Free Radical substitution reactions and Their applications BY

Free Radical substitution reactions and Their applications BY DR. GHULAM ABBAS
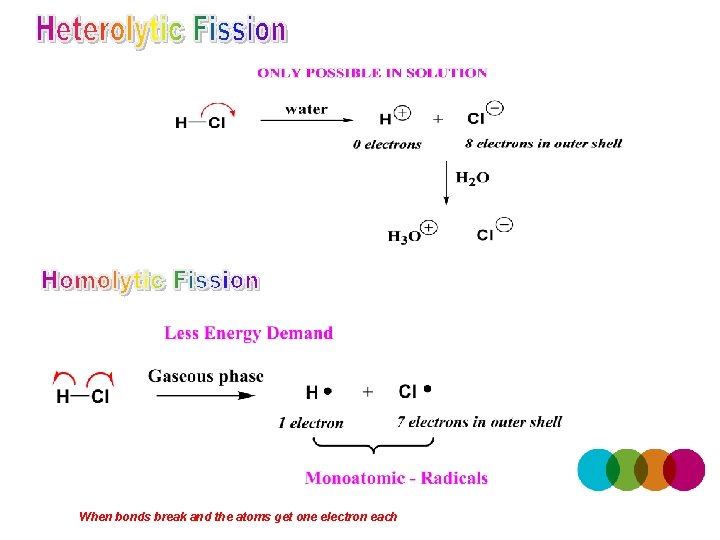
When bonds break and the atoms get one electron each
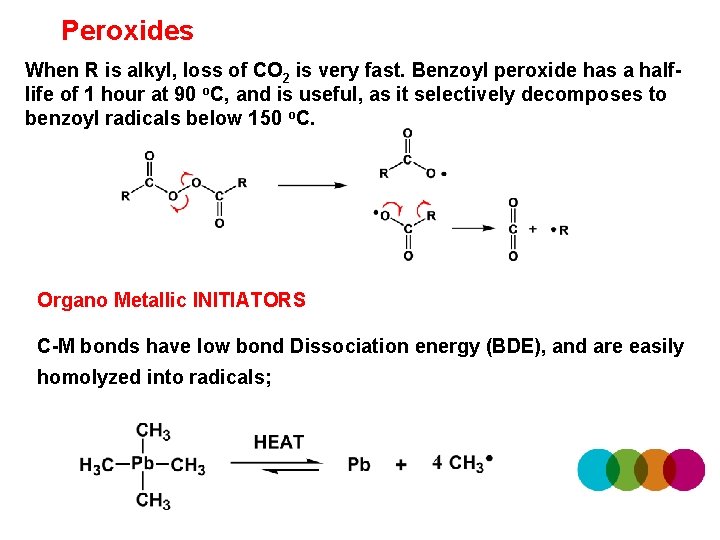
Peroxides When R is alkyl, loss of CO 2 is very fast. Benzoyl peroxide has a halflife of 1 hour at 90 o. C, and is useful, as it selectively decomposes to benzoyl radicals below 150 o. C. Organo Metallic INITIATORS C-M bonds have low bond Dissociation energy (BDE), and are easily homolyzed into radicals;
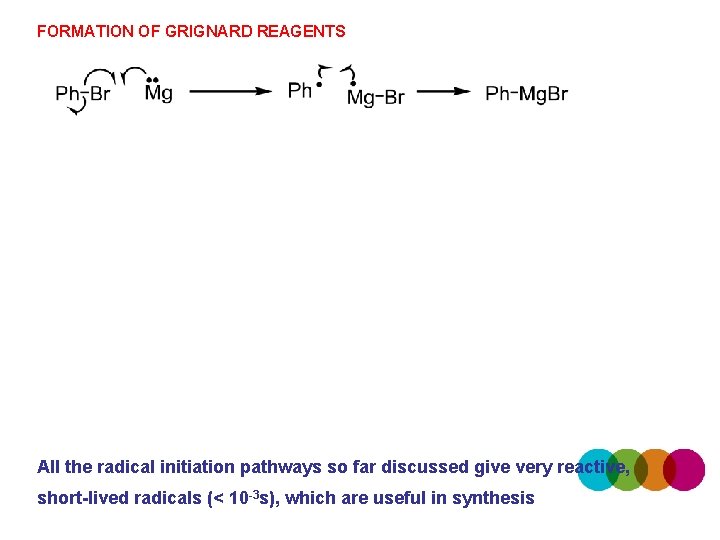
FORMATION OF GRIGNARD REAGENTS All the radical initiation pathways so far discussed give very reactive, short-lived radicals (< 10 -3 s), which are useful in synthesis

Very Stable Radicals (Half-life = years) Thermodynamic Stabilisation is most important. These radicals can be stored on the bench, and handled like other ordinary chemicals, without any adverse reaction in air or light.
