Fractional distillation fill in the gaps A fractionating
- Slides: 2

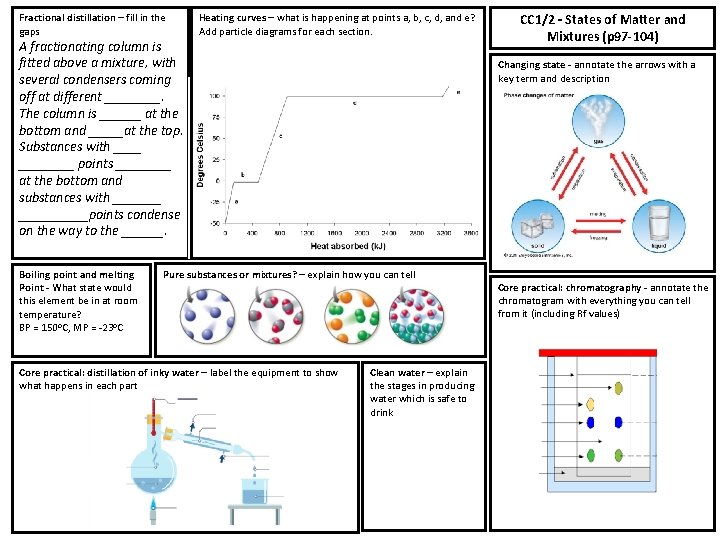
Fractional distillation – fill in the gaps A fractionating column is fitted above a mixture, with several condensers coming off at different ____. The column is ______ at the bottom and _____at the top. Substances with ________ points ____ at the bottom and substances with __________points condense on the way to the ______. Boiling point and melting Point - What state would this element be in at room temperature? BP = 150 o. C, MP = -23 o. C Heating curves – what is happening at points a, b, c, d, and e? Add particle diagrams for each section. Changing state - annotate the arrows with a key term and description Pure substances or mixtures? – explain how you can tell Core practical: distillation of inky water – label the equipment to show what happens in each part CC 1/2 – States of Matter and Mixtures (p 97 -104) Clean water – explain the stages in producing water which is safe to drink Core practical: chromatography - annotate the chromatogram with everything you can tell from it (including Rf values)

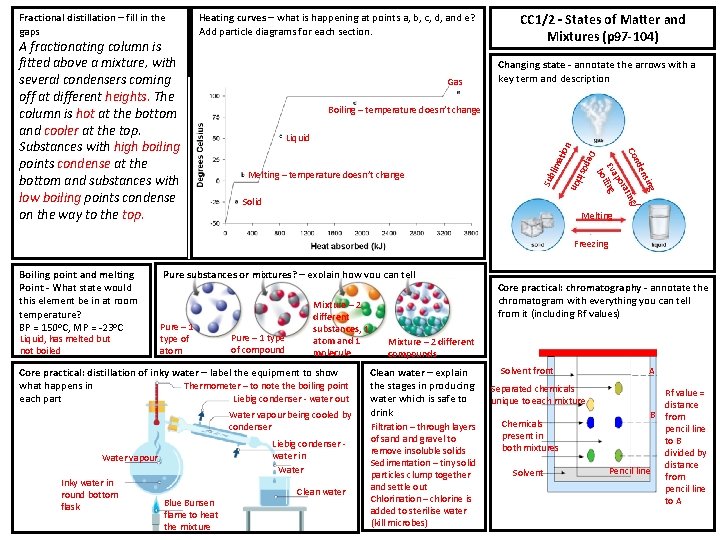
Gas CC 1/2 – States of Matter and Mixtures (p 97 -104) Changing state - annotate the arrows with a key term and description Boiling – temperature doesn’t change Melting – temperature doesn’t change lim atio n Liquid Solid g sin den g/ Con tin ora ap Ev iling bo De p o siti on A fractionating column is fitted above a mixture, with several condensers coming off at different heights. The column is hot at the bottom and cooler at the top. Substances with high boiling points condense at the bottom and substances with low boiling points condense on the way to the top. Heating curves – what is happening at points a, b, c, d, and e? Add particle diagrams for each section. Sub Fractional distillation – fill in the gaps Melting Freezing Boiling point and melting Point - What state would this element be in at room temperature? BP = 150 o. C, MP = -23 o. C Liquid, has melted but not boiled Pure substances or mixtures? – explain how you can tell Pure – 1 type of atom Pure – 1 type of compound Mixture – 2 different substances, 1 atom and 1 molecule Core practical: distillation of inky water – label the equipment to show Thermometer – to note the boiling point what happens in Liebig condenser - water out each part Water vapour being cooled by condenser Liebig condenser water in Water vapour Inky water in round bottom flask Blue Bunsen flame to heat the mixture Clean water Core practical: chromatography - annotate the chromatogram with everything you can tell from it (including Rf values) Mixture – 2 different compounds Clean water – explain the stages in producing water which is safe to drink Filtration – through layers of sand gravel to remove insoluble solids Sedimentation – tiny solid particles clump together and settle out Chlorination – chlorine is added to sterilise water (kill microbes) Solvent front Separated chemicals unique to each mixture Chemicals present in both mixtures Solvent A Rf value = distance B from pencil line to B divided by distance Pencil line from pencil line to A