Fre E coli A New Series of Escherichia





- Slides: 37

Fre. E coli™: A New Series of Escherichia coli Strains for Endotoxin-Free Production of Recombinant Proteins and Plasmid DNA Uwe Mamat Structural Biochemistry, Research Center Borstel, Leibniz-Center for Medicine and Biosciences, Borstel, Germany 1

Topics 1. Introduction 2. Features of the Fre. E coli™ system 3. Application: Protein expression 4. Application: Plasmid DNA production 2

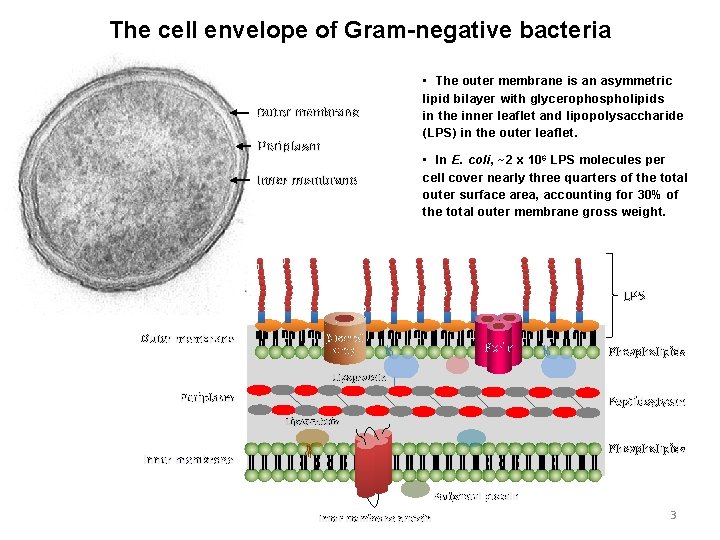
The cell envelope of Gram-negative bacteria Outer membrane Periplasm Inner membrane • The outer membrane is an asymmetric lipid bilayer with glycerophospholipids in the inner leaflet and lipopolysaccharide (LPS) in the outer leaflet. • In E. coli, ~2 x 10 6 LPS molecules per cell cover nearly three quarters of the total outer surface area, accounting for 30% of the total outer membrane gross weight. LPS Outer membrane b-Barrel Omp Porin Phospholipids Lipoprotein Periplasm Peptidoglycan Lipoprotein Phospholipids Inner membrane Peripheral protein Inner membrane protein 3

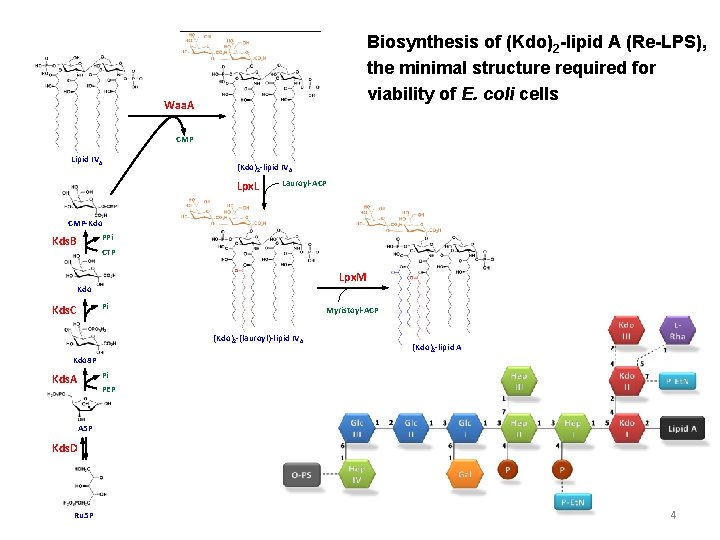
Biosynthesis of (Kdo)2 -lipid A (Re-LPS), the minimal structure required for viability of E. coli cells Waa. A CMP Lipid IVA (Kdo)2 -lipid IVA Lpx. L Lauroyl-ACP CMP-Kdo PPi Kds. B CTP Lpx. M Kdo Pi Kds. C Myristoyl-ACP (Kdo)2 -(lauroyl)-lipid IV A (Kdo)2 -lipid A Kdo 8 P Pi Kds. A PEP A 5 P Kds. D Ru 5 P 4

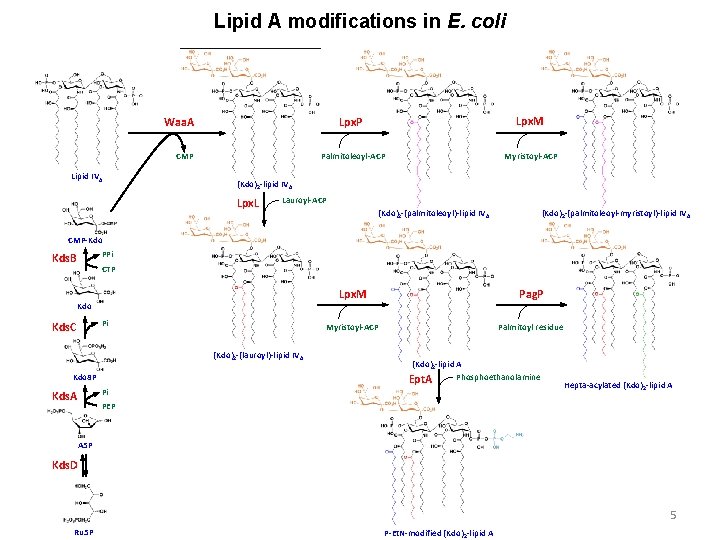
Lipid A modifications in E. coli Lpx. P Lpx. M Palmitoleoyl-ACP Myristoyl-ACP Waa. A CMP Lipid IVA (Kdo)2 -lipid IVA Lpx. L Lauroyl-ACP (Kdo)2 -(palmitoleoyl)-lipid IV A (Kdo)2 -(palmitoleoyl-myristoyl)-lipid IV A CMP-Kdo PPi Kds. B CTP Kdo Pi Kds. C (Kdo)2 -(lauroyl)-lipid IV A Kdo 8 P Pi Kds. A Lpx. M Pag. P Myristoyl-ACP Palmitoyl residue (Kdo)2 -lipid A Phosphoethanolamine Ept. A Hepta-acylated (Kdo)2 -lipid A PEP A 5 P Kds. D 5 Ru 5 P P-Et. N-modified (Kdo)2 -lipid A

