Free Radical Substitution Homolytic Fission Methane Chloromethane H

Free Radical Substitution Homolytic Fission
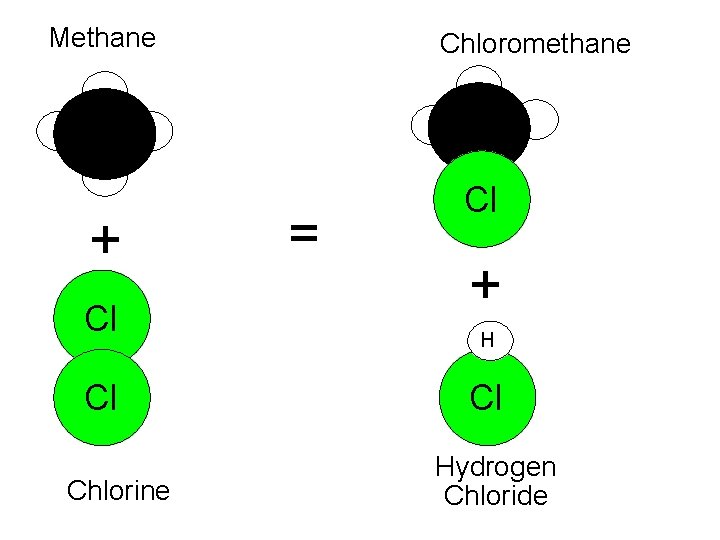
Methane Chloromethane H + Cl Cl Chlorine = Cl + H Cl Hydrogen Chloride
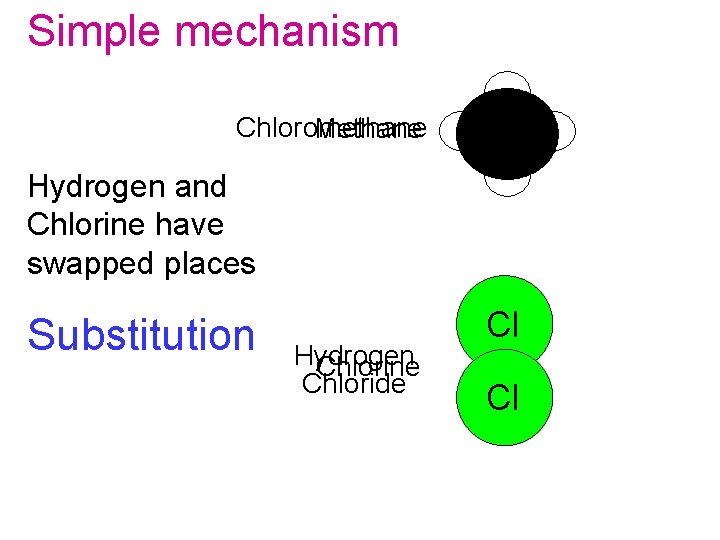
Simple mechanism Chloromethane Methane H Hydrogen and Chlorine have swapped places Substitution Hydrogen Chlorine Chloride Cl Cl

• How does this happen? • What causes it? • How do we know that we suggest is correct?

Stage 1 Initiation Getting Started
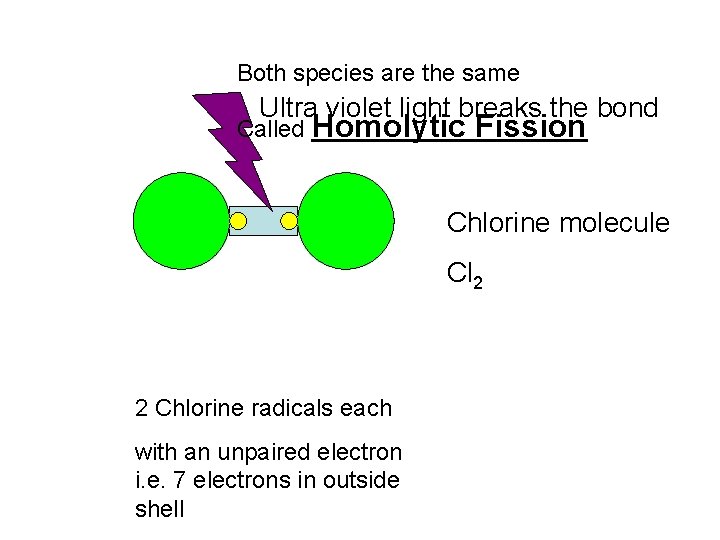
Both species are the same Ultra violet light breaks the bond Called Homolytic Fission Chlorine molecule Cl 2 2 Chlorine radicals each with an unpaired electron i. e. 7 electrons in outside shell