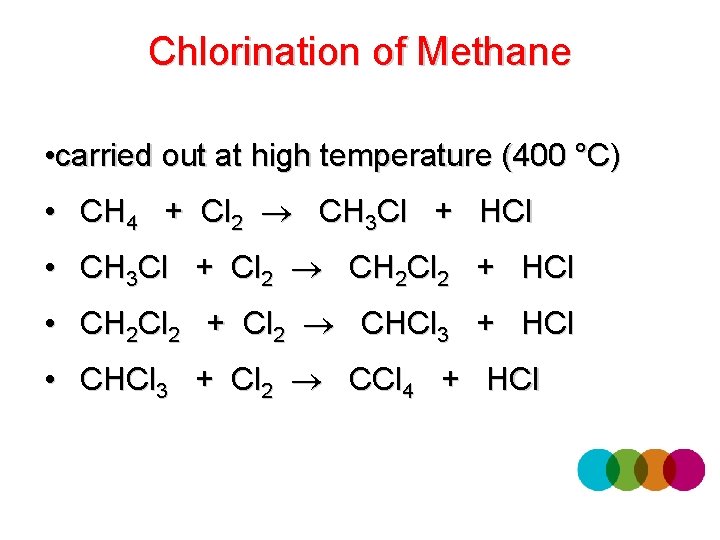
Chlorination of Methane • carried out at high temperature (400 °C) • CH 4 + Cl 2 CH 3 Cl + HCl • CH 3 Cl + Cl 2 CH 2 Cl 2 + HCl • CH 2 Cl 2 + Cl 2 CHCl 3 + HCl • CHCl 3 + Cl 2 CCl 4 + HCl
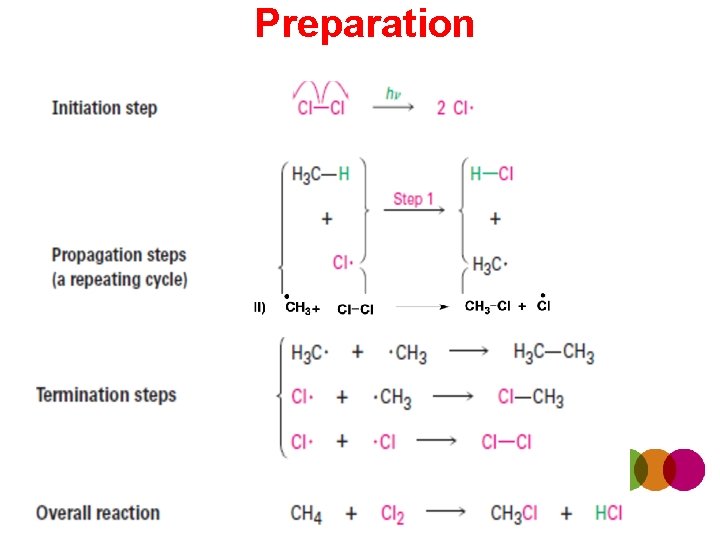
Preparation

• The relative stabilities of radicals follows the same trend as for carbocations – The most substituted radical is most stable – Radicals are electron deficient, as are carbocations, and are therefore also stabilized by hyperconjugation
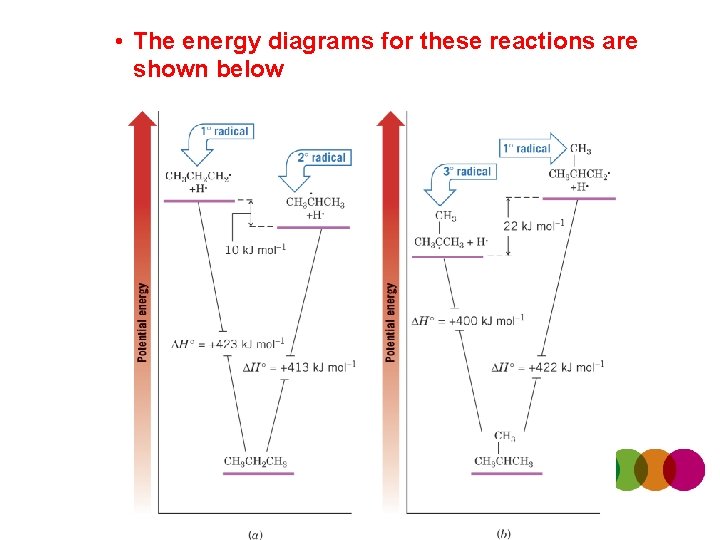
• The energy diagrams for these reactions are shown below

