FRCR I Basic Physics Nick Harding Clinical Scientist

FRCR I – Basic Physics Nick Harding Clinical Scientist Radiotherapy Department Castle Hill Hospital Hull University Teaching Hospitals NHS Trust email: nicholas. harding@hey. nhs. uk

FRCR LECTURES § Lecture I – 10/09/2020: § Structure of Matter: the Atom and the Nucleus § Lecture II – 10/09/2020: § Radioactivity § Lecture III – 14/09/2020: § Interactions of EM Radiation with Matter § Lecture IV – 14/09/2020: § Interactions of Electrons with Matter

Learning Outcomes Describe: • structure and properties of matter; • the phenomena of radioactivity and magnetism; • the nature of ionising radiation; • their interactions with matter;

FRCR LECTURES § All slides available at: http: //www. hullrad. org. uk/ Main Menu -> Training -> FRCR Physics

BIBLIOGRAPHY § Radiological Physics § P. Dendy, B. Heaton – Physics for Radiologists § Medical Imaging J. Bushberg et al – The Essential Physics of Medical Imaging § S. Webb – The Physics of Medical Imaging § P. Allisy-Roberts, J. Williams – Farr’s Physics for Medical Imaging § § Radiotherapy § F Khan – The Physics of Radiation Physics

Atoms & Nuclei

WHAT IS AN ATOM? § Definition: § Άτομο {átomo} – something that cannot be divided any further § The smallest division of an element in which the physical and chemical properties of the element are maintained
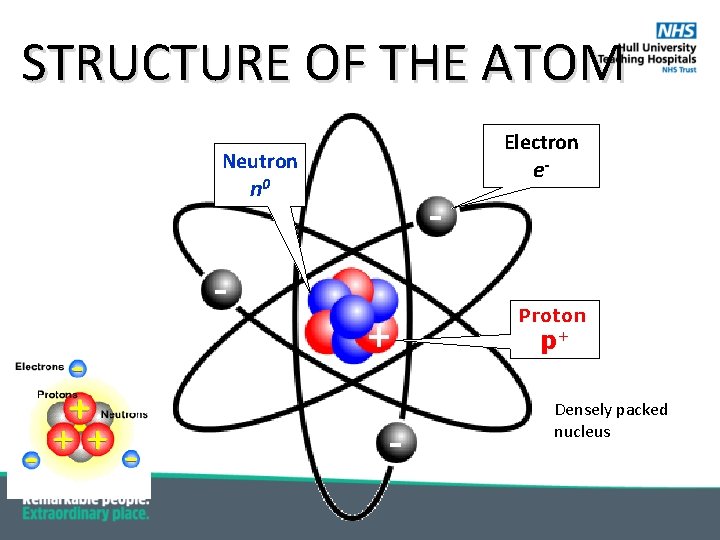
STRUCTURE OF THE ATOM Electron Neutron - n 0 + - e- Proton p+ Densely packed nucleus

SIZE OF THE ATOM § Radius of an atom is: § 10 -10 m § Radius of the nucleus is: § 10 -14 m § Thus, size of atom is 104=10, 000 times more than that of nucleus § Football vs 2 km
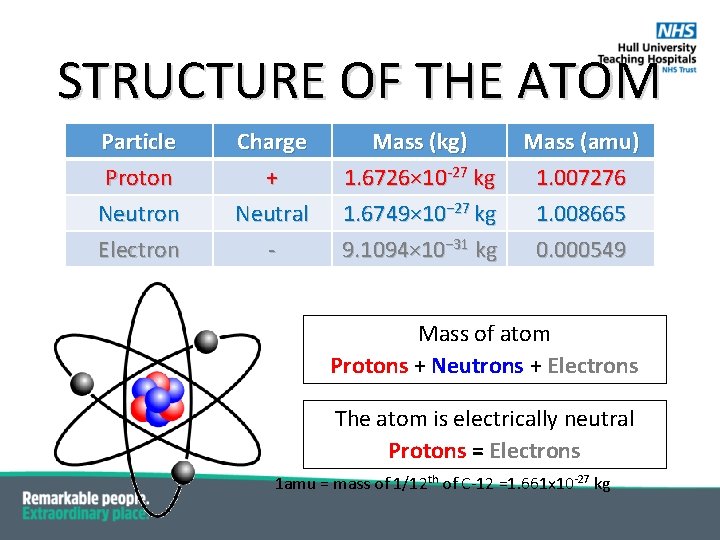
STRUCTURE OF THE ATOM Particle Proton Neutron Electron Charge + Neutral - Mass (kg) 1. 6726× 10 -27 kg 1. 6749× 10− 27 kg 9. 1094× 10− 31 kg Mass (amu) 1. 007276 1. 008665 0. 000549 Mass of atom Protons + Neutrons + Electrons The atom is electrically neutral Protons = Electrons 1 amu = mass of 1/12 th of C-12 =1. 661 x 10 -27 kg

