Formation of Styrene Carbonate from Styrene Oxide and

Formation of Styrene Carbonate from Styrene Oxide and CO 2 Honors Organic Chemistry Lab Spring Semester 2017 Dr. Deborah Lieberman & Dr. Allan Pinhas

Background to Honors Organic Lab Projects Synthesis of cyclic carbonates from epoxides and CO 2 provides tremendous promise for converting this hazardous waste into industrially useful raw materials Engineering plastics Cosmetics Polar solvents Honors Organic Lab investigates this synthesis of cyclic carbonates using variously substituted oxiranes and various catalysts Significant findings
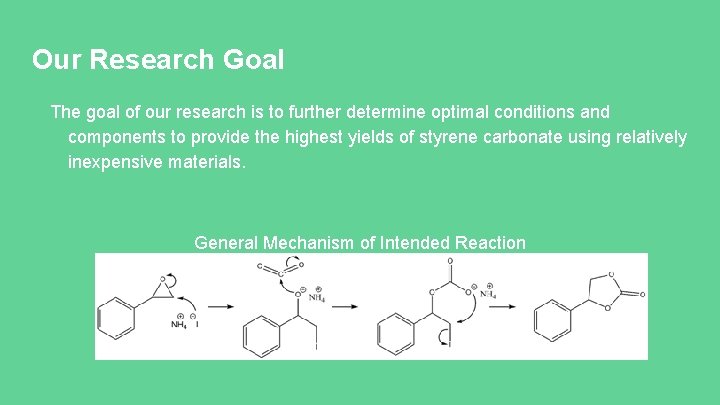
Our Research Goal The goal of our research is to further determine optimal conditions and components to provide the highest yields of styrene carbonate using relatively inexpensive materials. General Mechanism of Intended Reaction

Areas We Investigated Which iodide catalysts produce the highest yield of product Which bromide catalysts produce the highest yield of product How temperature is related to product formation Which proportions of reactants produce the highest yields Which solvent between THF, H 2 O, and no solvent works the best Which method of adding CO 2 works the best

Methods: GCMS Sample Preparation 1. Open steel vial noting if there is a hiss heard. 2. Run IR 3. Wash with diethyl ether until sample being retrieved is clear (about 3 pipets). 4. Remove bottom layer. a. If water, use pipet b. If salt, gravity filter 5. Place 3 -5 drops of sample into GCMS vial. 6. Fill GCMS vial to top line with isopropyl alcohol.

Which Iodide catalysts will produce the highest yield of product with and without H 2 O as a solvent? Erica Fastnacht and Sarah Han

