Fractional Distillation and Cracking Boiling point of the

Fractional Distillation and Cracking Boiling point of the alkane (°C) 30/01/2022 Bar chart showing the link between the number of carbons in an alkane chain and the temperature at which the alkane boils 200 150 100 As the chain length of the hydrocarbon increases its boiling point increases. 50 0 -50 -100 -150 -200 1 2 3 4 5 6 7 8 9 10 1 carbon in chain (methane) b. p. = -160°C; 4 carbons (butane) = -4°C; 10 carbons 170°C Number of carbons in alkane chain (no units) Use the bar chart above to explain the link between the length of a hydrocarbon chain (in this case an alkane) and the temperature at which it boils. How might we use these differences to separate them?
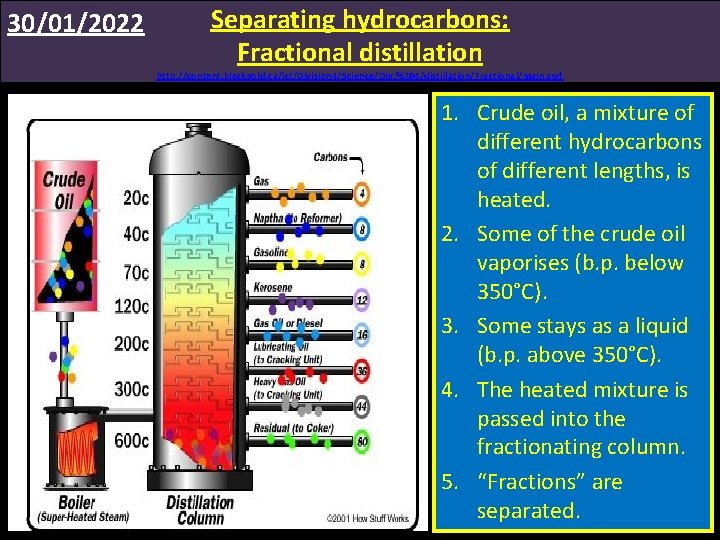
30/01/2022 Separating hydrocarbons: Fractional distillation http: //content. blackgold. ca/ict/Division 4/Science/Div. %204/distillation/Fractional/main. swf 1. Crude oil, a mixture of different hydrocarbons of different lengths, is heated. 2. Some of the crude oil vaporises (b. p. below 350°C). 3. Some stays as a liquid (b. p. above 350°C). 4. The heated mixture is passed into the fractionating column. 5. “Fractions” are separated.
How does fractional distillation work? 3 of 21 © Boardworks Ltd 2011
Forces between molecules 4 of 21 © Boardworks Ltd 2011

30/01/2022 Fractional Distillation: Getting the vocabulary right! • When discussing fractional distillation your language MUST be comparative: Longer chained hydrocarbons. Shorter chained hydrocarbons. Greater/weaker intermolecular forces. More/Less energy requires to overcome the intermolecular forces. – This leads to a higher/lower boiling point. – Difference in boiling points allows for separation. – –
30/01/2022 Question time! • Have a go at the questions on pages:
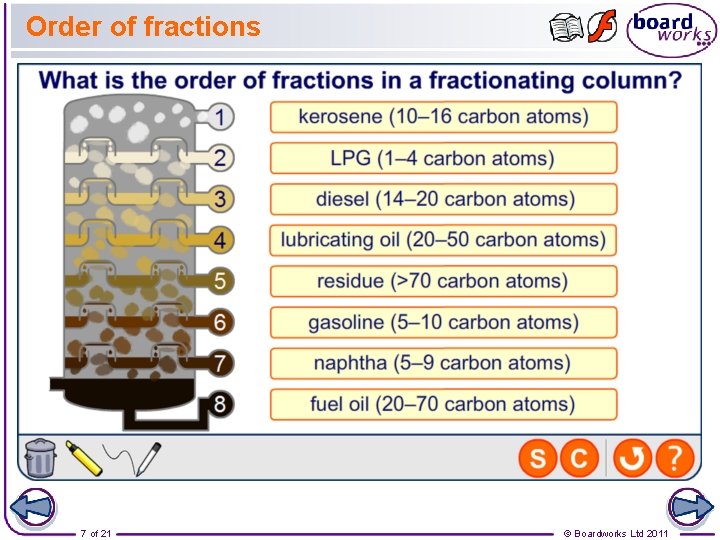
Order of fractions 7 of 21 © Boardworks Ltd 2011