NUCLEAR FORCES § Two main forces in the nucleus: § Coulombic (electrical) forces – between protons § Repulsive § Strong nuclear forces – between all nucleons § Attractive § Strong forces operate over short nuclear distances
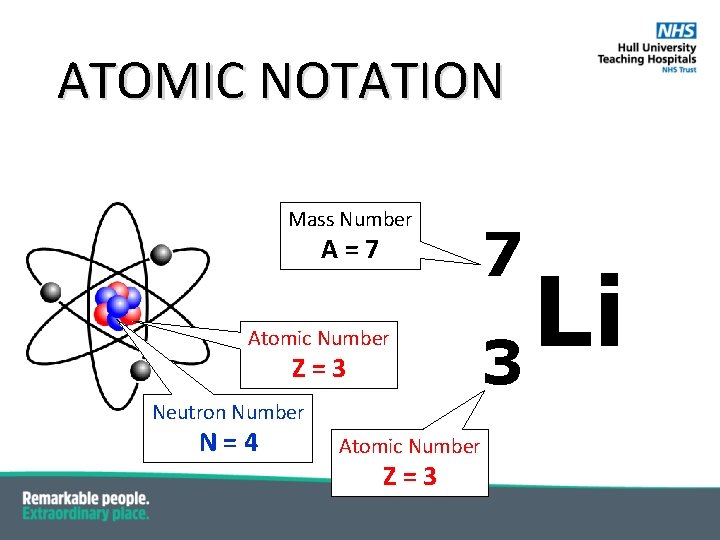
ATOMIC NOTATION Mass Number Α=7 Atomic Number Z=3 7 Li 3 Neutron Number Ν=4 Atomic Number Z=3
The Periodic Table 7 Li 3

ELEMENTS §Elements are groups of atoms with the same § number of protons (Z) § physical properties § e. g. density, melting and boiling point, electrical conductivity § chemical properties § e. g. reactions with water, oxygen, acids §There are more than 120 chemical ELEMENTS § 92 naturally occurring
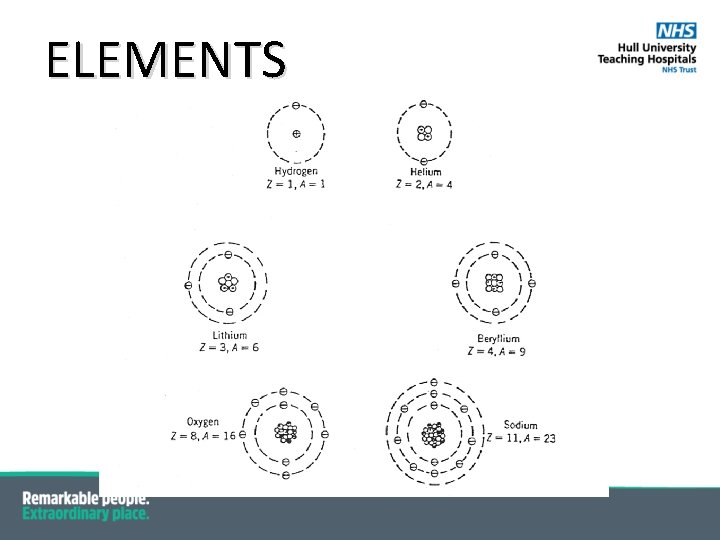
ELEMENTS