Free Radical Resonance • When drawing resonance for free radicals, use fishhook arrows to show electron movement • An allylic radical is stabilized by resonance
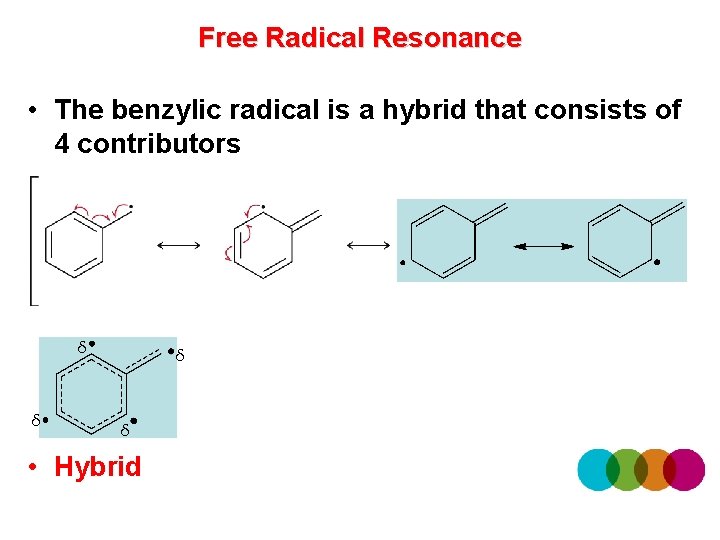
Free Radical Resonance • The benzylic radical is a hybrid that consists of 4 contributors δ δ • Hybrid
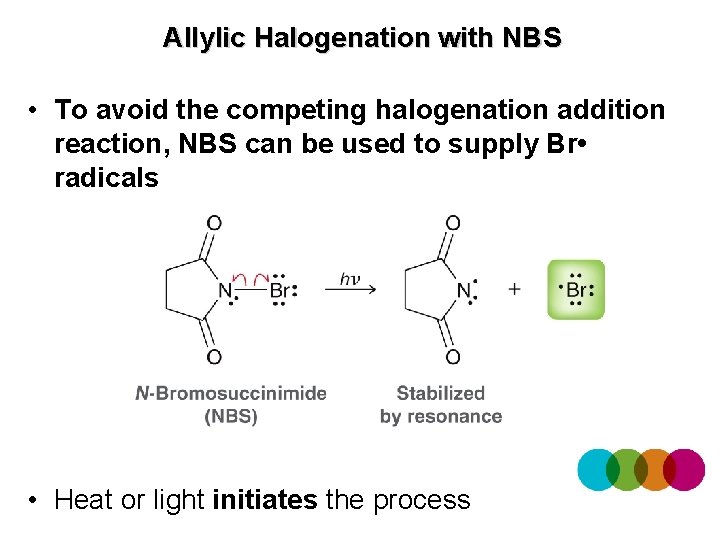
Allylic Halogenation with NBS • To avoid the competing halogenation addition reaction, NBS can be used to supply Br • radicals • Heat or light initiates the process
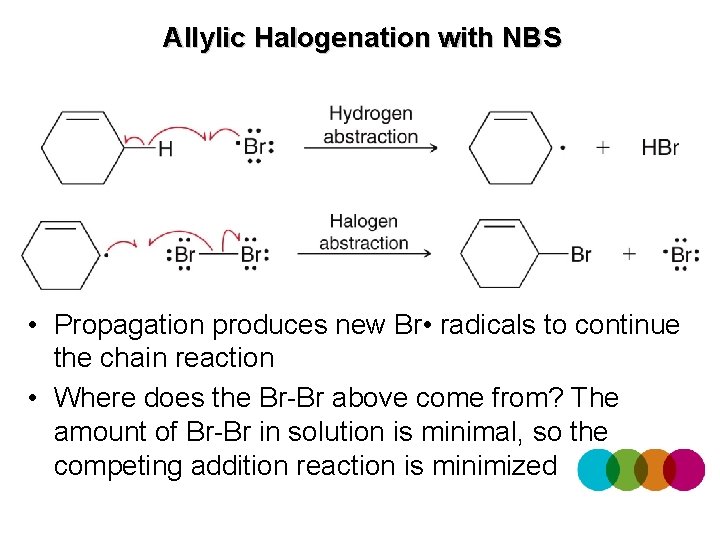
Allylic Halogenation with NBS • Propagation produces new Br • radicals to continue the chain reaction • Where does the Br-Br above come from? The amount of Br-Br in solution is minimal, so the competing addition reaction is minimized
