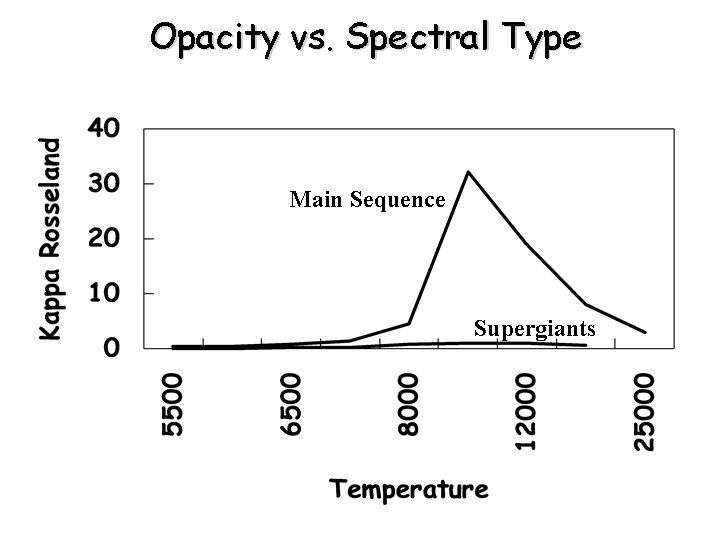
Opacity vs. Spectral Type Main Sequence Supergiants
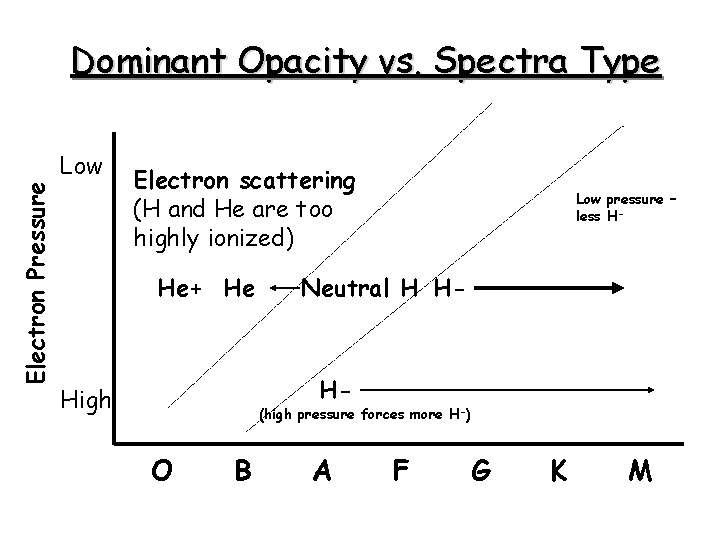
Dominant Opacity vs. Spectra Type Electron Pressure Low Electron scattering (H and He are too highly ionized) He+ He Low pressure – less H- Neutral H H- H- High (high pressure forces more H-) O B A F G K M
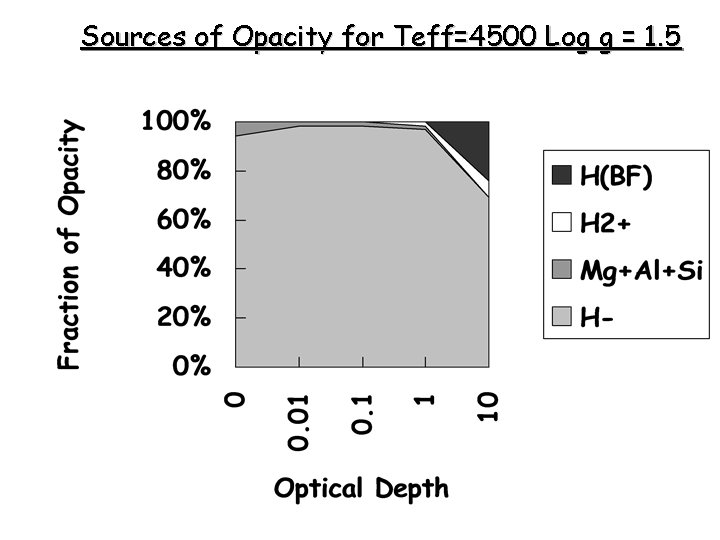
Sources of Opacity for Teff=4500 Log g = 1. 5

Class Exercise – Electron Scattering • Estimate the absorption coefficient for electron scattering for the models provided at a level where T=Teff • Recall that • and • with m in AMU and k=1. 38 x 10 -16 • How does ke compare to k. Rosseland
- Slides: 14