Forming Simple Ionic Compounds by Balancing Oxidation Numbers

Forming Simple Ionic Compounds by Balancing Oxidation Numbers How do we figure out how many chlorines will combine with a calcium atom to form calcium chloride? When one element forms positive ions and the other forms negative ions, it’s easy to determine the chemical formula!

Forming Simple Ionic Compounds l Get your periodic table of elements ready. l I’m not kidding. You need it now. l We’ll be looking at the alkali metals first.

Forming Simple Ionic Compounds. l Every alkali metal has just one valence electron. l How will those elements get a full octet (set of 8 to be like a noble gas)? l Gaining 7 electrons is too tough. l Each alkali metal will lose its only valence electron, leaving a full octet in the energy level below.
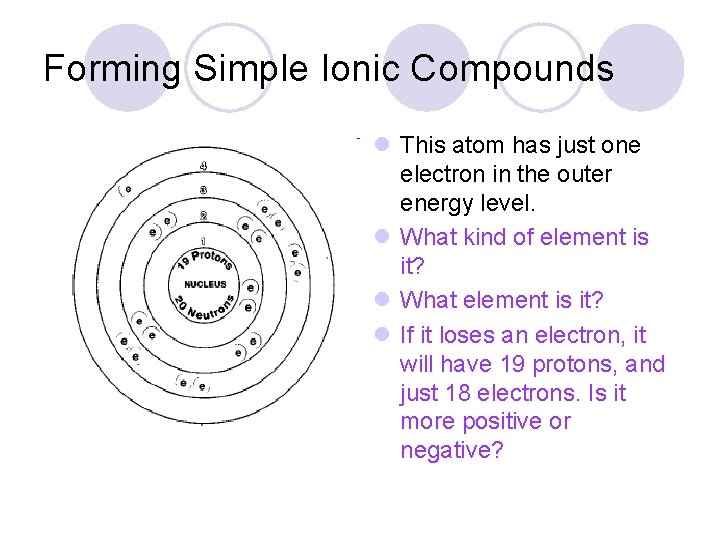
Forming Simple Ionic Compounds l This atom has just one electron in the outer energy level. l What kind of element is it? l What element is it? l If it loses an electron, it will have 19 protons, and just 18 electrons. Is it more positive or negative?

Forming Simple Ionic Compounds l When an atom loses an electron, it becomes a 1+ ion, because it has one more proton than electron. l All alkali metals become 1+ ions. l Their oxidation number is 1+. l What is cesium’s oxidation number? l What kind of ion does sodium form? l What is the charge of a lithium ion?
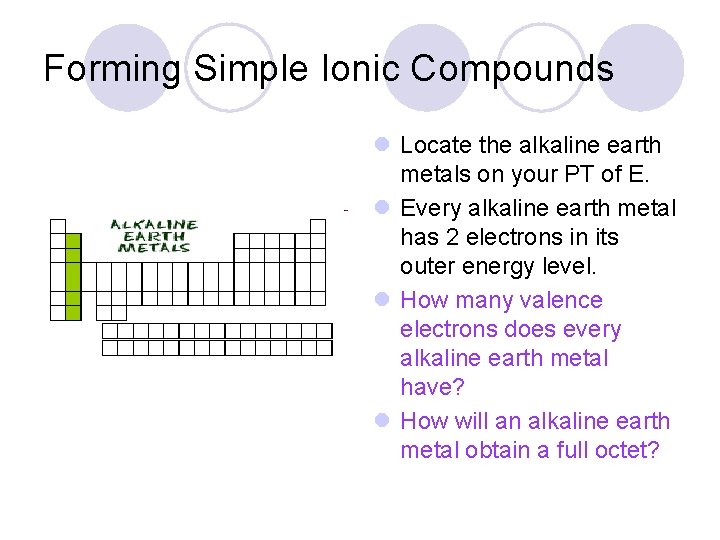
Forming Simple Ionic Compounds l Locate the alkaline earth metals on your PT of E. l Every alkaline earth metal has 2 electrons in its outer energy level. l How many valence electrons does every alkaline earth metal have? l How will an alkaline earth metal obtain a full octet?
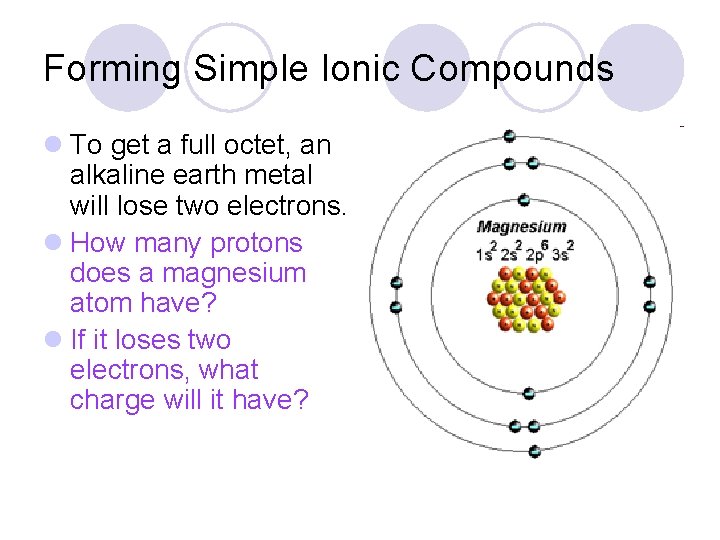
Forming Simple Ionic Compounds l To get a full octet, an alkaline earth metal will lose two electrons. l How many protons does a magnesium atom have? l If it loses two electrons, what charge will it have?