ISOTOPES § Isoto. Pes are atoms of the same element but with different mass number (A) § same atomic number (Z) i. e. same number of Protons § different neutron number § they have the same physical and chemical properties § but different nuclear properties
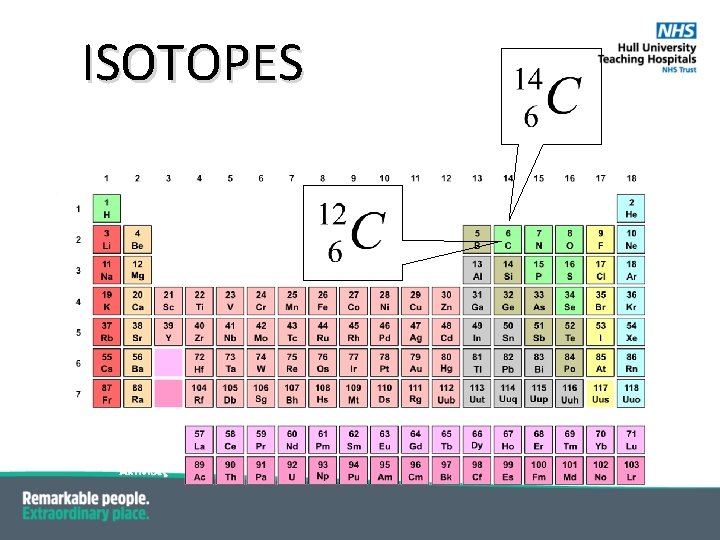
ISOTOPES

ISOBARS / ISOTONES / ISOMERS q. Nuclides with the same mass number (A) are called ISOBARS eg. Mo-99 and Tc-99 q. Nuclides with the same number of Neutrons (A – Z) are called ISOTONES eg. I-131 (Z=53) and Xe-132(Z=54) A-Z = (131 -53) = (132 – 54) = 78 q. Nuclides with the same atomic (Z) and mass numbers (A) but different nuclear Energy states are called ISOMERS eg. Tc-99 and Tc-99 m