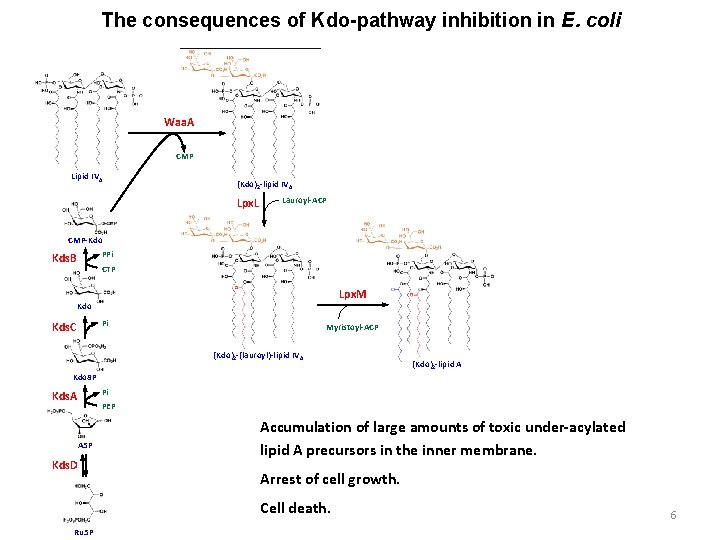
The consequences of Kdo-pathway inhibition in E. coli Waa. A CMP Lipid IVA (Kdo)2 -lipid IVA Lpx. L Lauroyl-ACP CMP-Kdo PPi Kds. B CTP Lpx. M Kdo Pi Kds. C Myristoyl-ACP (Kdo)2 -(lauroyl)-lipid IV A (Kdo)2 -lipid A Kdo 8 P Pi Kds. A PEP A 5 P Kds. D Accumulation of large amounts of toxic under-acylated lipid A precursors in the inner membrane. Arrest of cell growth. Cell death. Ru 5 P 6

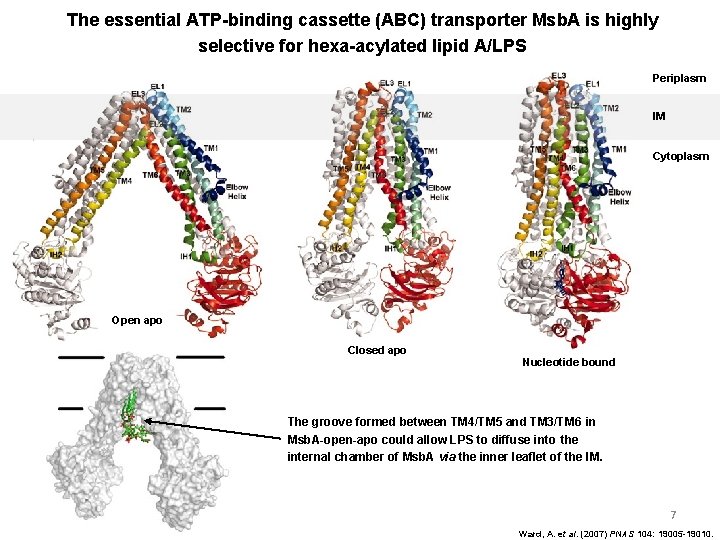
The essential ATP-binding cassette (ABC) transporter Msb. A is highly selective for hexa-acylated lipid A/LPS Periplasm IM Cytoplasm Open apo Closed apo Nucleotide bound The groove formed between TM 4/TM 5 and TM 3/TM 6 in Msb. A-open-apo could allow LPS to diffuse into the internal chamber of Msb. A via the inner leaflet of the IM. 7 Ward, A. et al. (2007) PNAS 104: 19005 -19010.

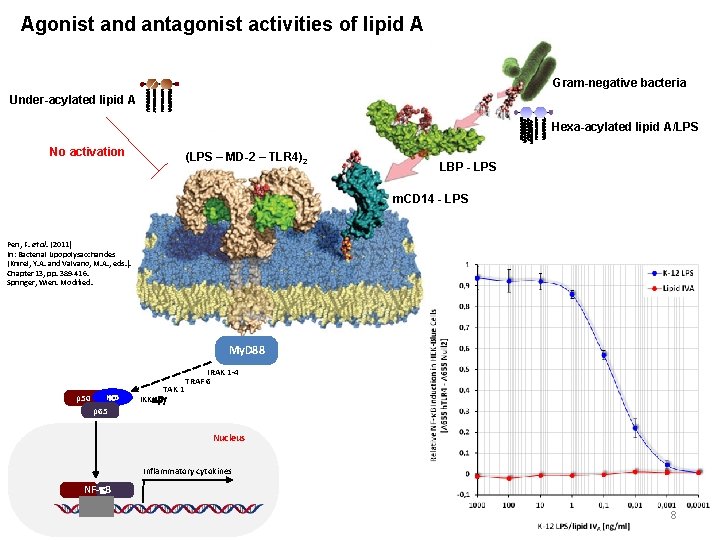
Agonist and antagonist activities of lipid A Gram-negative bacteria Under-acylated lipid A Hexa-acylated lipid A/LPS No activation (LPS – MD-2 – TLR 4)2 LBP - LPS m. CD 14 - LPS Peri, F. et al. (2011) In: Bacterial Lipopolysaccharides (Knirel, Y. A. and Valvano, M. A. , eds. ). Chapter 13, pp. 389 -416. Springer, Wien. Modified. My. D 88 p 50 Ik. B p 65 TAK 1 IKKabg IRAK 1 -4 TRAF 6 Nucleus Inflammatory cytokines NF-k. B 8

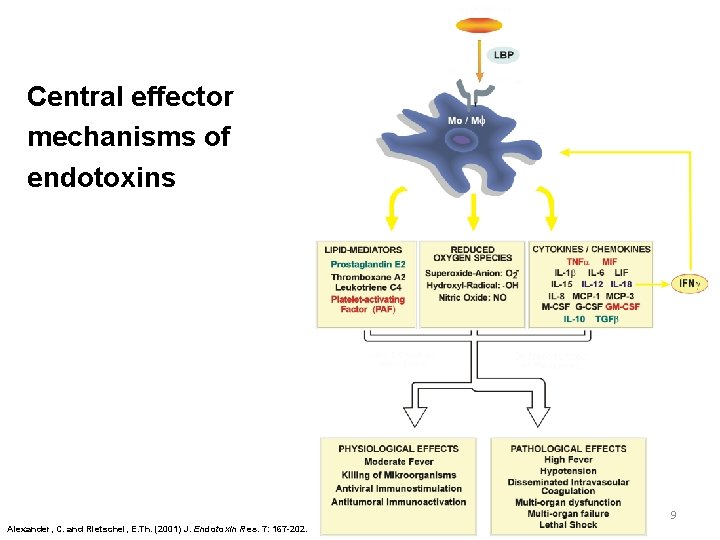
Central effector mechanisms of endotoxins 9 Alexander, C. and Rietschel, E. Th. (2001) J. Endotoxin Res. 7: 167 -202.