Methods Each reaction was run under controlled conditions. The components were added to a steel vial in the following order: - Salt catalyst (0. 15 mmol) - ammonium iodide (NH 4), tetrabutylammonium iodide (TBAI), tetramethylammonium iodide (TMAI) - Either 1. 0 m. L of solvent (water) or no solvent - Styrene oxide (1 mmol) - 0. 1 g or 2. 27 mmol of CO 2 (dry ice) Vials (w/ O-rings) were capped tightly and left at room temperature for one week, unless otherwise noted.
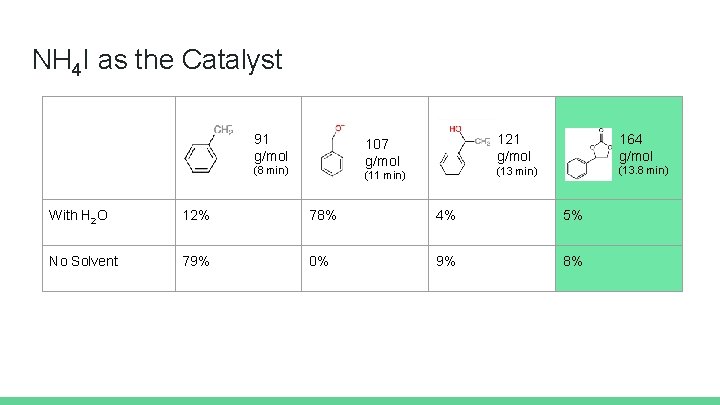
NH 4 I as the Catalyst 91 g/mol 107 g/mol (8 min) (11 min) 121 g/mol 164 g/mol (13 min) (13. 8 min) With H 2 O 12% 78% 4% 5% No Solvent 79% 0% 9% 8%
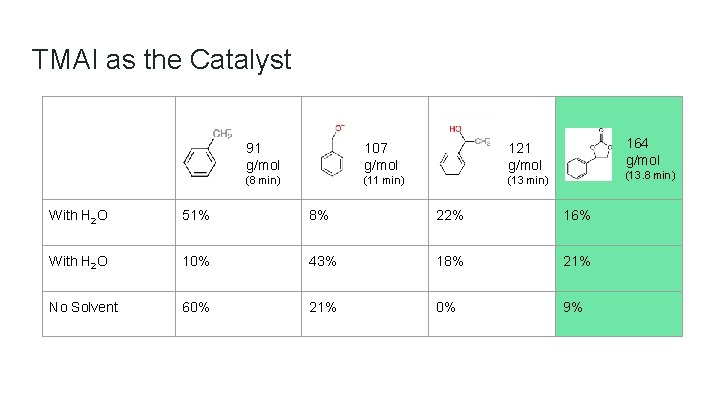
TMAI as the Catalyst 91 g/mol 107 g/mol 121 g/mol (8 min) (11 min) (13 min) 164 g/mol (13. 8 min) With H 2 O 51% 8% 22% 16% With H 2 O 10% 43% 18% 21% No Solvent 60% 21% 0% 9%
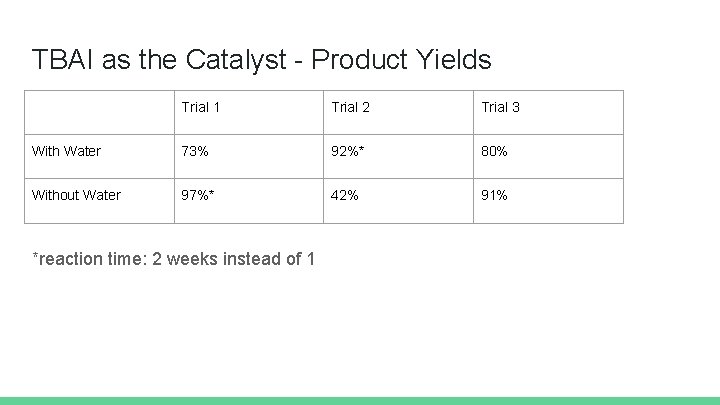
TBAI as the Catalyst - Product Yields Trial 1 Trial 2 Trial 3 With Water 73% 92%* 80% Without Water 97%* 42% 91% *reaction time: 2 weeks instead of 1
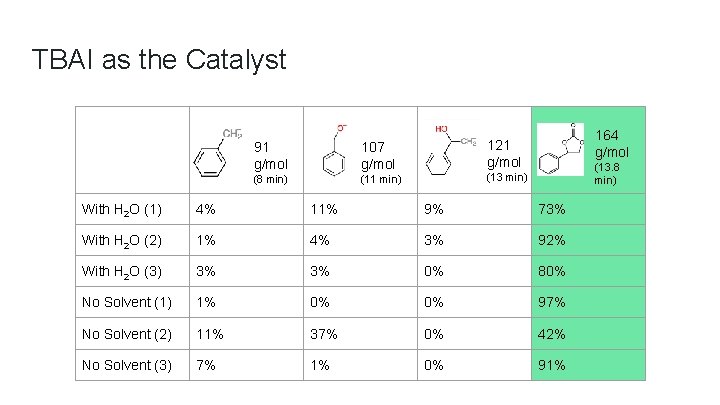
TBAI as the Catalyst 91 g/mol 107 g/mol (8 min) (11 min) 164 g/mol 121 g/mol (13. 8 min) (13 min) With H 2 O (1) 4% 11% 9% 73% With H 2 O (2) 1% 4% 3% 92% With H 2 O (3) 3% 3% 0% 80% No Solvent (1) 1% 0% 0% 97% No Solvent (2) 11% 37% 0% 42% No Solvent (3) 7% 1% 0% 91%

Conclusion ● TBAI worked the best as a catalyst for the formation of styrene carbonate from styrene oxide and CO 2 for the given reaction conditions, followed by TMAI and NH 4 I ● Results from the GCMS showed that the carbonate was the major product only when TBAI was used as the catalyst ● There is not enough data to conclude whether using water as a solvent has an effect on the yield of product

Possible Explanations - Solubility ● TBAI was visibly more soluble in styrene oxide than water ● NH 4 I was visibly more soluble in the water than the styrene oxide