Fractional distillation – true or false? 8 of 21 © Boardworks Ltd 2011

What are fractions used for? 9 of 21 © Boardworks Ltd 2011
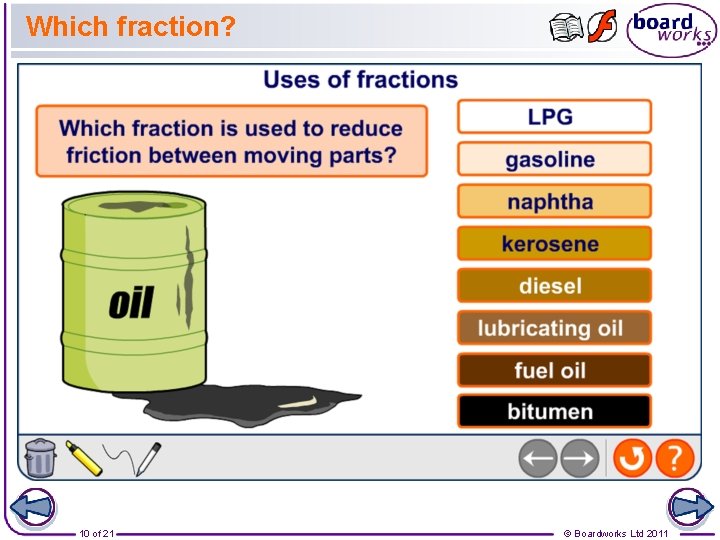
Which fraction? 10 of 21 © Boardworks Ltd 2011
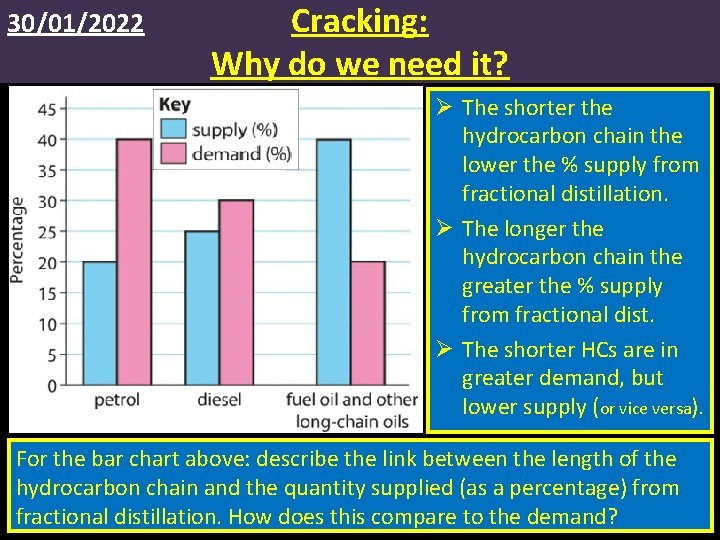
30/01/2022 Cracking: Why do we need it? Ø The shorter the hydrocarbon chain the lower the % supply from fractional distillation. Ø The longer the hydrocarbon chain the greater the % supply from fractional dist. Ø The shorter HCs are in greater demand, but lower supply (or vice versa). For the bar chart above: describe the link between the length of the hydrocarbon chain and the quantity supplied (as a percentage) from fractional distillation. How does this compare to the demand?

30/01/2022 Supply and demand • Shorter chained hydrocarbons/alkanes are more beneficial and valuable: they are easier to ignite and burn with a cleaner flame, making them better fuels than longer chained hydrocarbons. • This makes the shorter chained alkanes in higher demand than the longer chained hydrocarbons. • Problem: from fractional distillation we obtain greater quantities of the longer chained alkanes. • Shorter chained alkanes: Higher demand, but low supply. • Longer chained alkanes: Lower demand, but higher supply.