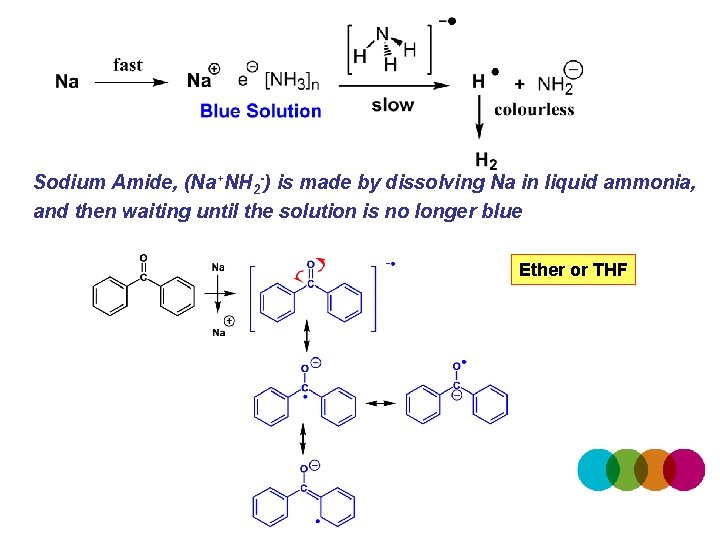
Sodium Amide, (Na+NH 2 -) is made by dissolving Na in liquid ammonia, and then waiting until the solution is no longer blue Ether or THF
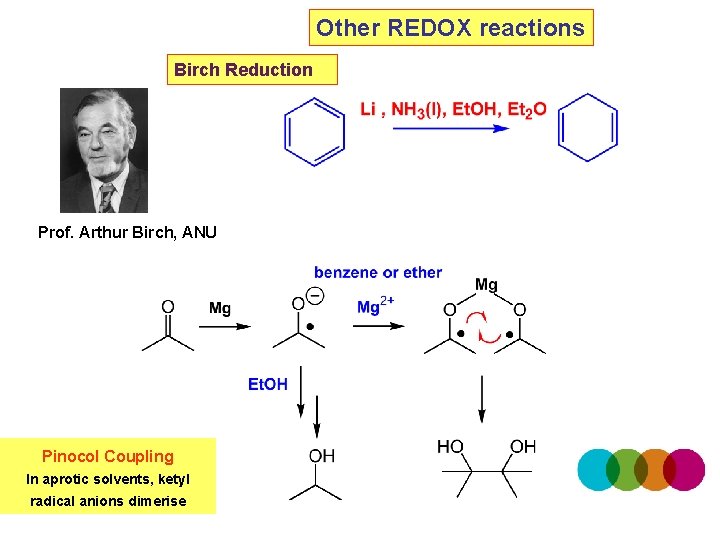
Other REDOX reactions Birch Reduction Prof. Arthur Birch, ANU Pinocol Coupling In aprotic solvents, ketyl radical anions dimerise
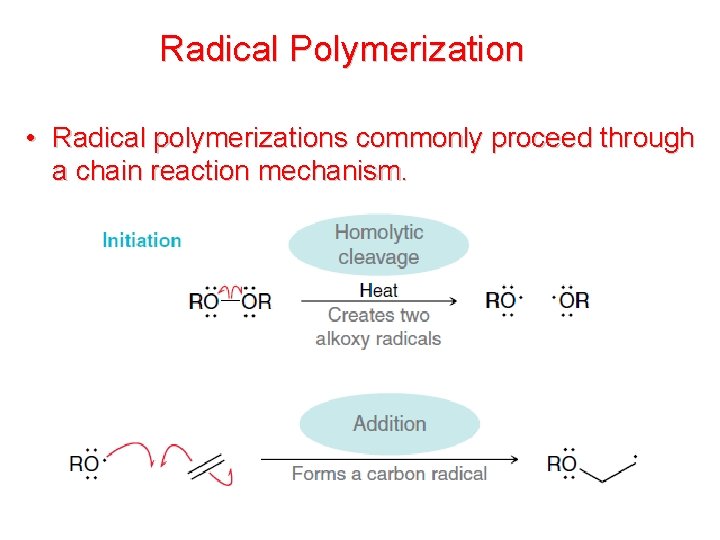
Radical Polymerization • Radical polymerizations commonly proceed through a chain reaction mechanism.
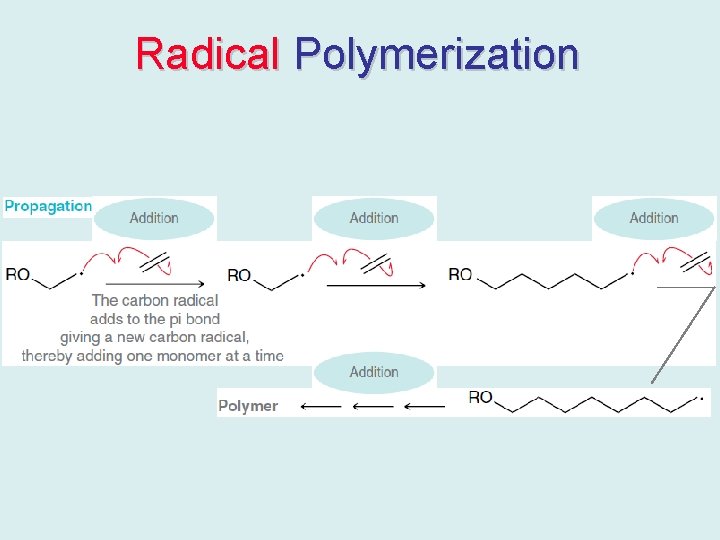
Radical Polymerization