Electron Orbitals

BOHR’S ATOMIC MODEL § Electrons orbit around the nucleus at fixed distances; § Each electron in a shell which has a discrete energy state; § Electron shells designated letters K, L, M… and quantum numbers 1, 2, 3… respectively. § Max number of electrons in each shell is 2 n 2 where n is the quantum number so 2 x 12, 2 x 22= 8, 2 x 32 = 18 and so on; § Outer shell is referred to as the valence shell; The Bohr model (1913)
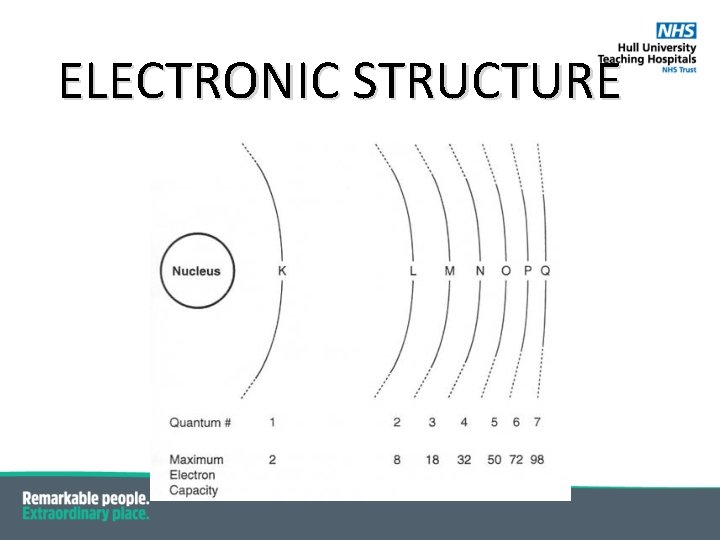
ELECTRONIC STRUCTURE
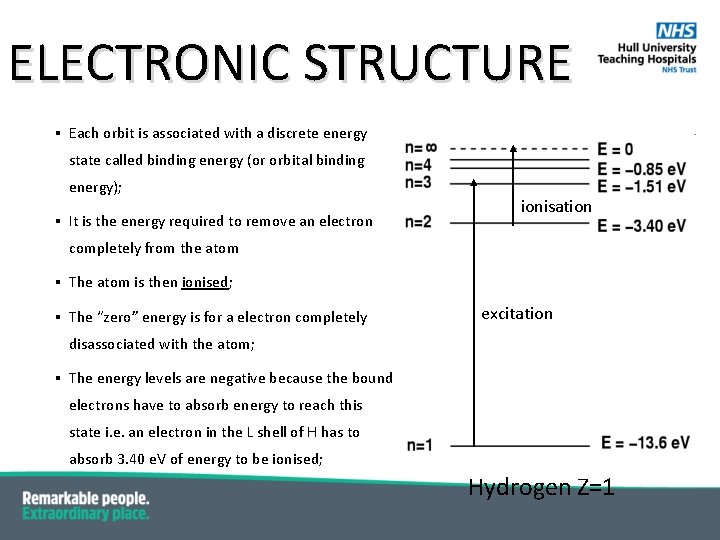
ELECTRONIC STRUCTURE § Each orbit is associated with a discrete energy state called binding energy (or orbital binding energy); § It is the energy required to remove an electron ionisation completely from the atom § The atom is then ionised; § The “zero” energy is for a electron completely excitation disassociated with the atom; § The energy levels are negative because the bound electrons have to absorb energy to reach this state i. e. an electron in the L shell of H has to absorb 3. 40 e. V of energy to be ionised; Hydrogen Z=1
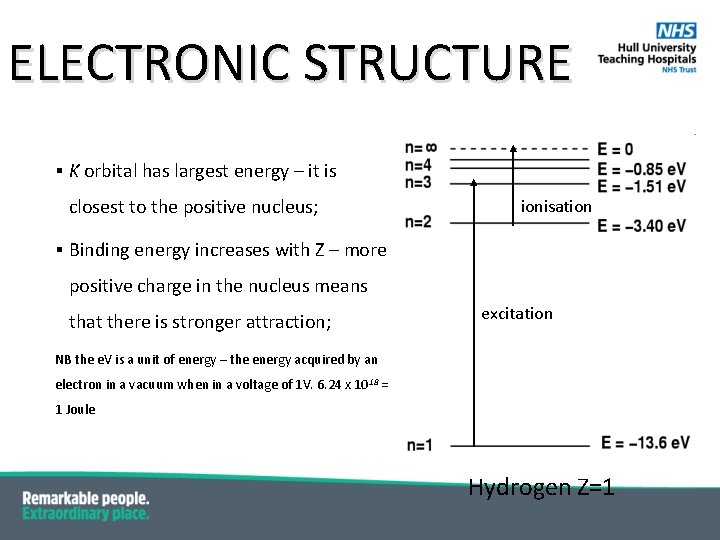
ELECTRONIC STRUCTURE § K orbital has largest energy – it is closest to the positive nucleus; ionisation § Binding energy increases with Z – more positive charge in the nucleus means that there is stronger attraction; excitation NB the e. V is a unit of energy – the energy acquired by an electron in a vacuum when in a voltage of 1 V. 6. 24 x 1018 = 1 Joule Hydrogen Z=1
ELECTRONIC STRUCTURE

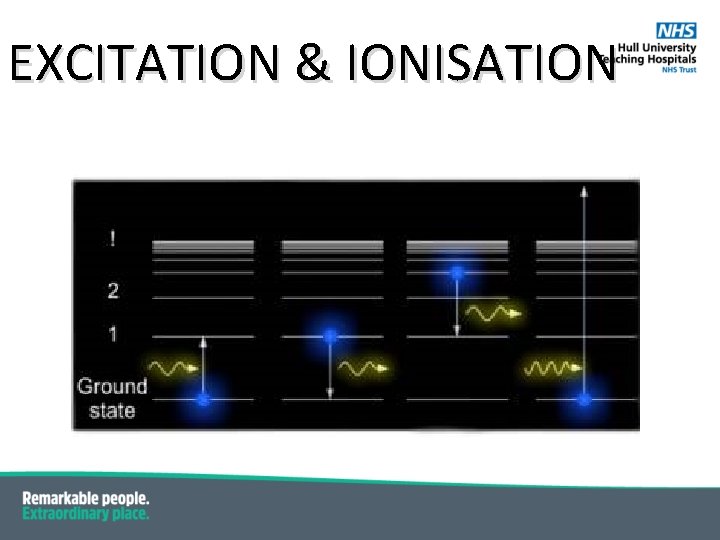
EXCITATION & IONISATION § Excitation of the atom § energy transferred to an orbiting electron § electron “jumps” from lower to higher energy levels § the atom is “excited” In the above diagram of hydrogen the energy required to excite an electron from the K shell to the M shell is: 13. 5 e. V – 1. 5 e. V = 12. 0 e. V

