Formulas Naming Compounds Chemical Formulas Describes a compounds

Formulas & Naming Compounds
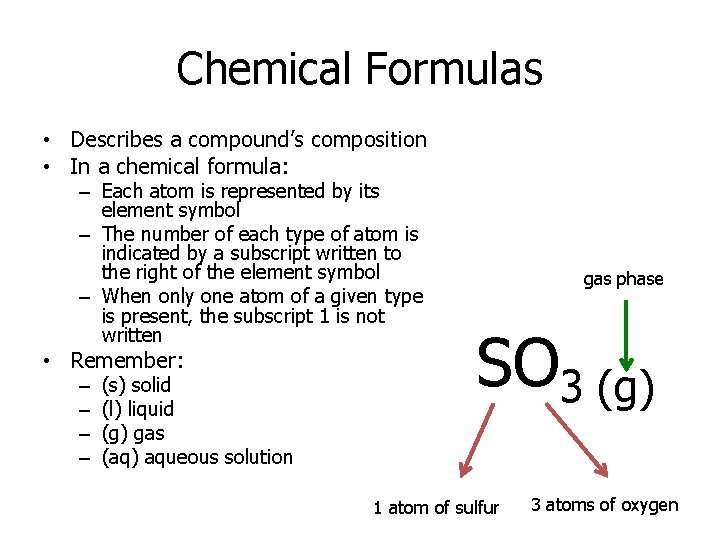
Chemical Formulas • Describes a compound’s composition • In a chemical formula: – Each atom is represented by its element symbol – The number of each type of atom is indicated by a subscript written to the right of the element symbol – When only one atom of a given type is present, the subscript 1 is not written • Remember: – – (s) solid (l) liquid (g) gas (aq) aqueous solution gas phase SO 3 (g) 1 atom of sulfur 3 atoms of oxygen
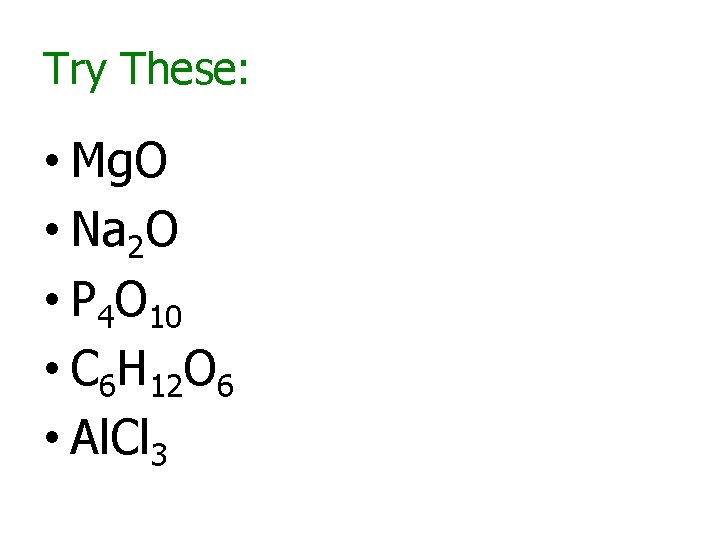
Try These: • Mg. O • Na 2 O • P 4 O 10 • C 6 H 12 O 6 • Al. Cl 3

Naming Ionic Compounds All ionic compounds are electrically neutral ---we will use this fact to make proper balanced formulas for the rest of the year!!!!

Binary Ionic Compounds • Compounds made up of only 2 elements Ø One component is from a cation of a metal such as Na+1, Li+1, Ca +2… Ø The other component is from an anion of a -1 -1 -2 nonmetal such as F , Cl , O … Ø Ex. Na. Cl, Li 2 O, Ca. F 2

Rules for Naming Type I Binary Compounds Ø A compound containing a metal and a nonmetal in which the metal present forms only one type of cation 1. The cation is always named first and the anion second 2. A simple cation takes its name from the name of the element 3. A simple anion is named by taking the first part of the element name (the root) and adding the ending –ide. Ex. KBr = Potassium Bromide Na. Cl = Sodium Chloride Al. F 3 = Aluminum Fluoride