Possible Explanation - Solubility

Possible Explanation - Solubility ● Since TBAI is the salt that is the most soluble in styrene oxide, it makes sense that it is the catalyst that produces the best yield, regardless of whether water is used ● NH 4 I and TMAI did not work as well because they are not as soluble in styrene oxide ○ When run in water, the salts dissolve in the water and do not come in contact with the styrene oxide, where the reaction is taking place ○ When run with no solvent, the salts still do not incorporate into the reaction because they do not become homogenous in the styrene oxide

Investigation of the Effects of Various Iodide Catalysts on Styrene Carbonate Formation in Tetrahydrofuran (THF) & Water (H 2 O) Alaina Werling and Andrew (Scottie) Emmert
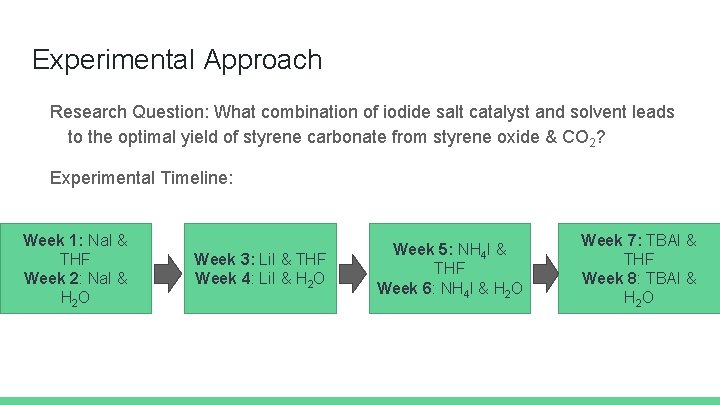
Experimental Approach Research Question: What combination of iodide salt catalyst and solvent leads to the optimal yield of styrene carbonate from styrene oxide & CO 2? Experimental Timeline: Week 1: Na. I & THF Week 2: Na. I & H 2 O Week 3: Li. I & THF Week 4: Li. I & H 2 O Week 5: NH 4 I & THF Week 6: NH 4 I & H 2 O Week 7: TBAI & THF Week 8: TBAI & H 2 O