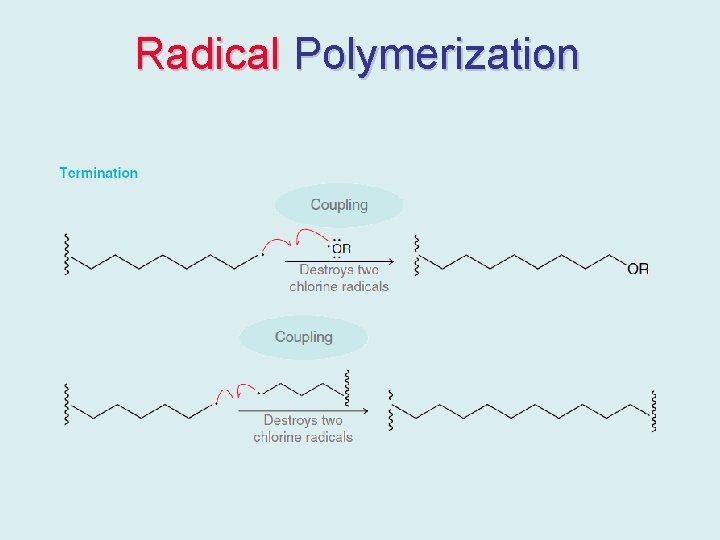
Radical Polymerization
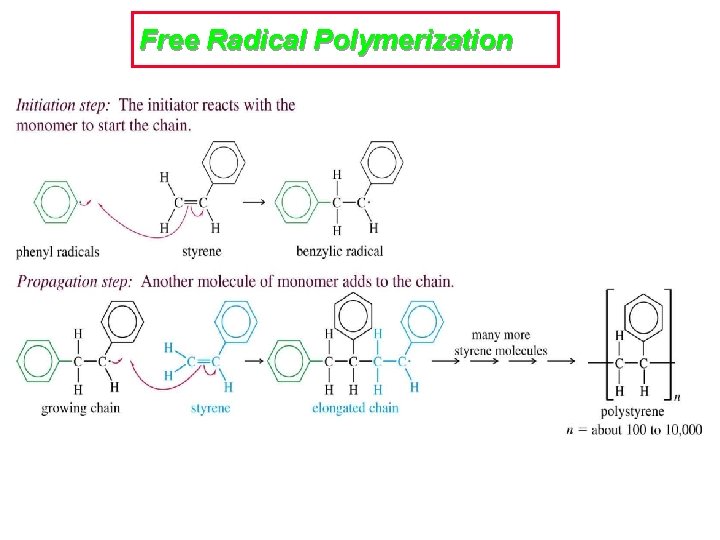
Free Radical Polymerization

• Reaction of Chloroalkanes with Ozone • The use of chlorinated organic compounds is being curtailed for two main reasons. • Some of the compounds are potentially carcinogenic (cancer forming) and they also have the potential to lower • the concentration of ozone in the ozone layer. • Ozone is continually being formed and depleted in the ozone layer between about 120 km and 50 km above the surface of the Earth. • The strong double bond in oxygen is broken by high energy • Ultraviolet light from the Sun to form radicals. One oxygen radical can then react with an oxygen • molecule to form ozone.

• Overall, the rate of production of ozone is equal to the rate of ozone destruction – this is known as • a steady state. However, compounds containing C—Cl bonds can alter this steady state. • Chlorinated hydrocarbons tend to be rather unreactive until they reach the stratosphere, where the ultraviolet light causes the C—Cl bond to break homolytically. • The chlorine radicals can then react with ozone molecules by a chain reaction. This causes a substantial lowering of the ozone • concentration, resulting in ‘holes’ in the ozone layer, particularly over the polar regions.

THE END
- Slides: 23