Try These: • KI • Cs. Br • Mg. I 2 • Li 2 S • Ca. F 2

Rules for Naming Type II Binary Compounds Ø A compound containing a metal and a nonmetal in which the metal present can form two or more cations that have different charges (many transition metals are like this) • Same rules apply as with Type I Binary Compounds but because the metals in a type II binary compound can have more than one charge, the ion present in the compound must be specified in the name by using a Roman numeral • Some transition elements only have one oxidation state. Do not use. Roman numerals to name them. Ex. Cu. Cl = Copper I Chloride (Cu+1 and Cl-1) (Copper can have a +1 or +2 charge) Fe 2 O 3 = Iron III Oxide (Fe+3 and O-2) (Iron can have a +2 or +3 charge) Mn. O 2 = Manganese IV Oxide (Mn+4 and O-2) (Manganese can have a +2, +3, +4, or +7 charge)

Try These: • Cr. Cl 3 • Co. Br 2 • Hg. O • Sn. O 2 • Ni. S
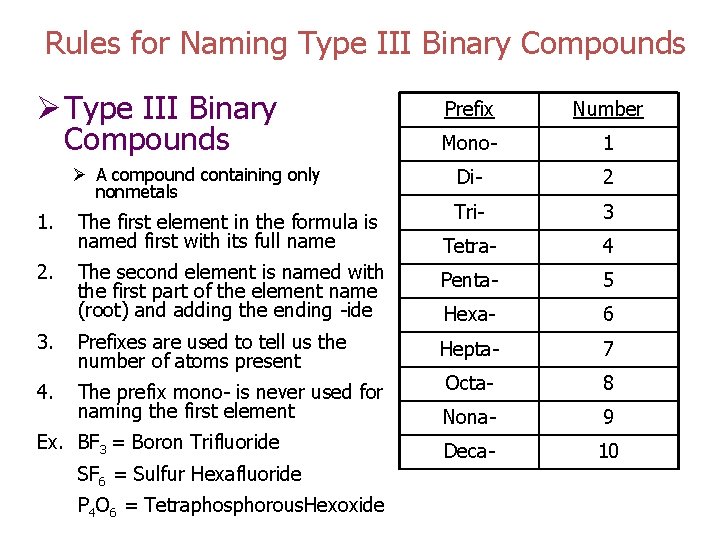
Rules for Naming Type III Binary Compounds Ø Type III Binary Compounds Prefix Number Mono- 1 Di- 2 Tri- 3 Tetra- 4 The second element is named with the first part of the element name (root) and adding the ending -ide Penta- 5 Hexa- 6 3. Prefixes are used to tell us the number of atoms present Hepta- 7 4. The prefix mono- is never used for naming the first element Octa- 8 Nona- 9 Deca- 10 Ø A compound containing only nonmetals 1. 2. The first element in the formula is named first with its full name Ex. BF 3 = Boron Trifluoride SF 6 = Sulfur Hexafluoride P 4 O 6 = Tetraphosphorous. Hexoxide

Try These: • CCl 4 • NO 2 • CO • N 2 O 5

Naming Compounds that contain Polyatomic Ions Ø An ion containing several atoms bound together • Same rules apply as with Type I and Type II Binary compounds • Use “Table E” for the name of the common polyatomic ions • Ex. Na 2 SO 4 = Sodium Sulfate KMn. O 4 = Potassium Permanganate Cu(NO 2)2 = Copper II Nitrite

Try These: • Na 2 CO 3 • Cs. Cl. O 4 • Fe(NO 3)3 • Zn 3(PO 4)2 • NH 4(C 2 H 3 O 2)

Naming Acids Ø Acid Ø A substance that produces hydrogen ions when dissolved in water 1. If the acid does not contain oxygen then use the prefix hydro- and the anion root plus the –ic ending 2. If the acid has an oxygen containing polyatomic ion ending in –ate then use the anion root plus the –ic ending 3. If the acid has an oxygen containing polyatomic ion ending in –ite then use the anion root plus the –ous ending Ex. HCl = Hydochloric Acid H 2 SO 4 = Sulfuric Acid HNO 2 = Nitrous Acid

Try These: • H 3 PO 4 • HBr • HCl. O • HNO 3 • HC 2 H 3 O 2

Writing Formulas

• To write a correct formula, the oxidation numbers of the elements must be known. • When elements are ions, their oxidation number is the same as their charge (look at periodic table for oxidation numbers). • Elements in the same group usually have the same charge. Group 1 2 13 16 17 Charge +1 +2 +3 -2 -1 Example Na+1 Ba+2 Al+3 O-2 Cl-1