Forming Simple Ionic Compounds l Every alkaline earth metal will lose two electrons to form a 2+ ion. l The oxidation number for an alkaline earth metal is 2+. l What is the charge of a calcium ion? l What kind of ion is formed by a barium atom? l How is a beryllium ion different from a lithium ion?
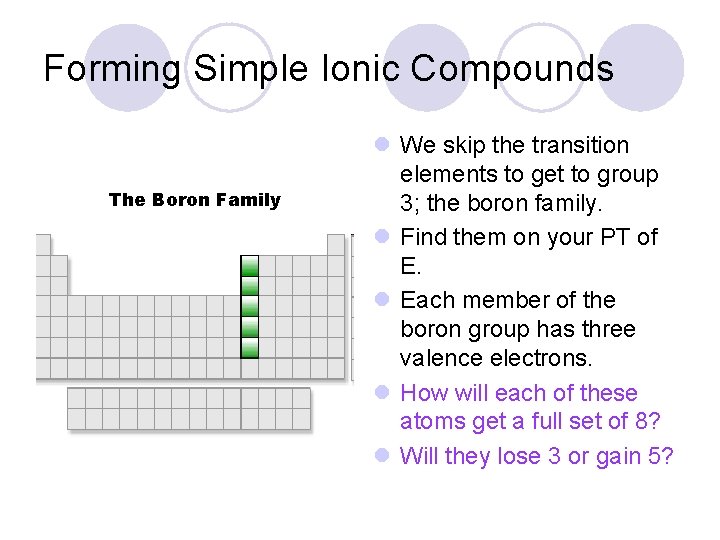
Forming Simple Ionic Compounds The Boron Family l We skip the transition elements to get to group 3; the boron family. l Find them on your PT of E. l Each member of the boron group has three valence electrons. l How will each of these atoms get a full set of 8? l Will they lose 3 or gain 5?

Forming Simple Ionic Compounds l What atom is shown here? l How do we know it is aluminum? l Whose model of the atom is being used to depict the distribution of subatomic particles? l What happens when this atom loses its three valence electrons?

Forming Simple Ionic Compounds l All elements on the boron group form ions by _______ three electrons. l The loss of three electrons causes every member of that group to form _____ ions. l The oxidation number for gallium is _____. l What kind of ion does indium form? l How is a boron ion different from a beryllium ion? l How is a boron ion like a beryllium ion?
Forming Simple Ionic Compounds l The carbon family, also known a group 4 A, contains elements with _____ valence electrons. l To get to a full octet, these elements usually _______ four electrons, giving them a 4+ charge. l Why does losing four electrons cause an atom to become an ion with a 4+ charge?

Forming Simple Ionic Compounds l The nitrogen group is next. Find it on your periodic table of elements. It is often known as Group 5 A of the PT of E. l Can you figure out which group is often called Group 4 A? 6 A? 7 A? 2 A? l How many valence electrons does each of the elements in Group 5 A have? l The nitrogen group elements form ions by gaining electrons. How many do they need to gain? l All elements in the nitrogen family form 3 - ions.

Forming Simple Ionic Compounds l The oxygen family is often known as Group _____ and has ____ valence electrons. l Do elements in the oxygen group gain or lose electrons to form ions? l What is the charge of an oxygen ion? l What is the oxidation number of polonium? l How many valence electrons does an atom of tellurium have? l How many valence electrons does a tellurium ion have?

Forming Simple Ionic Compounds l Group 7 A is the ____ family. l This group is more commonly known as the halogens. l Notice the ending of the name of every halogen. They all end with “-ine”. Any element that ends with “-ine” is a halogen. l Since the halogens have seven valence electrons, they form 1 - ions. l Explain why halogens have the oxidation number 1 -.

Forming Simple Ionic Compounds l The noble gases are group ______. l Their outer electron energy levels are full. l They do not form ions. l Oxidation number = 0. l They do not form compounds, generally speaking. l We can ignore them for the remainder of this lesson.

Forming Simple Ionic Compounds l Ionic compounds form because opposites attract. l Positive ions are electrostatically attracted to negative ions. l Positive charges must be balanced by negative charges for any ionic compound. l Positive and negative charges must add up to 0 for any ionic compound.