How does catalytic cracking work? 13 of 18 © Boardworks Ltd 2011

Catalytic cracking in the lab 14 of 18 © Boardworks Ltd 2011
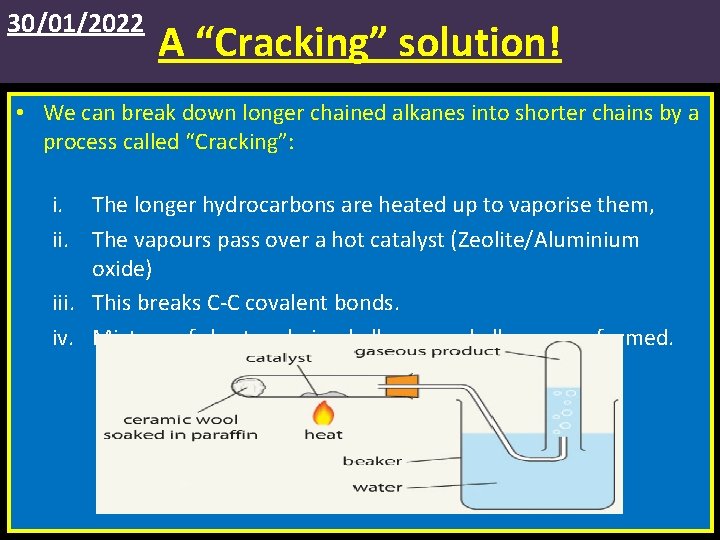
30/01/2022 A “Cracking” solution! • We can break down longer chained alkanes into shorter chains by a process called “Cracking”: i. The longer hydrocarbons are heated up to vaporise them, ii. The vapours pass over a hot catalyst (Zeolite/Aluminium oxide) iii. This breaks C-C covalent bonds. iv. Mixture of shorter chained alkanes and alkenes are formed.
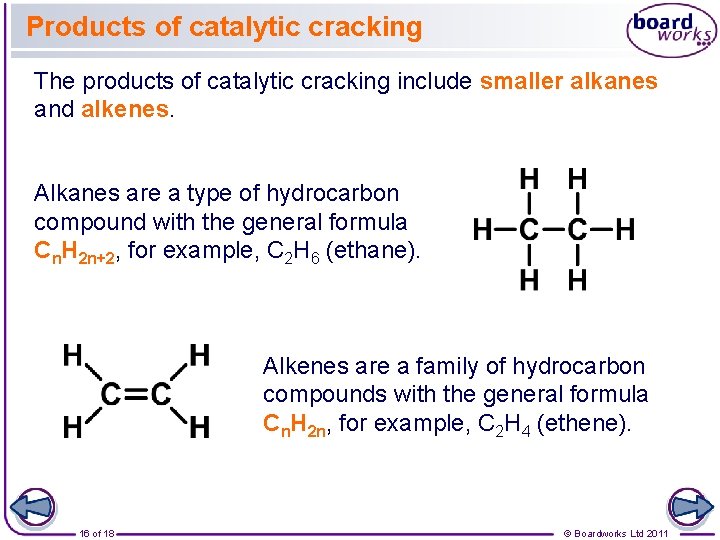
Products of catalytic cracking The products of catalytic cracking include smaller alkanes and alkenes. Alkanes are a type of hydrocarbon compound with the general formula Cn. H 2 n+2, for example, C 2 H 6 (ethane). Alkenes are a family of hydrocarbon compounds with the general formula Cn. H 2 n, for example, C 2 H 4 (ethene). 16 of 18 © Boardworks Ltd 2011
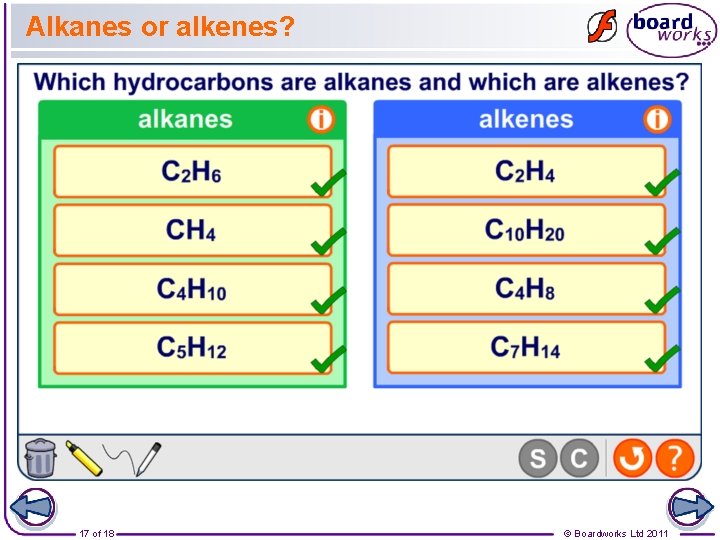
Alkanes or alkenes? 17 of 18 © Boardworks Ltd 2011
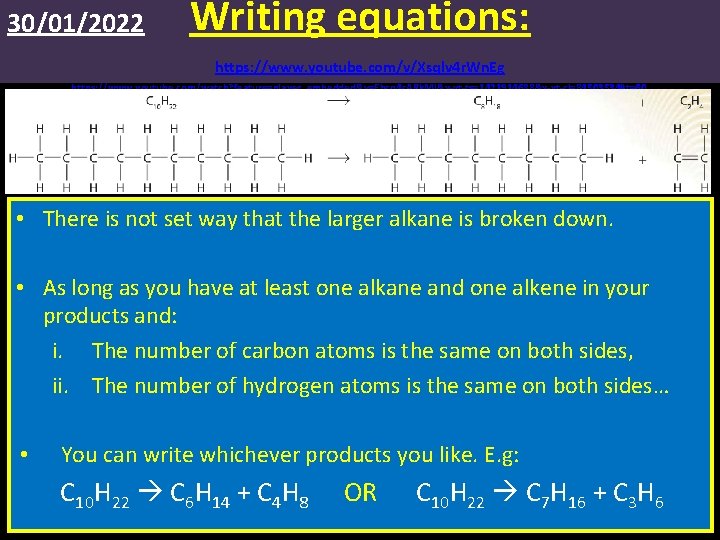