Stage 2 Propagation Keeping it going
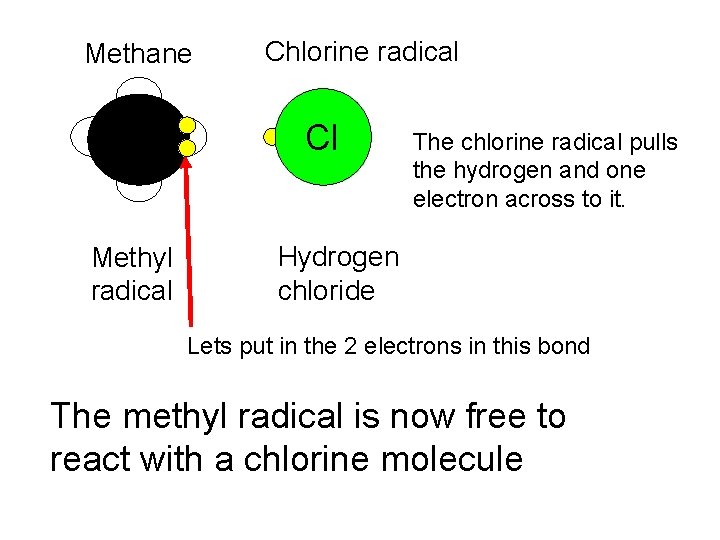
Methane H Methyl radical Chlorine radical Cl The chlorine radical pulls the hydrogen and one electron across to it. Hydrogen chloride Lets put in the 2 electrons in this bond The methyl radical is now free to react with a chlorine molecule
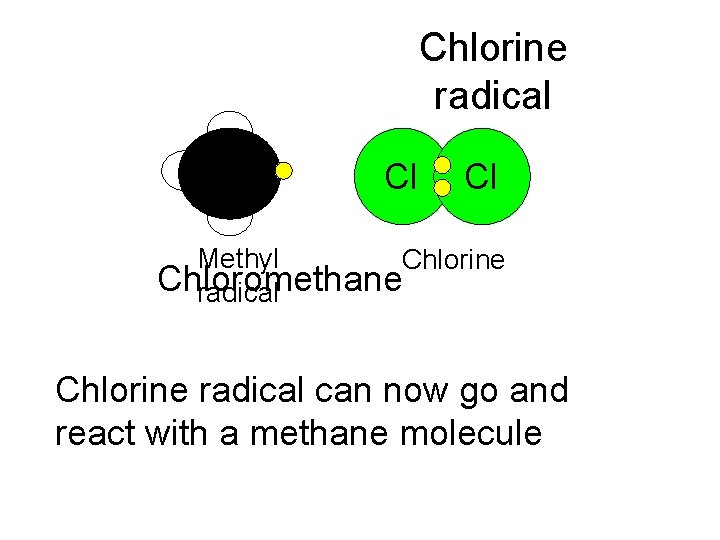
Chlorine radical Cl Cl Methyl Chlorine Chloromethane radical Chlorine radical can now go and react with a methane molecule
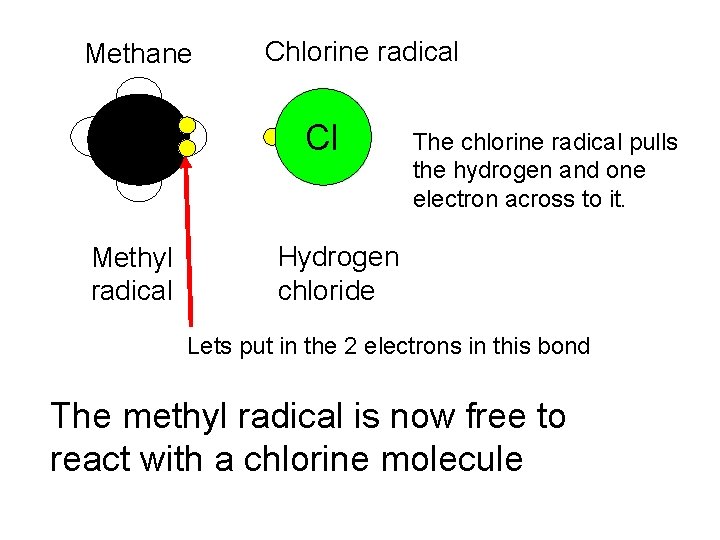
Methane H Methyl radical Chlorine radical Cl The chlorine radical pulls the hydrogen and one electron across to it. Hydrogen chloride Lets put in the 2 electrons in this bond The methyl radical is now free to react with a chlorine molecule
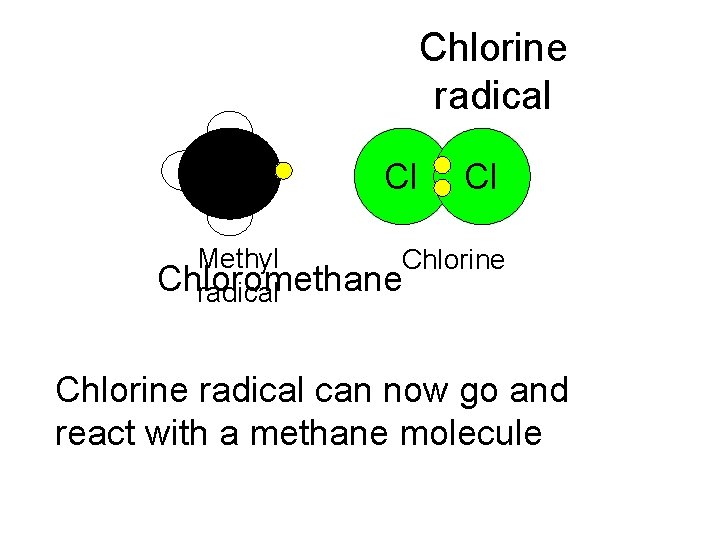
Chlorine radical Cl Cl Methyl Chlorine Chloromethane radical Chlorine radical can now go and react with a methane molecule