EXCITATION & IONISATION § Ionisation of the atom § energy transferred to an orbiting electron § electron removed from the electric field of nucleus § the atom is “ionised” § As described above, the ionisation energy is the energy of the shell i. e. the ionisation energy of the K shell of hydrogen is 13. 6 e. V.
EXCITATION & IONISATION

ELECTRON CASCADE § Electron removed from its shell (i. e. ionisation) by § an X-Ray photon § a γ-Ray photon § a charged particle (e. g. electron, proton) § Vacancy created in shell § usually filled by an electron from outer shell § Secondary vacancy in outer shell § filled by an electron transition from a more outer shell § The phenomenon is called Electron Cascade

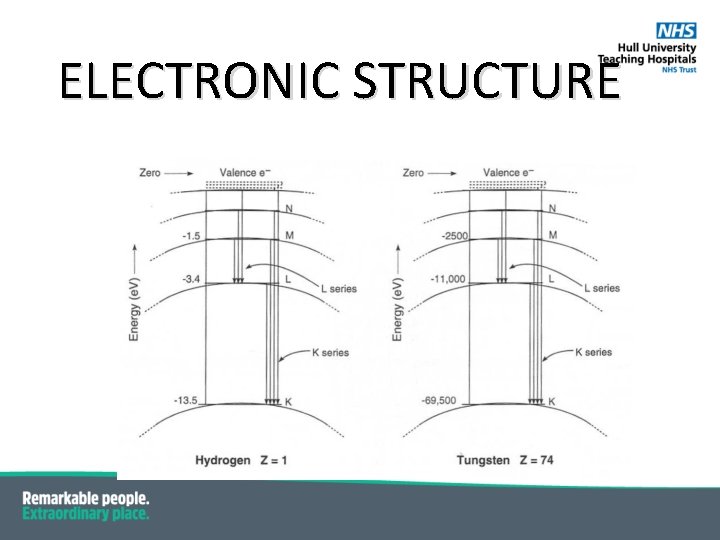
CHARACTERISTIC X-RAYS § Electrons moving between shells have to lose their energy – they do this by § § § § emitting a photon i. e. light in the visible, UV or X-ray part of the spectrum; Electron transitions ----> emission of radiation These are so called Characteristic X-rays; Characteristic of the atom itself due to the different energy levels in different atoms; Naming convention comes from the orbital in which the vacancy occurred i. e. a vacancy in the K-shell is a K-characteristic X-ray. If a vacancy is filled by an adjacent shell is given a subscript α (alpha) i. e. a vacancy in the K-shell being filled by an electron from the L-shell is Kα; If a vacancy is filled by a non-adjacent shell it is given subscript beta i. e. a vacancy in the K-shell being filed by an electron from the M-shell is Kβ. The energy of the characteristic X-ray is the difference in the binding energies so for Tungsten below: Energy of Kα (L-shell to K-shell) = 69. 5 ke. V – 11 ke. V = 58. 5 ke. V