Methods Reaction Preparation: All reactions were performed under controlled conditions in a steel vial containing the following reagents: Iodide salt (in 1: 1 molar equivalent with styrene oxide) Na. I, Li. I, NH 4 I, C 16 H 36 NI (TBAI) Styrene Oxide (100 u. L) Solvent (THF or H 2 O, 1000 u. L) CO 2 (extreme excess, “stuffed”)
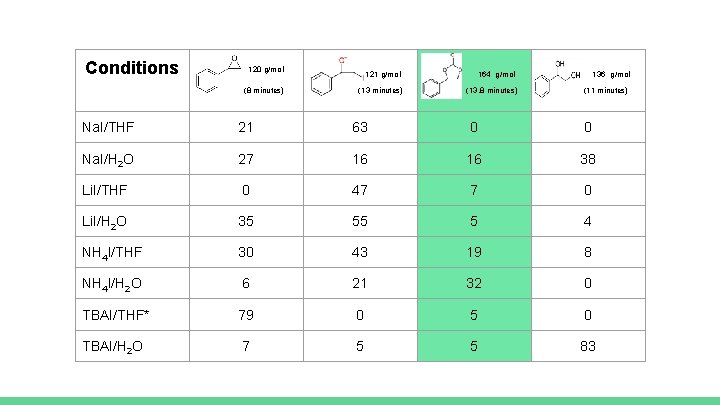
Conditions 120 g/mol (8 minutes) 121 g/mol (13 minutes) 164 g/mol (13. 8 minutes) 136 g/mol (11 minutes) Na. I/THF 21 63 0 0 Na. I/H 2 O 27 16 16 38 Li. I/THF 0 47 7 0 Li. I/H 2 O 35 55 5 4 NH 4 I/THF 30 43 19 8 NH 4 I/H 2 O 6 21 32 0 TBAI/THF* 79 0 5 0 TBAI/H 2 O 7 5 5 83
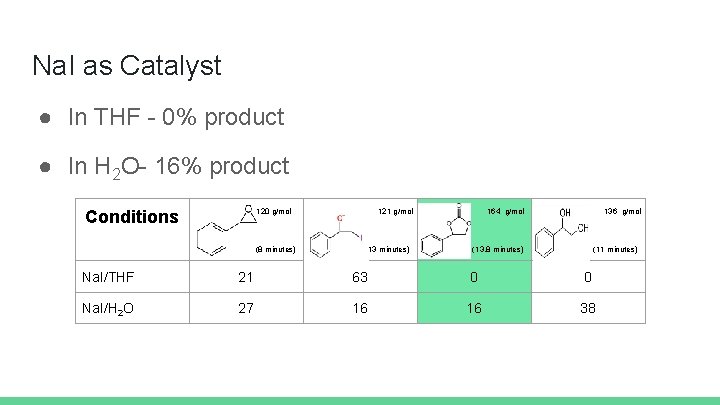
Na. I as Catalyst ● In THF - 0% product ● In H 2 O- 16% product 120 g/mol Conditions (8 minutes) 121 g/mol (13 minutes) 164 g/mol 136 g/mol (13. 8 minutes) (11 minutes) Na. I/THF 21 63 0 0 Na. I/H 2 O 27 16 16 38
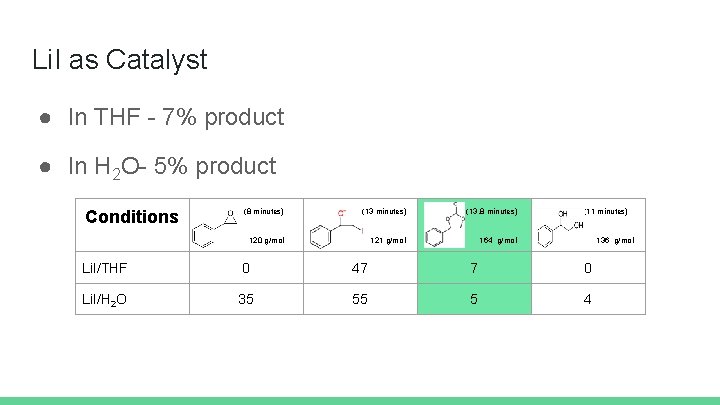
Li. I as Catalyst ● In THF - 7% product ● In H 2 O- 5% product Conditions (8 minutes) (13. 8 minutes) 120 g/mol 121 g/mol 164 g/mol (11 minutes) 136 g/mol Li. I/THF 0 47 7 0 Li. I/H 2 O 35 55 5 4
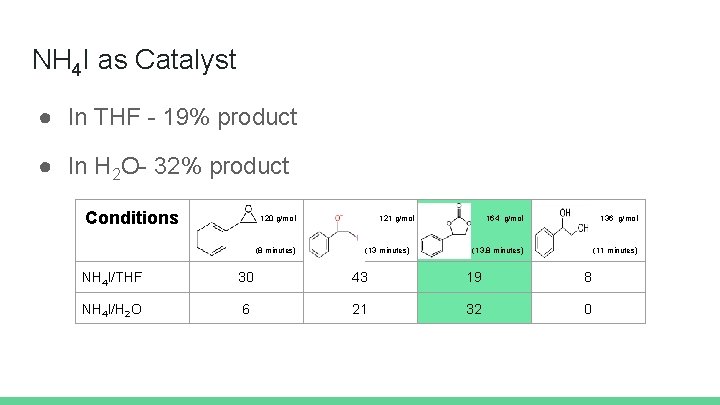
NH 4 I as Catalyst ● In THF - 19% product ● In H 2 O- 32% product Conditions 120 g/mol (8 minutes) 121 g/mol (13 minutes) 164 g/mol 136 g/mol (13. 8 minutes) (11 minutes) NH 4 I/THF 30 43 19 8 NH 4 I/H 2 O 6 21 32 0
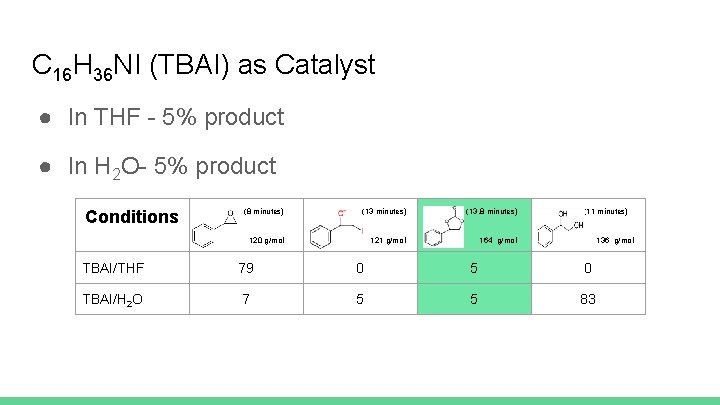
C 16 H 36 NI (TBAI) as Catalyst ● In THF - 5% product ● In H 2 O- 5% product Conditions (8 minutes) (13. 8 minutes) 120 g/mol 121 g/mol 164 g/mol (11 minutes) 136 g/mol TBAI/THF 79 0 5 0 TBAI/H 2 O 7 5 5 83
Conclusions Extreme excess of CO 2 (i. e. , packing vial with dry ice) possible culprit of low styrene carbonate yields for time-tested salts TBAI/H 2 O reaction yields Erica & Sarah: 80% yield Alberta & Divya: 71% Alaina & Scottie: 5% Iodide salts involving alkali and alkali earth cations (e. g. , Na+ and Li 2+) prevent ring closure due to strong, covalent character of oxygen-cation bond Ring opens, but then sits there unable to react.
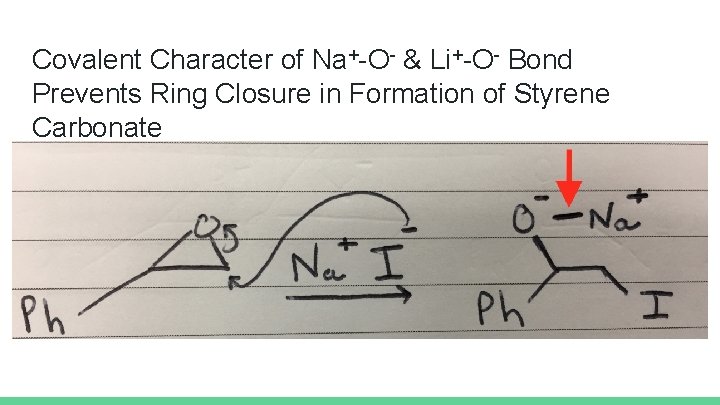
Covalent Character of Na+-O- & Li+-O- Bond Prevents Ring Closure in Formation of Styrene Carbonate