Criss-Cross Method • The oxidation state of the 1 stelement becomes the subscript of the 2 ndelement • The oxidation state of the 2 ndelement becomes the subscript of the 1 stelement • NOTE: Signs are not used Sodium Oxide Na+1 O-2 Na 2 O Na 2 O Never write a number 1 for a subscript
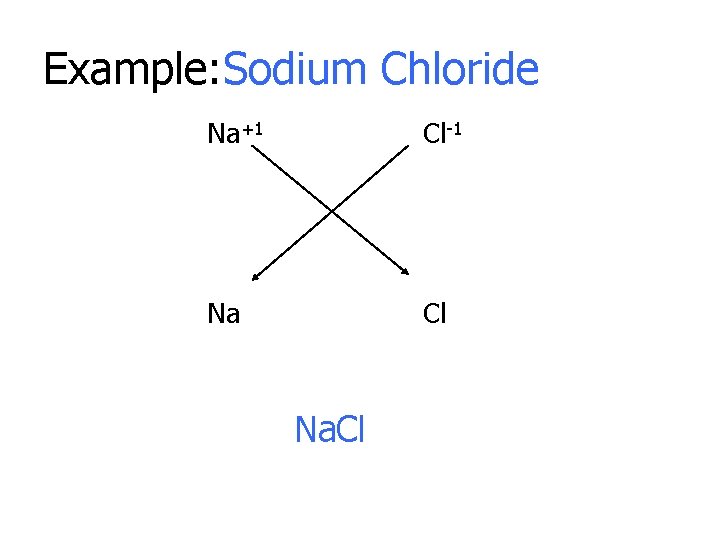
Example: Sodium Chloride Na+1 Cl-1 Na Cl Na. Cl
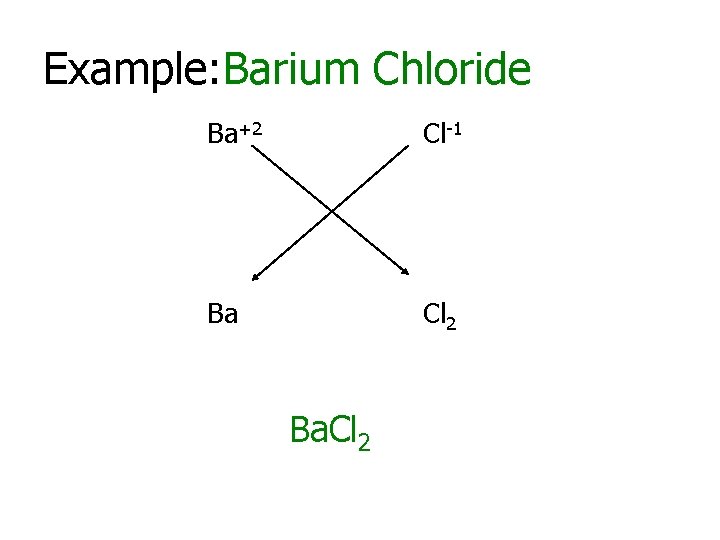
Example: Barium Chloride Ba+2 Cl-1 Ba Cl 2 Ba. Cl 2
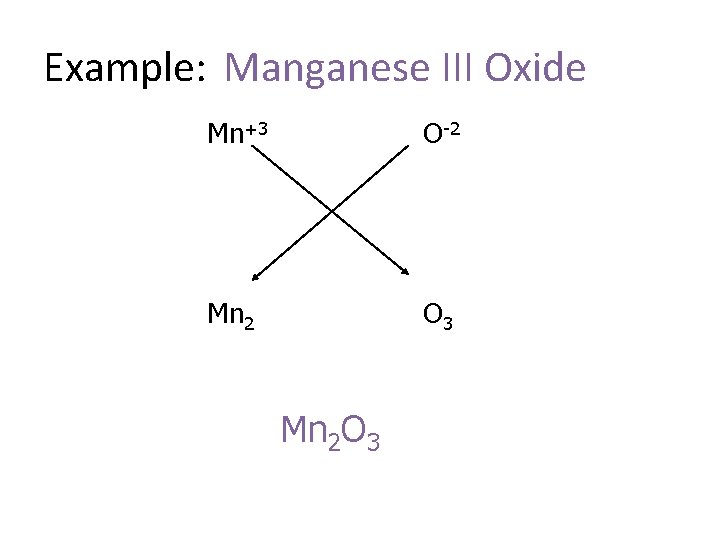
Example: Manganese III Oxide Mn+3 O-2 Mn 2 O 3 Mn 2 O 3
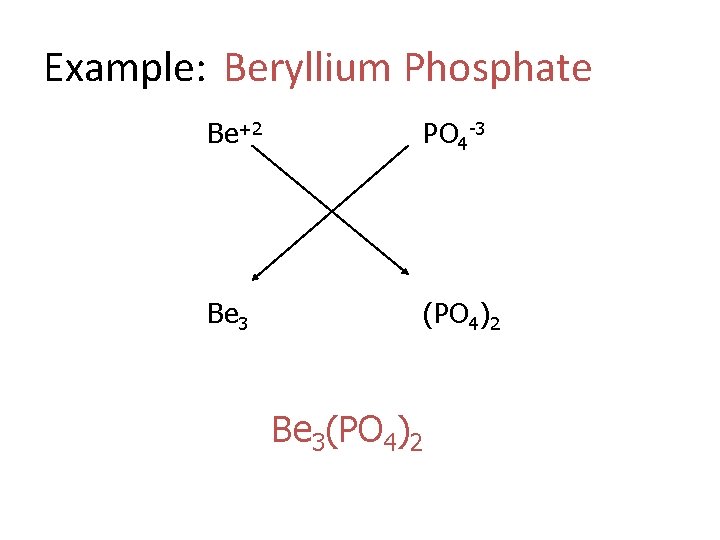
Example: Beryllium Phosphate Be+2 PO 4 -3 Be 3 (PO 4)2 Be 3(PO 4)2