CHARACTERISTIC X-RAYS De-excitation of a tungsten atom
X-RAY TUBE
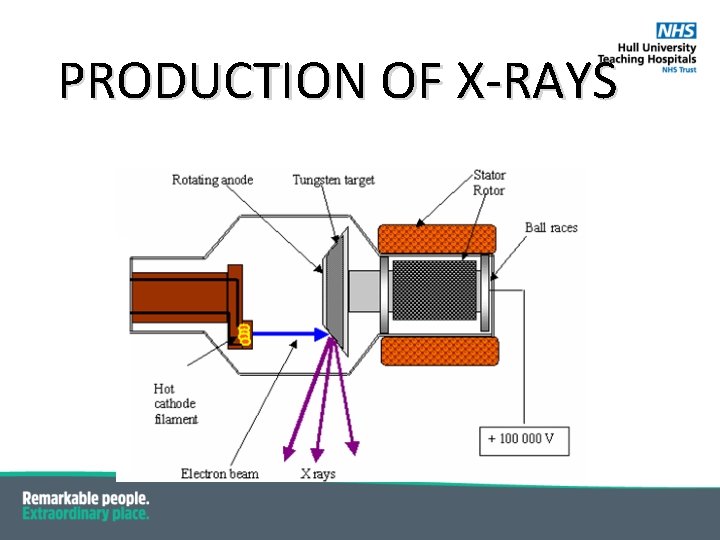
PRODUCTION OF X-RAYS
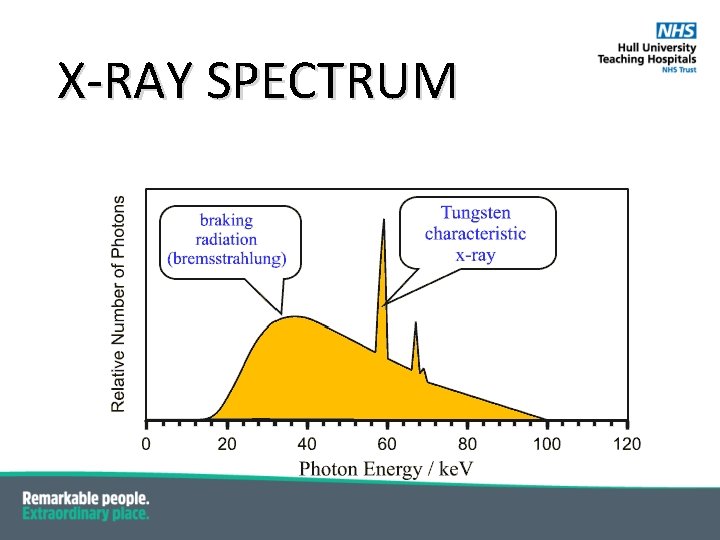
X-RAY SPECTRUM

AUGER ELECTRONS § Energy not always released as a photon; § Predominant in low-Z elements is Auger electron emission; § Energy released an orbital electron § Ejected Auger electron has a kinetic energy equal to: the difference between the transition energy and the binding energy of the ejected electron (69. 5 – 2. 5) – 2. 5 = 64. 5 ke. V
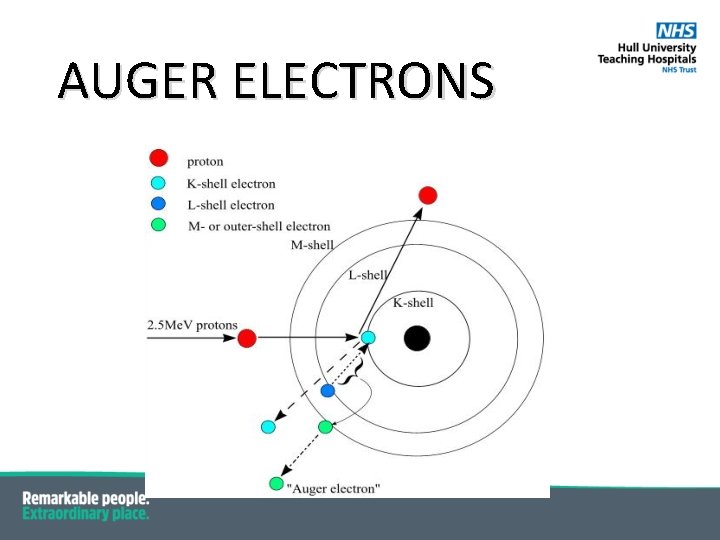
AUGER ELECTRONS

FLUORESCENT YIELD § Fluorescent yield (ω) is the probability § characteristic radiation emitted § Auger emission predominates in § low-Z elements § in electron transitions of the outer shells § K-shell fluorescent yield is essentially § <1% for elements with Z<10 (i. e. majority of soft tissue) § 15% for Calcium (Z=20) § 65% for Iodine (Z=53) § ~ 80% for elements with Z>60

Luminescence • Above we talked about the emission of characteristic X-rays; • It is possible for the difference between the electron shells to be in the visible light range; • This phenomenon of the emission of light is called luminescence; • If the electrons move between shells immediately -> fluorescent; • If the electrons have a delayed relaxation -> phosphorescent; Worth noting that luminescence is not a phenomenon unique to ionising radiation. Applications include scintillation detectors in our field.