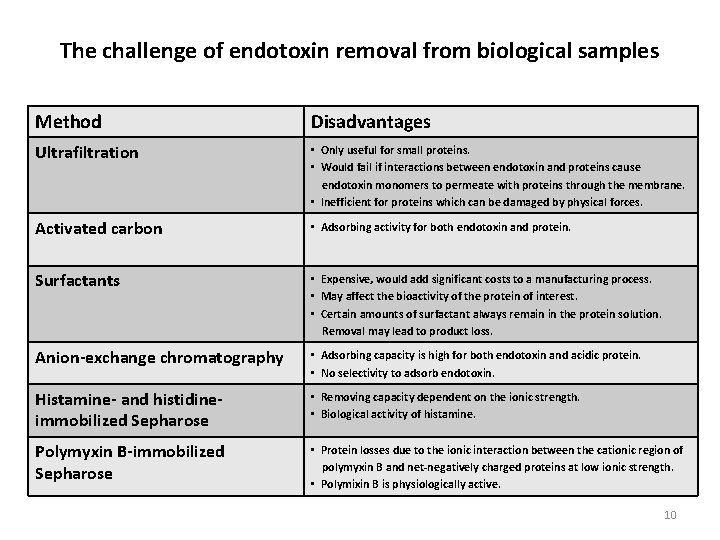
The challenge of endotoxin removal from biological samples Method Disadvantages Ultrafiltration • Only useful for small proteins. • Would fail if interactions between endotoxin and proteins cause endotoxin monomers to permeate with proteins through the membrane. • Inefficient for proteins which can be damaged by physical forces. Activated carbon • Adsorbing activity for both endotoxin and protein. Surfactants • Expensive, would add significant costs to a manufacturing process. • May affect the bioactivity of the protein of interest. • Certain amounts of surfactant always remain in the protein solution. Removal may lead to product loss. Anion-exchange chromatography • Adsorbing capacity is high for both endotoxin and acidic protein. • No selectivity to adsorb endotoxin. Histamine- and histidineimmobilized Sepharose • Removing capacity dependent on the ionic strength. • Biological activity of histamine. Polymyxin B-immobilized Sepharose • Protein losses due to the ionic interaction between the cationic region of polymyxin B and net-negatively charged proteins at low ionic strength. • Polymixin B is physiologically active. 10

Topics 1. Introduction 2. Features of the Fre. E coli™ system 3. Application: Protein expression 4. Application: Plasmid DNA production 11

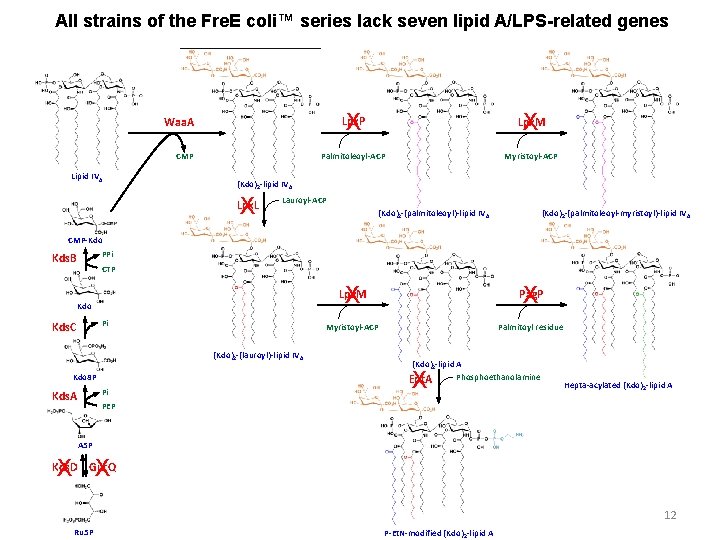
All strains of the Fre. E coli™ series lack seven lipid A/LPS-related genes Lpx. P X Lpx. M Palmitoleoyl-ACP Myristoyl-ACP Waa. A CMP Lipid IVA X (Kdo)2 -lipid IVA X Lpx. L Lauroyl-ACP (Kdo)2 -(palmitoleoyl)-lipid IV A (Kdo)2 -(palmitoleoyl-myristoyl)-lipid IV A CMP-Kdo PPi Kds. B CTP Kdo Pi Kds. C (Kdo)2 -(lauroyl)-lipid IV A Kdo 8 P Pi Kds. A Lpx. M X Pag. P X Myristoyl-ACP Palmitoyl residue (Kdo)2 -lipid A Phosphoethanolamine Ept. A X Hepta-acylated (Kdo)2 -lipid A PEP A 5 P X X Kds. D Gut. Q 12 Ru 5 P P-Et. N-modified (Kdo)2 -lipid A

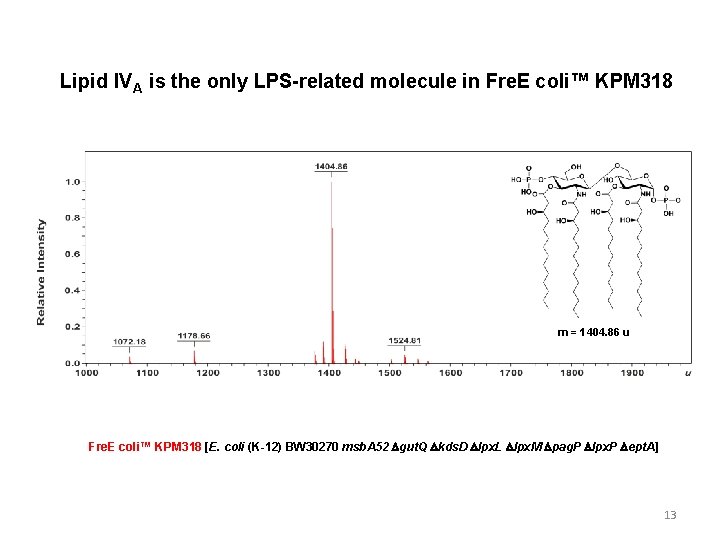
Lipid IVA is the only LPS-related molecule in Fre. E coli™ KPM 318 m = 1404. 86 u Fre. E coli™ KPM 318 [E. coli (K-12) BW 30270 msb. A 52 Dgut. Q Dkds. D Dlpx. L Dlpx. M Dpag. P Dlpx. P Dept. A] 13

