FORMULA MASS 1 Write out the symbols 2

FORMULA MASS 1) Write out the symbols 2) Write out how many you have 3) Multiply by the atomic mass (3 SD) 4) Total up the masses (3 SD) EXAMPLE = Ca (OH)2 Ca 1 X 40. 1 = 40. 1 O 2 X 16. 0 = 32. 0 H 2 X 1. 0 1= 2. 02 74. 0 grams
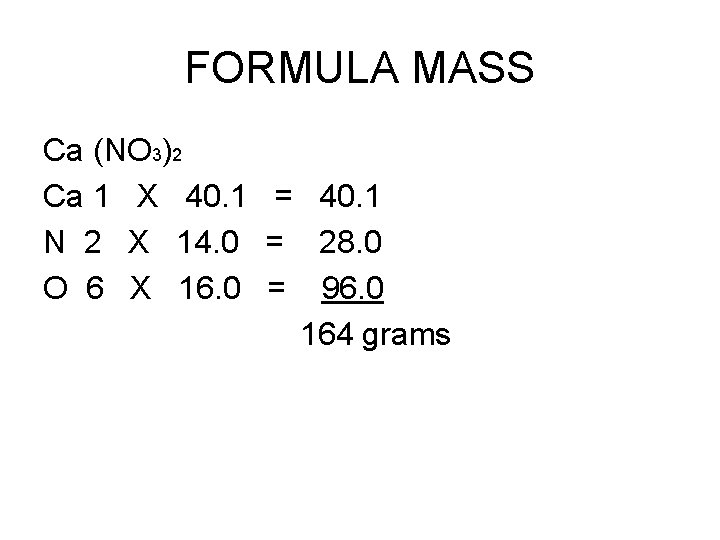
FORMULA MASS Ca (NO 3)2 Ca 1 X 40. 1 = 40. 1 N 2 X 14. 0 = 28. 0 O 6 X 16. 0 = 96. 0 164 grams

FACTOR LABEL METHOD 85. 2 INCHES = ? ? FEET GIVEN x CONVERSION = ? ? FACTOR 85. 2 INCHES x 1 FOOT = 7. 10 FT 12 INCHES THE UNITS INCHES CANCEL OUT LEAVING FEET IN THE ANSWER

PERCENT COMPOSITION Mg 1 X 24. 3 = 24. 3 N 2 X 14. 0 = 28. 0 formula mass O 6 X 16. 0 = 96. 0 148. 3 = 148 Mg 24. 3 = X 100 = 16. 4 % 148 N 28. 0 = X 100 = 18. 9 % 148 O 96. 0 = X 100 = 64. 9 % 148

CONVERT GRAMS INTO MOLES 85. 0 grams of Ag (NO 3) = ? ? moles Ag 1 x 108 = 108 N 1 x 14. 0= 14. 0 O 3 x 16. 0= 48. 0 170. g = 1 mole 85. 0 g x 1. 00 mole =. 500 moles 170. g

CONVERT MOLES INTO GRAMS 2. 70 MOLES of Ag (NO 3) = ? ? GRAMS Ag 1 x 108 = 108 N 1 x 14. 0= 14. 0 O 3 x 16. 0= 48. 0 170. g = 1 mole 2. 70 moles x 170 grams = 459 grams 1. 00 mole

Steps for empirical formulas given Ni = 20% convert to grams 20 g find moles mole ratio formula 20/ 58. 7. 341/. 341 =. 341 m =1 Ni =1 From atomic mass chart Br = 80% 80 g 80/ 79. 9 1. 00/. 341 = 1. 00 m =3 Br = 3 Ni Br 3

MOLECULAR FORMULA EMPIRICAL FORMULA MOLECULAR FORMULA EF = CO 2 MF =C 2 O 4 EMPIRICAL MASS MOLECULAR MASS EM = 44 MM = 88 88/44= 2

MAKE SURE YOU HAVE A CALCULATOR!!!!!
- Slides: 9