Forming New Substances Chapter 9 1 Chemical Reactions

Forming New Substances Chapter 9 - 1

Chemical Reactions • A chemical reaction is a process in which one or more substances change to make one or more new substances. • The chemical and physical properties of the new substance are different from the original substances. + =

Signs of Chemical Reactions • Some reactions form precipitates, which are solid substances in a liquid. • Some other signs include gas production, color change, and energy given off in the form of light, heat, or electrical energy.

A Change of Properties • The most important sign of a chemical reaction is the formation of a new substance. • The properties of these new substances are very different from those of the starting substances.
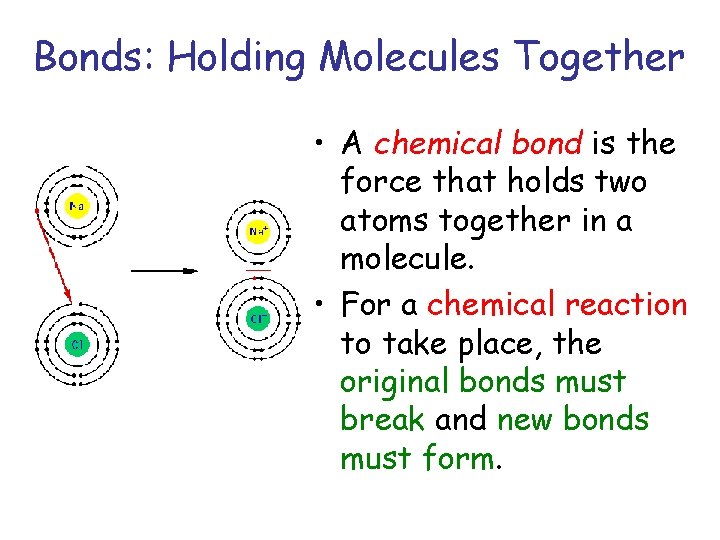
Bonds: Holding Molecules Together • A chemical bond is the force that holds two atoms together in a molecule. • For a chemical reaction to take place, the original bonds must break and new bonds must form.
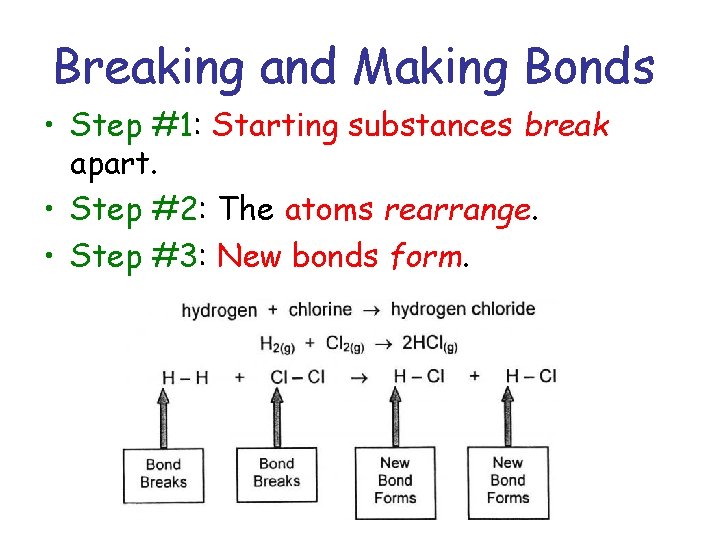
Breaking and Making Bonds • Step #1: Starting substances break apart. • Step #2: The atoms rearrange. • Step #3: New bonds form.

Reactions and Energy • Energy is needed to break chemical bonds in chemical reactions. • As new substances form, energy is released or absorbed.

Exothermic Reactions • A chemical reaction in which energy is released is called an exothermic reaction. • Exo means “go out” or “exit. ”

Endothermic Reaction • A chemical reaction in which energy is taken in is called an endothermic reaction. • The energy that is “taken in” is absorbed from the surrounding area, leaving it cold.

The Law of Conservation of Energy • The law of conservation of energy states that energy cannot be created or destroyed. • However, energy can change form and it can be transferred from one object to another.

THE END • LET’S TRY IT!
- Slides: 11