30/01/2022 Writing equations: https: //www. youtube. com/v/Xsqlv 4 r. Wn. Eg https: //www. youtube. com/watch? feature=player_embedded&v=Ehcq 4 c. ARk. MI&x-yt-ts=1421914688&x-yt-cl=84503534#t=60 • There is not set way that the larger alkane is broken down. • As long as you have at least one alkane and one alkene in your products and: i. The number of carbon atoms is the same on both sides, ii. The number of hydrogen atoms is the same on both sides… • You can write whichever products you like. E. g: C 10 H 22 C 6 H 14 + C 4 H 8 OR C 10 H 22 C 7 H 16 + C 3 H 6

30/01/2022 Writing equations: https: //www. youtube. com/v/Xsqlv 4 r. Wn. Eg https: //www. youtube. com/watch? feature=player_embedded&v=Ehcq 4 c. ARk. MI&x-yt-ts=1421914688&x-yt-cl=84503534#t=60 • How many different combinations of products can be made from C 10 H 22? • Write a word and symbol equation for each possible combination.

30/01/2022 • • C 10 H 22 Many possibilities! C 10 H 22 CH 4 + C 9 H 18 C 10 H 22 C 2 H 6 + C 8 H 16 C 10 H 22 C 3 H 8 + C 7 H 14 C 10 H 22 C 4 H 10 + C 6 H 12 C 10 H 22 C 5 H 12 + C 5 H 10 C 10 H 22 C 6 H 14 + C 4 H 8 C 10 H 22 C 7 H 16 + C 3 H 6 C 10 H 22 C 8 H 18 + C 2 H 4 • We could even have more products: • C 10 H 22 C 5 H 12 + C 2 H 4 + C 3 H 6
30/01/2022 Question time! • Have a go at the questions on pages:
- Slides: 21