Forming Chemical Bonds and ions Compounds Remember compounds

Forming Chemical Bonds and ions

Compounds • Remember compounds are two or more different elements that combine chemically – Can be broken down by chemical means – Have properties different from those of its component element – Thousands of compounds exist

Chemical Bonds • A chemical bond is a force that holds two atoms together • Chemical bonds usually only involve valence electrons
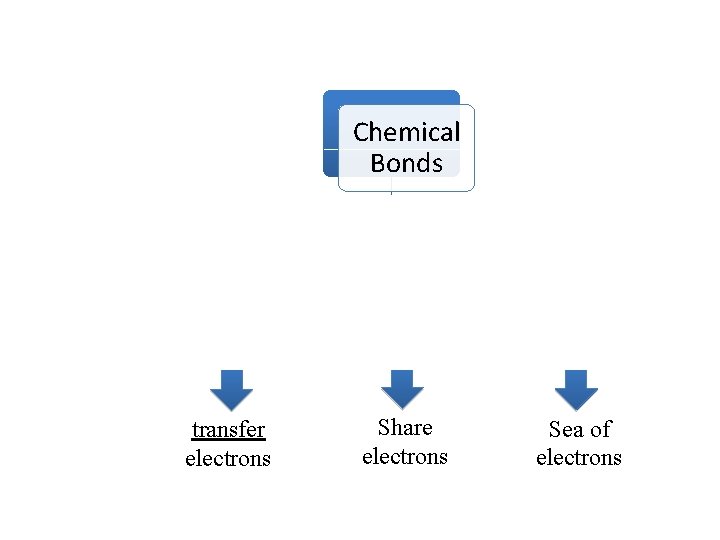
Chemical Bonds Ionic bonds transfer electrons Covalent bonds Metallic bonds Share electrons Sea of electrons

Valence Electrons • Valence electrons are electrons in the highest occupied s and p energy level. • The number of valence electrons largely determines the chemical properties of an element. • To find the number of valence electrons in a representative (group A) element, simply look at its group number.
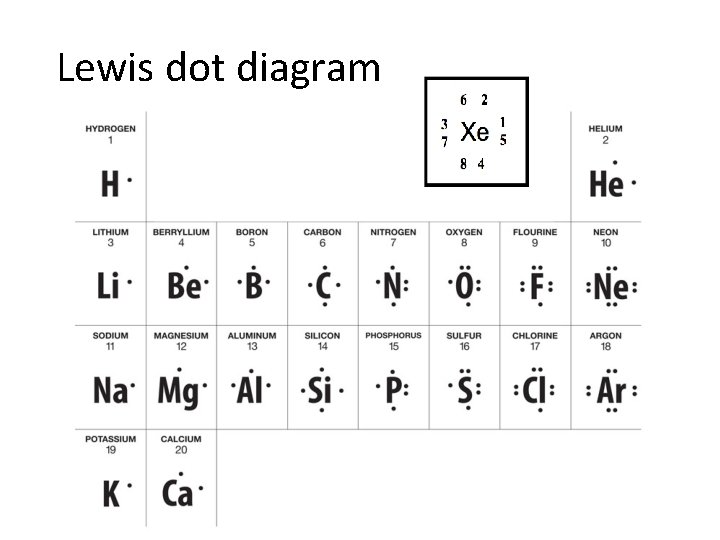
Lewis dot diagram • Diagrams that show valence electrons as dots

Why do elements react? • Noble Gases typically do not react and are chemically stable because they have eight valence electrons called an octet • Elements tend to react in order to acquire a full octet or “eight is great” (gain stability) • Octet rule –when forming compounds, atoms tend to achieve the stable electron configuration of a noble gas