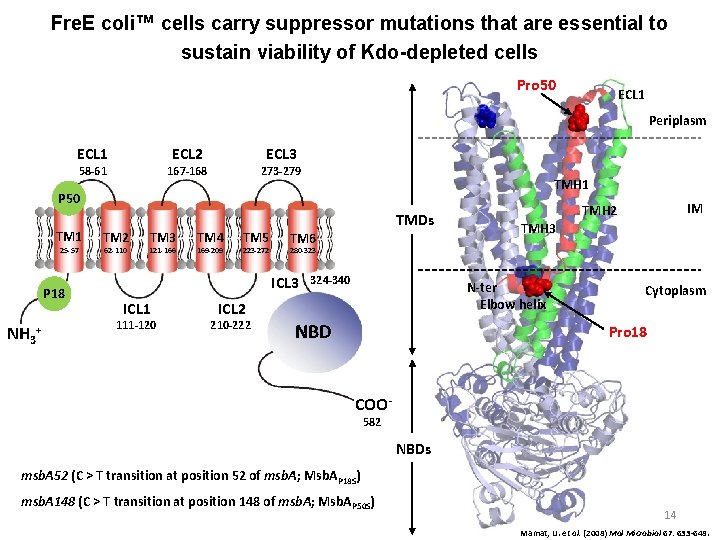
Fre. E coli™ cells carry suppressor mutations that are essential to sustain viability of Kdo-depleted cells Pro 50 ECL 1 Periplasm ECL 1 ECL 2 58 -61 ECL 3 167 -168 273 -279 TMH 1 P 50 TM 1 25 -57 P 18 NH 3+ TM 2 62 -110 TM 3 121 -166 TM 4 169 -209 TMDs TM 5 223 -272 TM 6 111 -120 324 -340 N-ter Elbow helix ICL 2 210 -222 TMH 3 280 -323 ICL 1 IM TMH 2 NBD Cytoplasm Pro 18 COO 582 NBDs msb. A 52 (C > T transition at position 52 of msb. A; Msb. AP 18 S) msb. A 148 (C > T transition at position 148 of msb. A; Msb. AP 50 S) 14 Mamat, U. et al. (2008) Mol Microbiol 67: 633 -648.

The Fre. E coli™ cells are capable of • increasing the rate of lipid IVA transport to supply enough lipid for biogenesis of the outer membrane and • removing lipid IVA from the inner membrane to attenuate toxic side effects caused by lipid IVA accumulation. 15

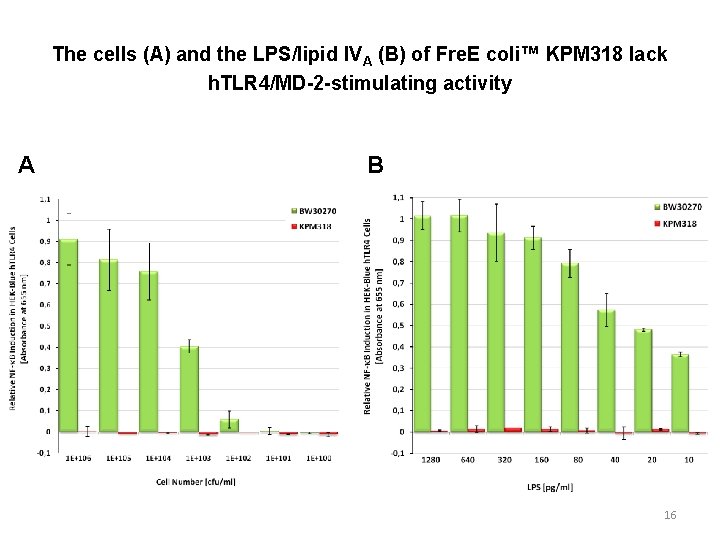
The cells (A) and the LPS/lipid IVA (B) of Fre. E coli™ KPM 318 lack h. TLR 4/MD-2 -stimulating activity A B 16

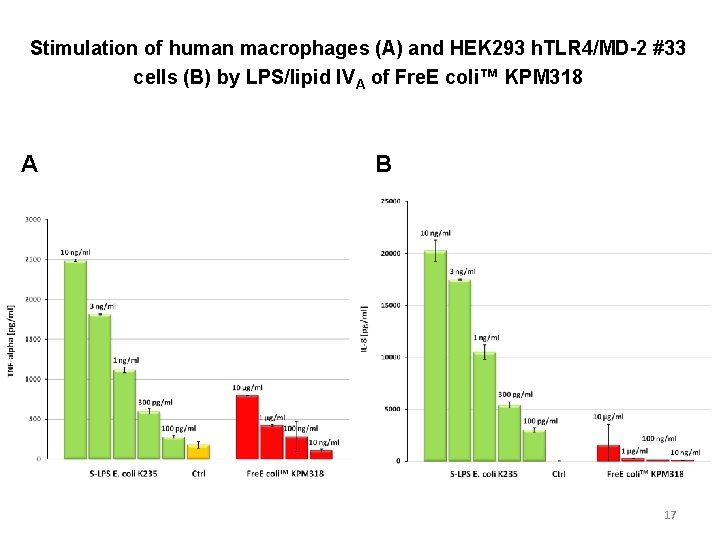
Stimulation of human macrophages (A) and HEK 293 h. TLR 4/MD-2 #33 cells (B) by LPS/lipid IVA of Fre. E coli™ KPM 318 A B 17

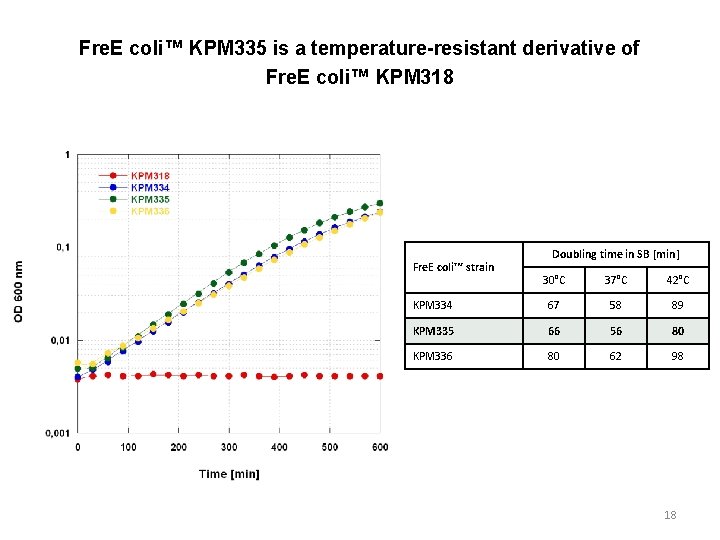
Fre. E coli™ KPM 335 is a temperature-resistant derivative of Fre. E coli™ KPM 318 Fre. E coli™ strain Doubling time in SB [min] 30°C 37°C 42°C KPM 334 67 58 89 KPM 335 66 56 80 KPM 336 80 62 98 18