Forming Simple Ionic Compounds l In this illustration, a sodium atom loses an electron, making it a 1+ ion. l The chlorine atom takes the electron, making it a 1 - ion. l (+1) + (-1) = 0 l When the ions join, they will form a compound, sodium chloride, with a neutral (0) charge. l One sodium ion was balanced by one chloride ion. l Na. Cl is the formula.
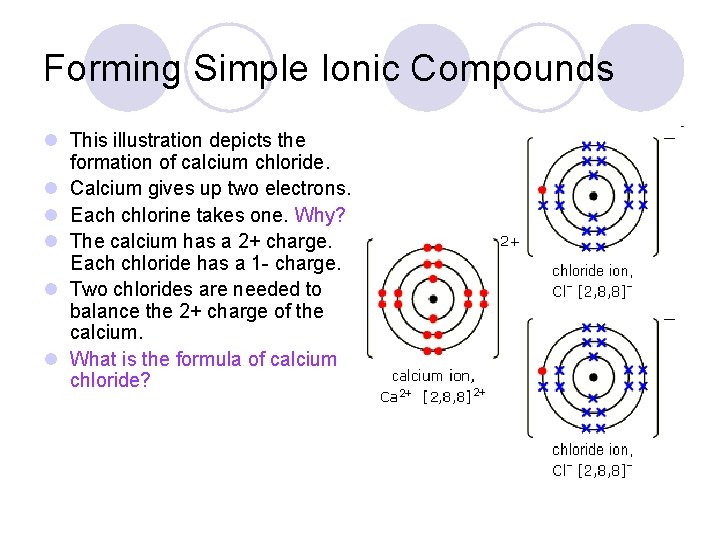
Forming Simple Ionic Compounds l This illustration depicts the formation of calcium chloride. l Calcium gives up two electrons. l Each chlorine takes one. Why? l The calcium has a 2+ charge. Each chloride has a 1 - charge. l Two chlorides are needed to balance the 2+ charge of the calcium. l What is the formula of calcium chloride?
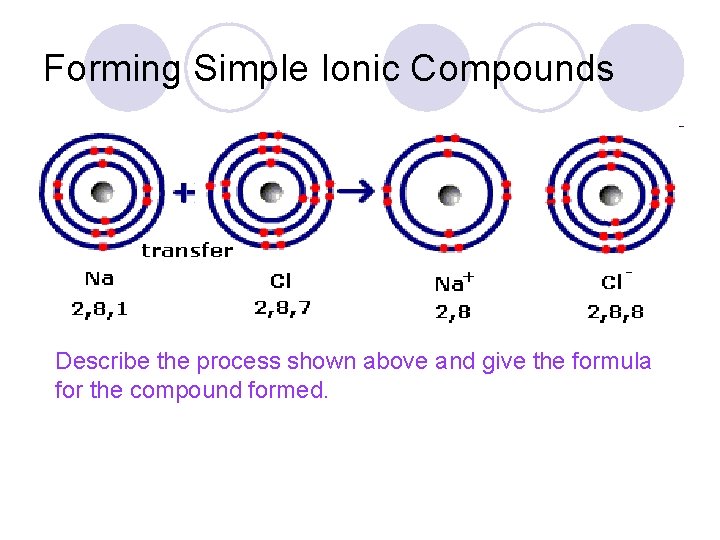
Forming Simple Ionic Compounds Describe the process shown above and give the formula for the compound formed.

Forming Simple Ionic Compounds l I want to find out the formula for the compound formed by potassium and sulfur. l I write their oxidation numbers. I know the compound must have a neutral charge. l It will take two +1 charges to balance out the 2 - charge of the sulfur. l I write the formula with the number of each type of atom written as a subscript. l I remove the charge from the compound. This is the formula for potassium sulfide.

Forming Simple Ionic Compounds l Notice that the positive ion (the element that loses electrons) is always listed first. l This method can only be used when one element forms positive ions and the other forms negative ions. l The negative ion always gets an –ide ending if it was formed by a single element. l This same method can be used for some compounds which are not, technically, ionic, but are formed by elements of which one has a positive oxidation number and the other has a negative oxidation number.

Forming Simple Ionic Compounds l We’ll use the criss-cross method to determine the formula for the compound formed by aluminum and oxygen l The oxidation numbers are added to each symbol. l As this combination is not obvious, we take each oxidation number and make it the subscript for the other element. l Those subscripts are used in the chemical formula, showing that for every 2 aluminum ions, three oxygen ions will be required. l The charge number is removed from the final formula. l 2 (+3) + 3 (-2) = 0

Forming Simple Ionic Compounds l You will be using your skills for determining ionic formulas for the rest of this course. l Polyatomic ions will make the process just a bit more complicated, but if you can do this, you can manage them, too. l You’ve had enough learning for now. Are you ready for a quiz?
- Slides: 24