Percent Composition

Percent Composition • Percent by mass of an element in a compound • Number of grams of the element divided by the mass in grams of the compound multiplied by 100 % Composition By Mass = T) Part x 100 (Formula in Table Whole

Example: When a 13. 60 g sample of a compound containing only magnesium and oxygen is decomposed, 5. 40 g of oxygen is obtained. What is the percent composition of this compound? % Mg = %O = mass of Mg x 100 = 8. 20 g mass of compound 13. 60 g mass of O mass of compound x 100 = 60. 3% x 100 = 5. 40 g x 100 = 39. 7% 13. 60 g 100%

Example: Propane (C 3 H 8), the fuel commonly used in gas grills, is one of the compounds obtained from petroleum. Calculate the percent composition of propane. %C= mass of. Cx 100 = 36. 0 gx 100 = 81. 8% mass of compound 44. 0 g %H = mass of H mass of compound x 100 = 8. 0 gx 100 = 18. 2% 44. 0 g 100%

Empirical & Molecular Formulas

Determining the Empirical Formula • Empirical Formula – Gives the lowest whole-number ratio of the atoms of the elements in a compound – To calculate, you need to know the: • gfm • % composition

Steps To Determine The Empirical Formula 1. Divide % by atomic mass (This tells us the number of moles. ) 2. Find the smallest whole # ratio. Divide all molar amounts by the smallest molar amount. (These #’s are the subscripts. )

Example: • A compound contains 40% carbon, 6. 67% hydrogen, 53. 3% oxygen by mass. What is the empirical formula? Step 1) C = 40. 0 H = 6. 67 O = 53. 3 12 1 16 C = 3. 33 H = 6. 67 O = 3. 33 Step 2) C = 3. 33 H = 6. 67 O = 3. 33 C= 1 H = 2 O = 1 The empirical formula is: CH 2 O

Determining the Molecular Formula • Molecular Formula – The same as the experimentally determined empirical formula, or it is a simple wholenumber multiple of its empirical formula – To calculate, you need to know the: • gfm • Empirical formula

Steps To Determine The Empirical Formula 1. Put the gfm of the substance in the numerator 2. Add the masses of the components in empirical formula 3. Divide step 1 by step 2 4. Multiply the subscripts of the empirical formula by the answer in step 3 Ø NOTE: The molecular formula is just a multiple of the empirical formula

Example: • If the empirical formula for a compound is CH 2 O and its gram formula mass is 180 g/mole, what is the molecular formula? Step 1) Step 2) 180/? C + H 2 + O 12 + 16 = 30 Step 3) 180 = 6 30 Step 4) CH 2 O x 6 The molecular formula is: C 6 H 12 O 6