Does THF as a solvent improve product yield? Alberta Negri and Divya Takkellapati

Methods ● Reaction Preparation: ○ Wash and dry steel vial, making sure to replace o-ring for each trial ○ Add reagents ■ 1 mmol of salt (TBAI or TEAI) ■ 114 u. L of styrene oxide ■ Approximately. 5 g of dry ice ■ If applicable, 1 m. L of solvent (H 2 O, THF, or dry THF) ○ Close vial immediately, using vice grip to ensure tightest seal
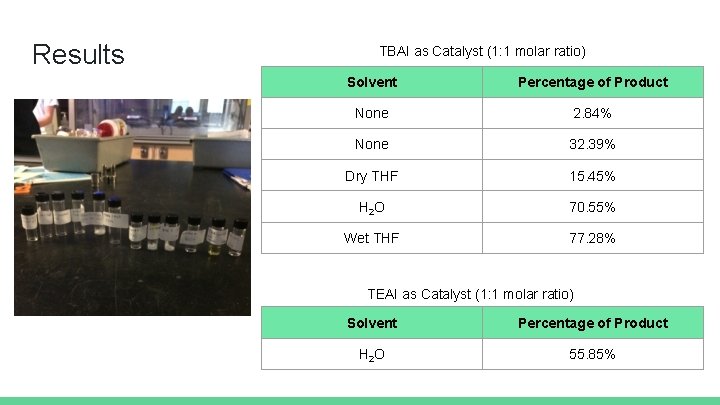
Results TBAI as Catalyst (1: 1 molar ratio) Solvent Percentage of Product None 2. 84% None 32. 39% Dry THF 15. 45% H 2 O 70. 55% Wet THF 77. 28% TEAI as Catalyst (1: 1 molar ratio) Solvent Percentage of Product H 2 O 55. 85%

Conclusions Initial results hinted THF might be an effective solvent (wet THF trial) Further trials with dry THF and H 2 O support that THF alone does noticeably increase product Product yields were highest for reactions with H 2 O as yield solvent TBAI is more effective salt than TEAI Bulkiness of catalyst may play a role Important to replace O-rings after every trial Glass vials should not be stuffed with dry ice