And on and on

Stage 3 Termination Grinding to a halt

• Three different ways this can happen
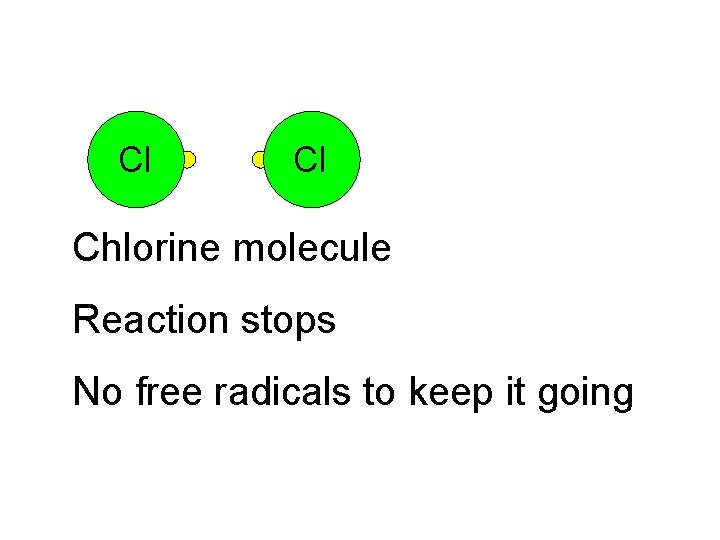
Cl Cl Chlorine molecule Chlorine radical Reaction stops No free radicals to keep it going

Cl Chlorine Methyl Chloromethane forms radical Reaction stops Because there are no free radicals to keep it going

Ethane Methyl stops Methyl Reaction because no free radicals radical to keep it going The formation of ethane proves that this is the mechanism Reaction speeded up by sources of free radicals such as tetraethyl lead.

End
- Slides: 18