Moles & Gram Formula Mass

Moles • A unit of measurement • One mole of any substance contains 6. 02 × 1023 particles (Avogadro’s number) • Particles may be atoms, molecules, ions, protons, electrons, etc. Think of a mole as a basket oranges 1 mole Fe H 2 O 1 mole 6. 02 × 1023 oranges atoms molecules He 1 mole protons 1 mole 6. 02 × 1023 atoms particles
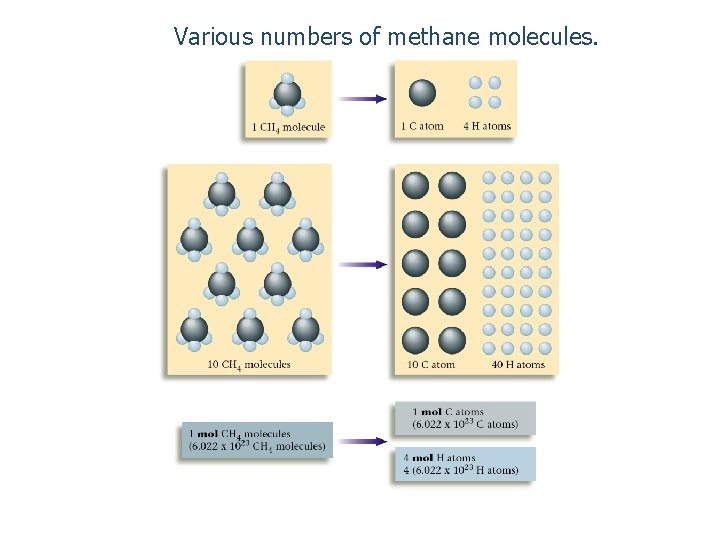
Various numbers of methane molecules.

Gram Formula Mass (Molar Mass) • Mass in grams of one mole of any substance 51. 9961 – (unit = g/mole) • Obtained by summing the masses of component atoms (values obtained for each element from the periodic table). Cr 24 +2 +3 +6

Example: • What is the mass of one mole of Fe ? • To obtain this value we look at the periodic table for the atomic mass value of Fe. • In this case: • Fe = 55. 8 g/mole round 56 g/mole

Example: • What is the mass of one mole of H 2 O ? • To obtain this value we look at the periodic table for the atomic mass values of H and O. • In this case: H = 1. 008 g/mole O = 16. 00 g/mole round 1 g/mole x 2 = 2 g/mole = 16 g/mole 18 g/mole

Try These: • Mass of 1 mole of Ar • Mass of 1 mole of CO 2 • Mass of 1 mole of N 2 • Mass of 1 mole of C 7 H 14 O 2

Mole-Molecule-Mass Conversions

It’s Back!!!!

Remember • Factor label method is used to convert a quantity expressed in one unit to a quantity expressed in a new unit Ø Uses conversion factors o A fraction relating the two units o Always equivalent to 1 Ø Choose the conversion factor that will cancel out the undesired units Ø Multiply the quantity by the conversion factor to give the quantity with the desired units

Moles & Mass • Determine the number of moles in 65 g of Li. Br. (grams moles) 65 g Li. Br x 1 mole =. 75 moles Li. Br 87 g Li. Br • Determine the mass of 1. 50 moles of Fe. Cl 3. (moles grams) 1. 50 moles Fe. Cl 3 x 161 g Fe. Cl 3 = 242 g Fe. Cl 3 1 mole

Moles & Molecules • Determine the number of molecules in 3. 75 moles. (moles molecules) 3. 75 moles x 6. 02 x 1023 molecules = 2. 26 x 1024 molecules 1 mole • Determine the number of moles in 2. 5 x 1018 molecules. (molecules moles) 2. 5 x 1018 molecules x 1 mole 6. 02 x 1023 molecules = 4. 2 x 10 -6 moles

Moles & Volume • Determine the volume of 5. 60 moles of CH 4. (Moles Volume) 5. 60 moles CH 4 x 22. 4 L CH 4 = 125 L CH 4 1 mole • Determine the number of moles in 37. 2 L of CO 2. (Volume Moles) 37. 2 L CO 2 x 1 mole = 1. 66 moles CO 2 22. 4 L CO 2

Two-Step Conversions • How many molecules are in 6. 5 g NH 3? grams molecules 6. 5 g NH 3 x 1 mole 17 g NH 3 x 6. 02 x 1023 molecules = 2. 3 x 1023 molecules NH 3 1 mole

• What volume would 25. 5 x 1012 molecules of SO 2 occupy? molecules moles liters 25. 5 x 1012 molecules SO 2 x 1 mole 6. 02 x 1023 molecules x 22. 4 L SO 2 = 9. 49 x 10 -10 L SO 2 1 mole
- Slides: 48