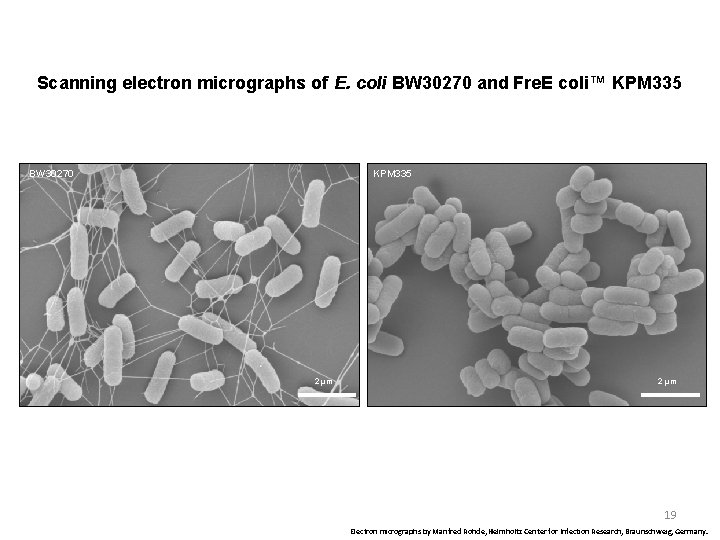
Scanning electron micrographs of E. coli BW 30270 and Fre. E coli™ KPM 335 BW 30270 KPM 335 2 µm 19 Electron micrographs by Manfred Rohde, Helmholtz Center for Infection Research, Braunschweig, Germany.

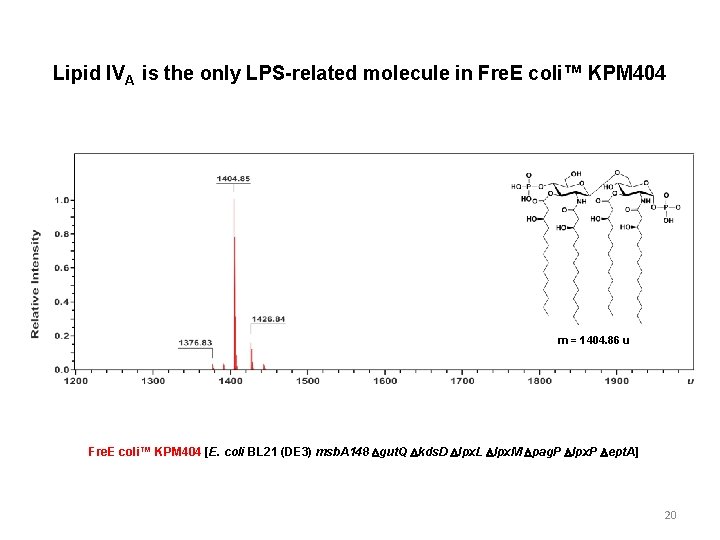
Lipid IVA is the only LPS-related molecule in Fre. E coli™ KPM 404 m = 1404. 86 u Fre. E coli™ KPM 404 [E. coli BL 21 (DE 3) msb. A 148 Dgut. Q Dkds. D Dlpx. L Dlpx. M Dpag. P Dlpx. P Dept. A] 20

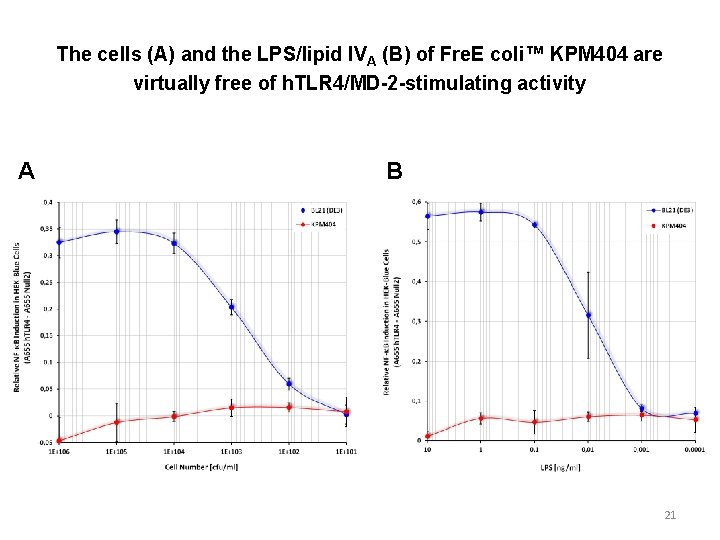
The cells (A) and the LPS/lipid IVA (B) of Fre. E coli™ KPM 404 are virtually free of h. TLR 4/MD-2 -stimulating activity A B 21

Topics 1. Introduction 2. Features of the Fre. E coli™ system 3. Application: Protein expression 4. Application: Plasmid DNA production 22

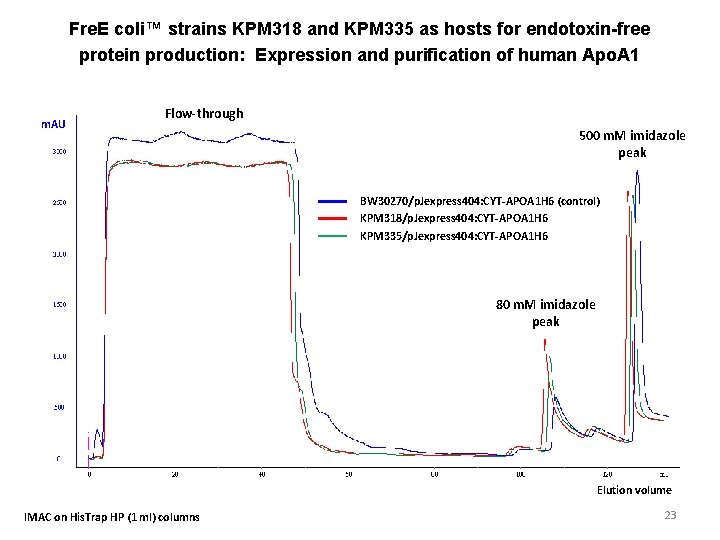
Fre. E coli™ strains KPM 318 and KPM 335 as hosts for endotoxin-free protein production: Expression and purification of human Apo. A 1 m. AU Flow-through 500 m. M imidazole peak BW 30270/p. Jexpress 404: CYT-APOA 1 H 6 (control) KPM 318/p. Jexpress 404: CYT-APOA 1 H 6 KPM 335/p. Jexpress 404: CYT-APOA 1 H 6 80 m. M imidazole peak Elution volume IMAC on His. Trap HP (1 ml) columns 23