Formation of Positive Ions-Cations • A cation is an atom or group of atoms with a positive charge because of a loss of valence electrons. Ca “+” ion • Metals only form cations • Reactivity of metals is based on the ease in which they lose a valence electron to achieve a stable octet. Brainiac Alkali Metals Video
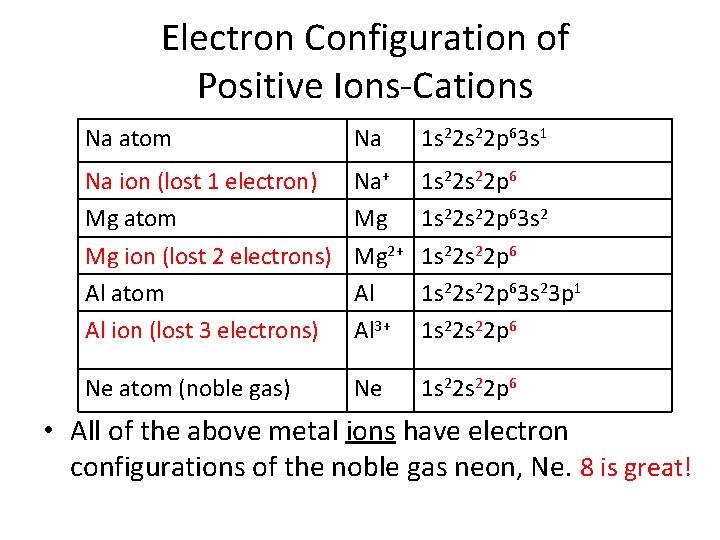
Electron Configuration of Positive Ions-Cations Na atom Na 1 s 22 p 63 s 1 Na ion (lost 1 electron) Mg atom Mg ion (lost 2 electrons) Al atom Al ion (lost 3 electrons) Na+ Mg Mg 2+ Al Al 3+ 1 s 22 s 22 p 63 s 23 p 1 1 s 22 p 6 Ne atom (noble gas) Ne 1 s 22 p 6 • All of the above metal ions have electron configurations of the noble gas neon, Ne. 8 is great!

Cation Charges • Group 1 A lose 1 electron, making a 1+ ion • Group 2 A loses 2 electrons, making a 2+ ion • Group 3 A loses 3 electrons, making a 3+ ion

Formation of Negative Ions-Anions • An anion is a negative atom or group of e atoms that gain electrons g a t i v e • Nonmetals tend to gain electrons

Electron Configuration of Negative Ions-”Anions” Cl atom Cl ion (gain 1 electron) S atom S ion (gain 2 electrons) Cl Cl. S S 2 - 1 s 22 p 63 s 23 p 5 1 s 22 p 63 s 23 p 6 1 s 22 p 63 s 23 p 4 1 s 22 p 63 s 23 p 6 Ar atom Ar 1 s 22 p 63 s 23 p 6 • Cl ion and S ion have the noble gas configuration of argon. 8 is great!

Anion Charges • 5 A tend to gain 3 electrons, making a 3 - ion • 6 A tend to gain 2 electrons, making a 2 - ion • 7 A tend to gain 1 electron, making a 1 - ion • The ion produced when atoms of halogens (group 7 A) gain electrons are called halide ions
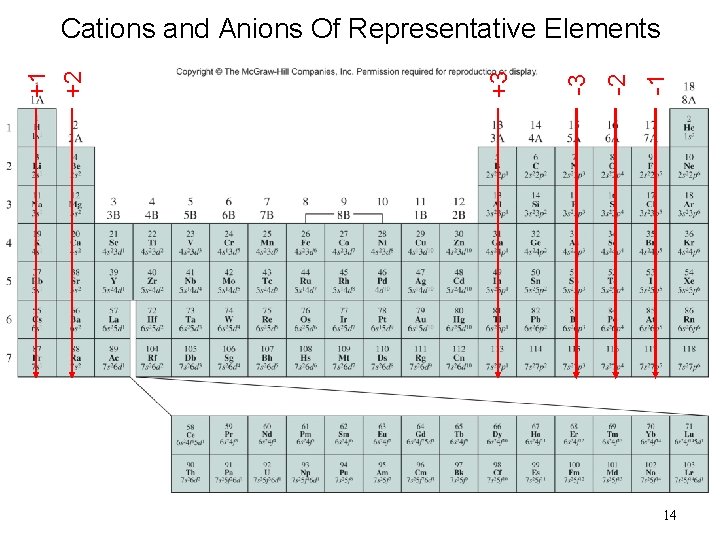
-1 -2 -3 +3 +1 +2 Cations and Anions Of Representative Elements 14

Cation is always smaller than atom from which it is formed. Anion is always larger than atom from which it is formed.
- Slides: 15