The Nucleus

NUCLEAR ENERGY LEVELS § The nucleus has energy levels § analogous to orbital electron shells § often much higher in energy § The lowest energy state is called the ground state § Nuclei with excess energy are in an excited state § Excited states (100 s years>t>10 -12 sec) referred to as § metastable or isomeric states (e. g. 99 Tcm)
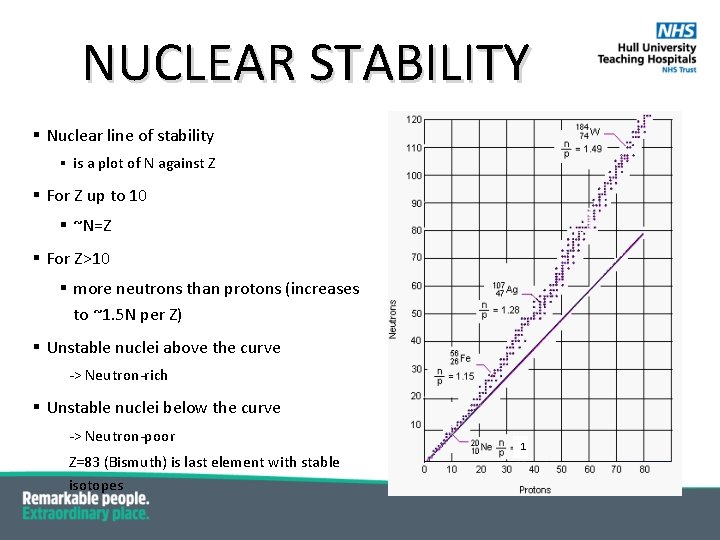
NUCLEAR STABILITY § Nuclear line of stability § is a plot of N against Z § For Z up to 10 § ~N=Z § For Z>10 § more neutrons than protons (increases to ~1. 5 N per Z) § Unstable nuclei above the curve -> Neutron-rich § Unstable nuclei below the curve -> Neutron-poor Z=83 (Bismuth) is last element with stable isotopes 1

UNSTABLE NUCLEI § Combinations of unstable nuclei DO exist § over time decay to stable nuclei § Two kinds of instability § neutron excess § neutron deficiency (proton excess) § Such nuclei have excess internal energy § Stability achieved through conversion of § a neutron to a proton § a proton to a neutron Emission of energy

RADIOACTIVITY § Nuclides (isotopes) decaying to more stable nuclei are § Radioactive § The process is called § Radioactive Decay or Radioactivity § A nucleus undergoes a series of radioactive decays until it reaches a stable configuration

GAMMA RAYS § Analogous to the emission of characteristic X-Rays but generally much more energetic; § Nucleus in excited state (often from nuclear decay); § Nucleus decays to a lower (more stable) energy state § Electromagnetic radiation emitted § This electromagnetic radiation is called a § γ-ray § Gamma rays stem from the nucleus;

Internal Conversion § Analogous to Auger electron process; § Nucleus in excited state; § Alternative mechanism of decay to gamma rays; § All de-excitation energy transferred to orbital electron; § Electron energy = excitation energy - binding energy; § Hole left be electron filled by electron cascade.