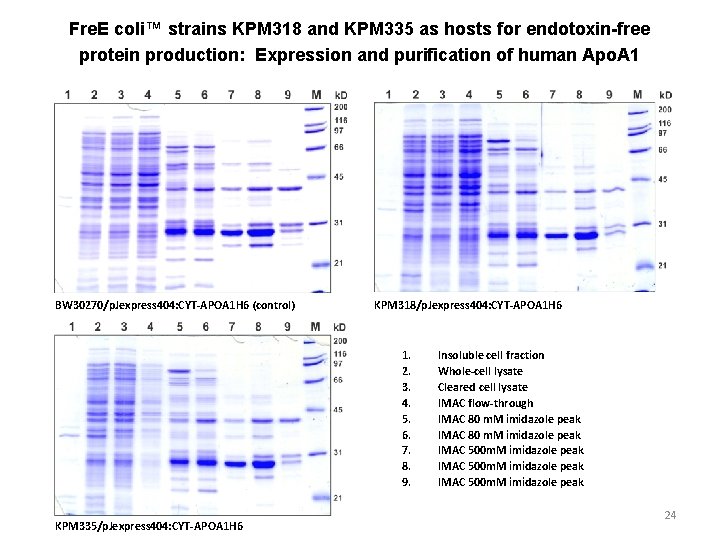
Fre. E coli™ strains KPM 318 and KPM 335 as hosts for endotoxin-free protein production: Expression and purification of human Apo. A 1 BW 30270/p. Jexpress 404: CYT-APOA 1 H 6 (control) KPM 318/p. Jexpress 404: CYT-APOA 1 H 6 1. 2. 3. 4. 5. 6. 7. 8. 9. KPM 335/p. Jexpress 404: CYT-APOA 1 H 6 Insoluble cell fraction Whole-cell lysate Cleared cell lysate IMAC flow-through IMAC 80 m. M imidazole peak IMAC 500 m. M imidazole peak 24

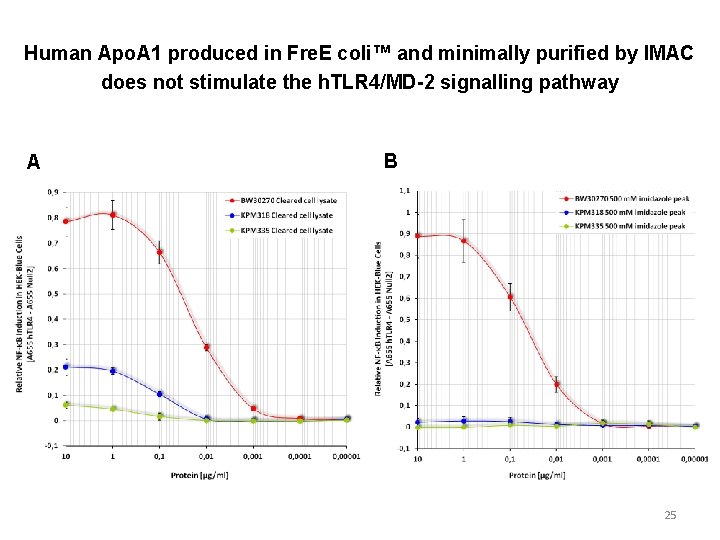
Human Apo. A 1 produced in Fre. E coli™ and minimally purified by IMAC does not stimulate the h. TLR 4/MD-2 signalling pathway A B 25

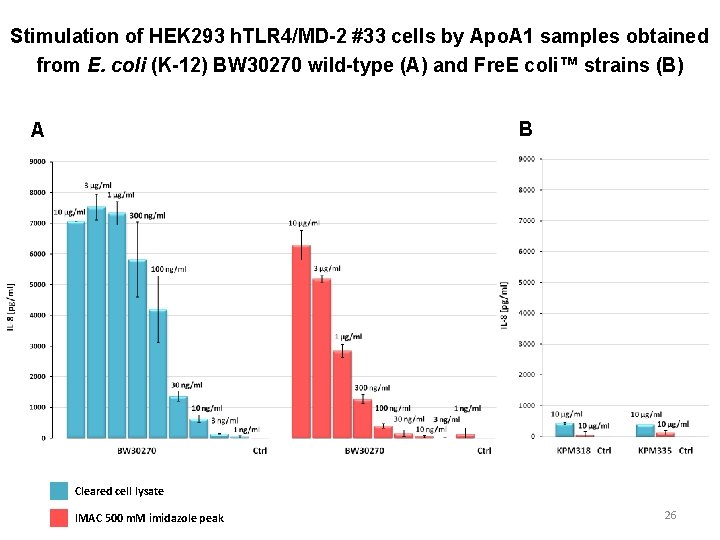
Stimulation of HEK 293 h. TLR 4/MD-2 #33 cells by Apo. A 1 samples obtained from E. coli (K-12) BW 30270 wild-type (A) and Fre. E coli™ strains (B) B A Cleared cell lysate IMAC 500 m. M imidazole peak 26

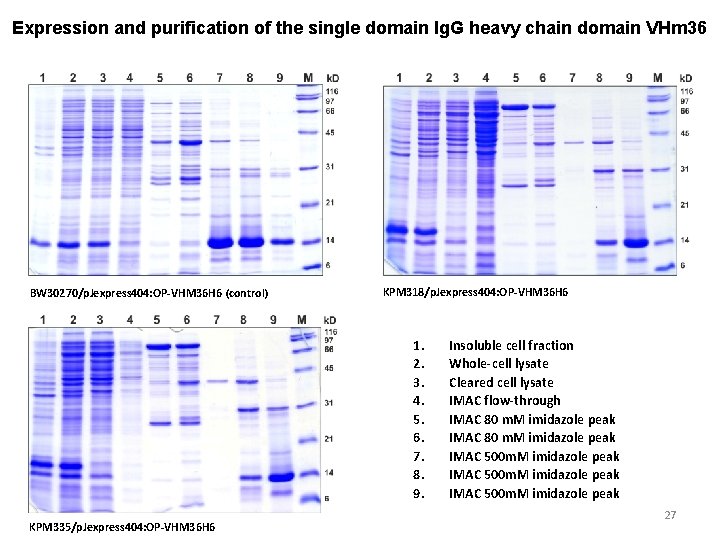
Expression and purification of the single domain Ig. G heavy chain domain VHm 36 BW 30270/p. Jexpress 404: OP-VHM 36 H 6 (control) KPM 318/p. Jexpress 404: OP-VHM 36 H 6 1. 2. 3. 4. 5. 6. 7. 8. 9. KPM 335/p. Jexpress 404: OP-VHM 36 H 6 Insoluble cell fraction Whole-cell lysate Cleared cell lysate IMAC flow-through IMAC 80 m. M imidazole peak IMAC 500 m. M imidazole peak 27