Atomic and Nuclear Binding Energy § Nuclear binding energy is energy required to completely separate nucleus into constituent parts; § Nuclear binding energy >>> electron binding energy; § Atomic binding energy is the sum of the two i. e. energy required to completely separate an atom into constituent parts; § Bringing two subatomic particles together their total energy decreases due to strong nuclear force; § ∴ bound sub-atomic particles have less energy than free particles

Mass Defect § E=mc 2 where c is speed of light = 3. 00 x 108 m/s; (1 amu = 931. 5 Me. V) § As above, energy of bound particles is less than free particles -> mass of bound particles less than sum of mass of free particles; § E. g. N-14 atom mass is 14. 00307 amu; § Mass of 7 p + 7 n + 7 e is 14. 11534 amu; § Mass defect = 14. 11534 – 14. 00307 = 0. 11227 amu = 104. 5 Me. V = atomic binding energy § Atomic binding energy per nucleon = 104. 5 / 14 = 7. 5 Me. V (EB / A)
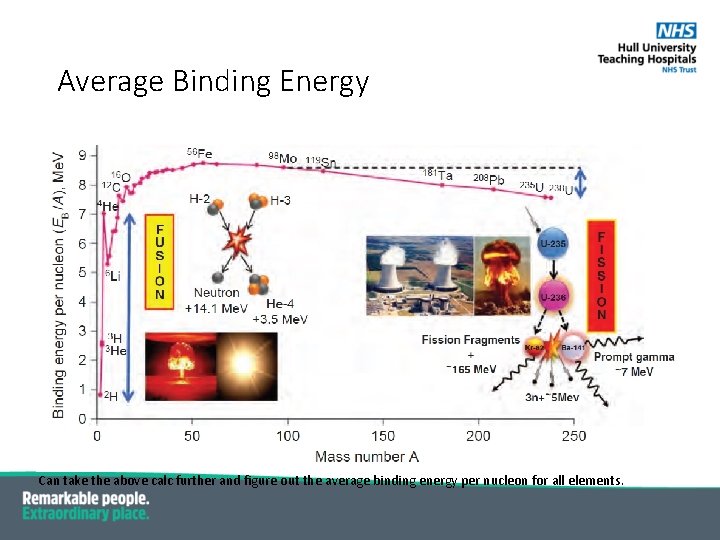
Average Binding Energy Can take the above calc further and figure out the average binding energy per nucleon for all elements.

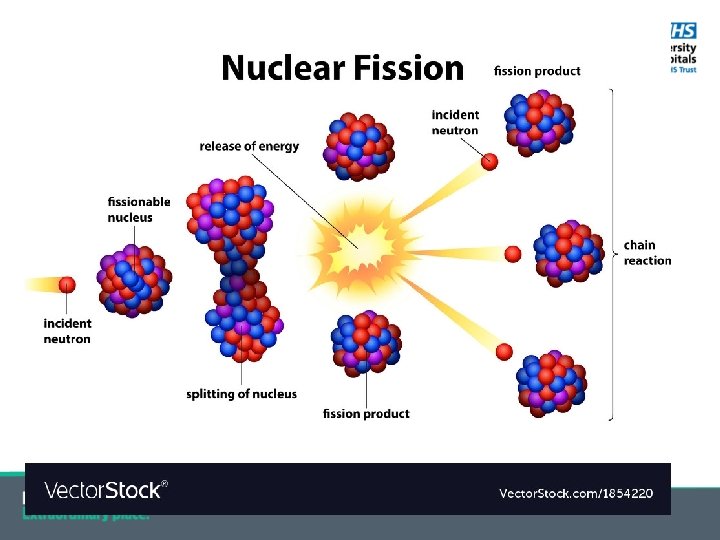
Nuclear Fission § Splitting of a large nucleus into two smaller parts; § Separate parts have a higher average binding energy; § Overall total nuclear binding energy increases; § This energy released as radiation and kinetic energy of the fragments; § Typically also releases energetic neutrons -> more fission; Eg. U-235 + n -> U-236 -> Sn-131 + Mo-102 + 3 n + energy § Process used in nuclear power plants and atom bombs;

Nuclear Fusion § Joining of two small nucleus atoms; Eg. H-3 + H-2 -> He-4 + n § Overall nuclear binding energy (greatly) increases; § Needs a large amount of (heat/kinetic) energy to initiate fusion – to overcome Coulomb forces; Eg. The sun, H-bomb (triggered by an atom bomb);

Any questions ? With thanks to Manos Papadopoulos for the original slides (2015).
- Slides: 51