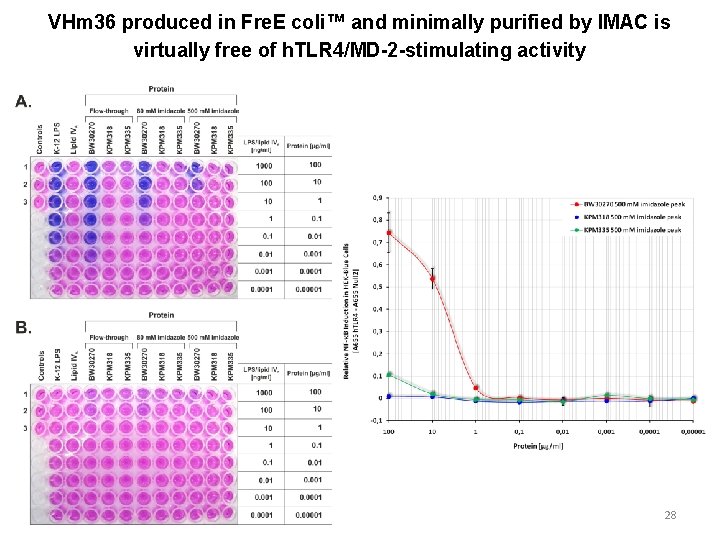
VHm 36 produced in Fre. E coli™ and minimally purified by IMAC is virtually free of h. TLR 4/MD-2 -stimulating activity 28

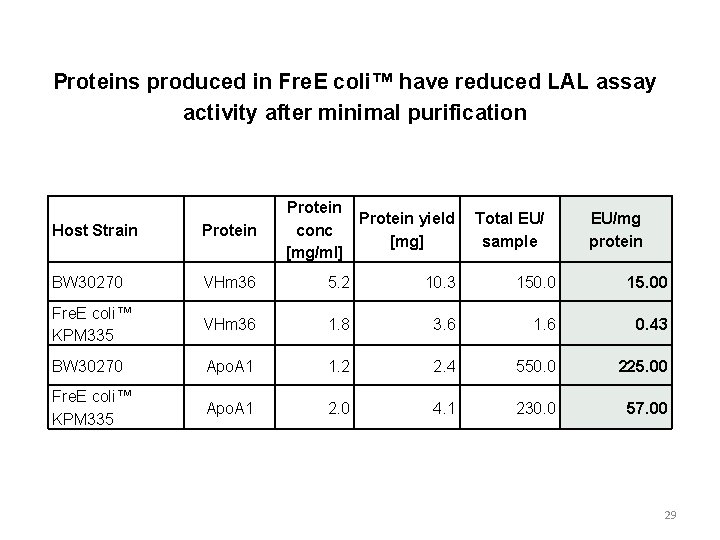
Proteins produced in Fre. E coli™ have reduced LAL assay activity after minimal purification Protein yield conc [mg] [mg/ml] Total EU/ sample EU/mg protein Host Strain Protein BW 30270 VHm 36 5. 2 10. 3 150. 0 15. 00 Fre. E coli™ KPM 335 VHm 36 1. 8 3. 6 1. 6 0. 43 BW 30270 Apo. A 1 1. 2 2. 4 550. 0 225. 00 Fre. E coli™ KPM 335 Apo. A 1 2. 0 4. 1 230. 0 57. 00 29

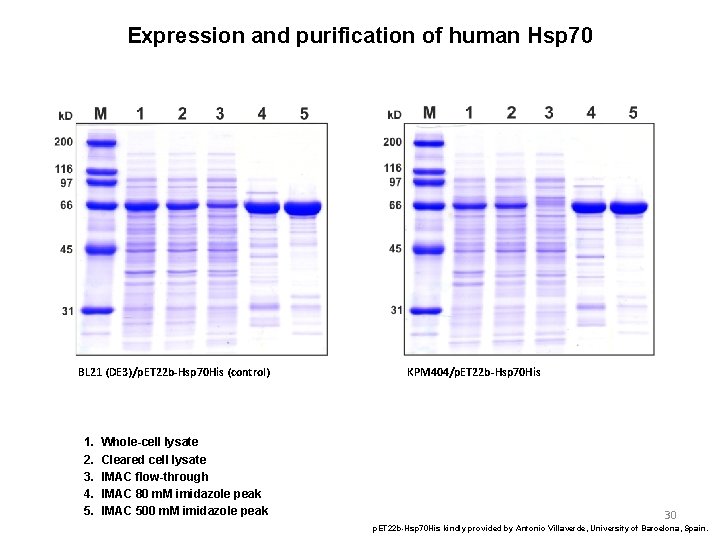
Expression and purification of human Hsp 70 BL 21 (DE 3)/p. ET 22 b-Hsp 70 His (control) 1. 2. 3. 4. 5. Whole-cell lysate Cleared cell lysate IMAC flow-through IMAC 80 m. M imidazole peak IMAC 500 m. M imidazole peak KPM 404/p. ET 22 b-Hsp 70 His 30 p. ET 22 b-Hsp 70 His kindly provided by Antonio Villaverde, University of Barcelona, Spain.

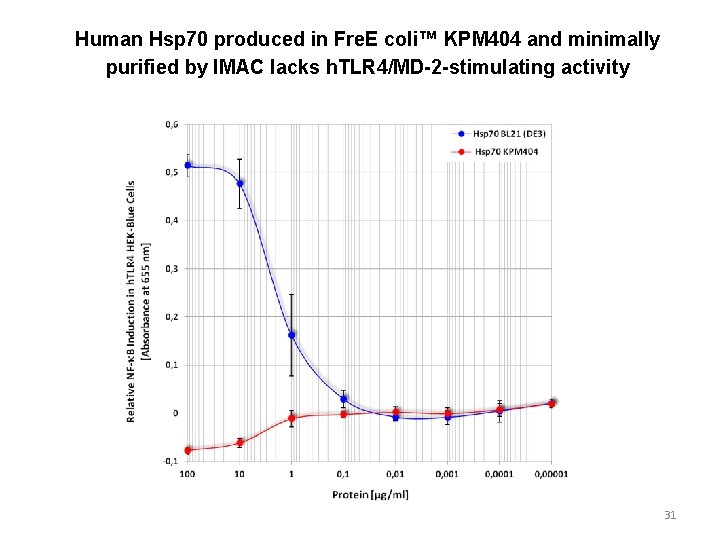
Human Hsp 70 produced in Fre. E coli™ KPM 404 and minimally purified by IMAC lacks h. TLR 4/MD-2 -stimulating activity 31

Topics 1. Introduction 2. Features of the Fre. E coli™ system 3. Application: Protein expression 4. Application: Plasmid DNA production 32

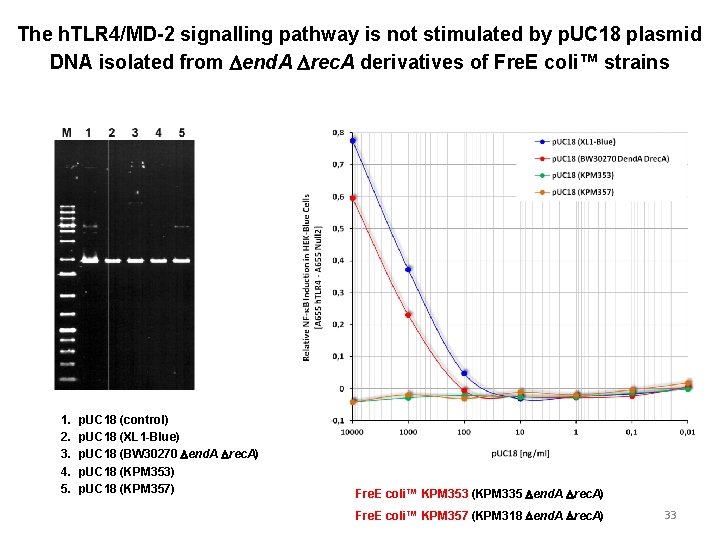
The h. TLR 4/MD-2 signalling pathway is not stimulated by p. UC 18 plasmid DNA isolated from Dend. A Drec. A derivatives of Fre. E coli™ strains 1. 2. 3. 4. 5. p. UC 18 (control) p. UC 18 (XL 1 -Blue) p. UC 18 (BW 30270 Dend. A Drec. A) p. UC 18 (KPM 353) p. UC 18 (KPM 357) Fre. E coli™ KPM 353 (KPM 335 Dend. A Drec. A) Fre. E coli™ KPM 357 (KPM 318 Dend. A Drec. A) 33

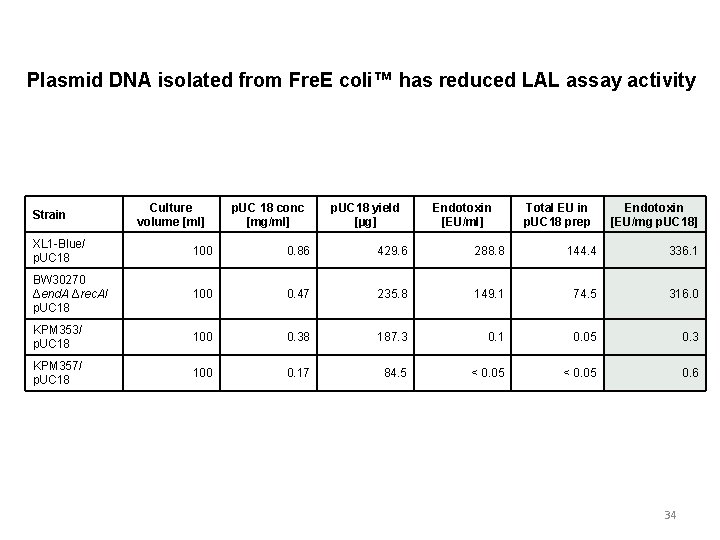
Plasmid DNA isolated from Fre. E coli™ has reduced LAL assay activity Strain Culture volume [ml] p. UC 18 conc [mg/ml] p. UC 18 yield [µg] Endotoxin [EU/ml] Total EU in p. UC 18 prep Endotoxin [EU/mg p. UC 18] XL 1 -Blue/ p. UC 18 100 0. 86 429. 6 288. 8 144. 4 336. 1 BW 30270 Δend. A Δrec. A/ p. UC 18 100 0. 47 235. 8 149. 1 74. 5 316. 0 KPM 353/ p. UC 18 100 0. 38 187. 3 0. 1 0. 05 0. 3 KPM 357/ p. UC 18 100 0. 17 84. 5 < 0. 05 0. 6 34

Summary (I) • The Fre. E coli™ strains are viable despite predominantly expressing the endotoxically inactive LPS precursor lipid IVA. • Due to deletions of seven LPS/lipid A-related genes in the genomes of the Fre. E coli™ suppressor strains, the mutants cannot easily regain the ability to synthesize normal LPS or endotoxically active lipid IVA derivatives. • Several example proteins have been expressed with the Fre. E coli™ system. Pre-purification yields for Fre. E coli™ are similar to the yield from wild-type comparator strains. • Crude and minimally purified proteins produced in Fre. E coli™ do not activate the endotoxin signaling pathway (human TLR 4/MD-2 signaling) meaning less time and effort is needed to purify proteins before screening in cell based assays. 35

Summary (II) • The outer membrane component of Fre. E coli™, lipid IVA, appears easier to remove from proteins than the traditional outer membrane component LPS and may provide substantial benefits for proteins where LPS is difficult to remove. • DNA has been produced in Dend. A Drec. A derivatives of Fre. E coli™ strains. • DNA produced in Fre. E coli™ does not activate the endotoxin signaling pathway (human TLR 4/MD-2 signaling) and has substantially lower activity in the LAL assay after purification using the Qiagen Maxi. Prep system. • The outer membrane component of Fre. E coli™, lipid IVA, appears easier to remove from DNA than the traditional outer membrane component LPS allowing for higher quality DNA after purification. This may provide for greater transfection efficiency. 36

Acknowledgements Research Corporation Technologies, Tucson, USA David Bramhill Chad Souvignier Research Center Borstel, Germany Kathleen Wilke Michael Weinkauf Kerstin Viertmann Brigitte Kunz Buko Lindner Sabrina Groth Andra Schromm University of Michigan, College of Pharmacy, Ann Arbor, USA Timothy C. Meredith Paul Kirchhoff Parag Aggarwal Ronald W. Woodard Helmholtz Center for Infection Research, Braunschweig, Germany, Ina Schleicher Manfred Rohde 37