Role of Carbon Dioxide’s Method of Addition in Styrene Carbonate Formation Kyle Necamp and Nate Ranly
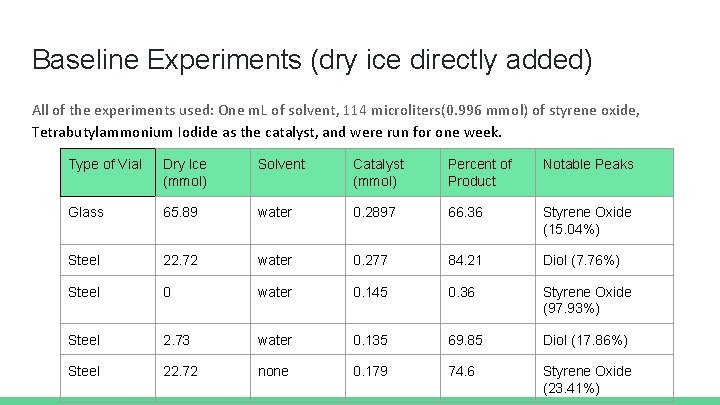
Baseline Experiments (dry ice directly added) All of the experiments used: One m. L of solvent, 114 microliters(0. 996 mmol) of styrene oxide, Tetrabutylammonium Iodide as the catalyst, and were run for one week. Type of Vial Dry Ice (mmol) Solvent Catalyst (mmol) Percent of Product Notable Peaks Glass 65. 89 water 0. 2897 66. 36 Styrene Oxide (15. 04%) Steel 22. 72 water 0. 277 84. 21 Diol (7. 76%) Steel 0 water 0. 145 0. 36 Styrene Oxide (97. 93%) Steel 2. 73 water 0. 135 69. 85 Diol (17. 86%) Steel 22. 72 none 0. 179 74. 6 Styrene Oxide (23. 41%)
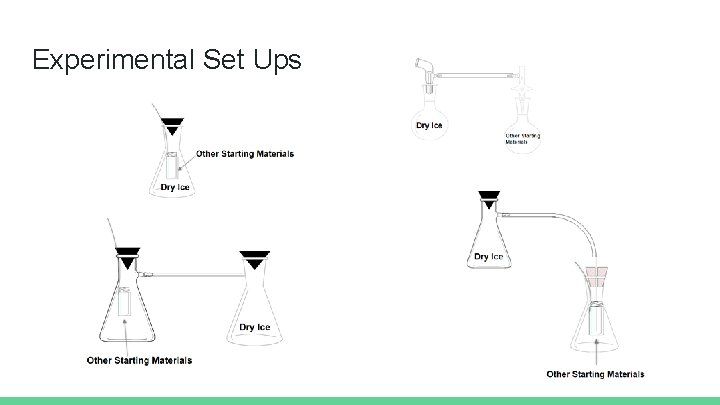
Experimental Set Ups
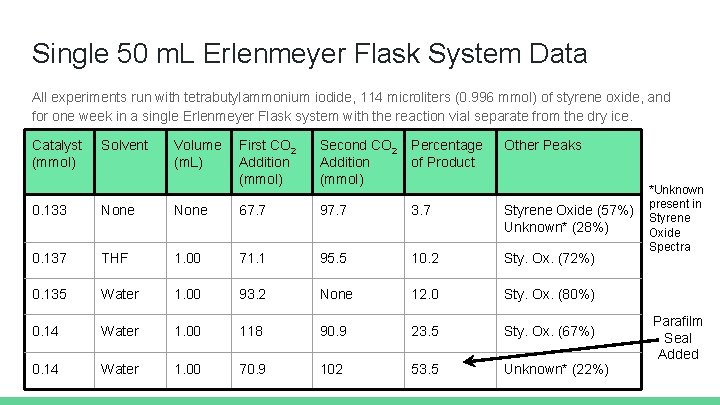
Single 50 m. L Erlenmeyer Flask System Data All experiments run with tetrabutylammonium iodide, 114 microliters (0. 996 mmol) of styrene oxide, and for one week in a single Erlenmeyer Flask system with the reaction vial separate from the dry ice. Catalyst (mmol) Solvent Volume (m. L) First CO 2 Addition (mmol) Second CO 2 Addition (mmol) Percentage of Product Other Peaks 0. 133 None 67. 7 97. 7 3. 7 Styrene Oxide (57%) Unknown* (28%) 0. 137 THF 1. 00 71. 1 95. 5 10. 2 Sty. Ox. (72%) 0. 135 Water 1. 00 93. 2 None 12. 0 Sty. Ox. (80%) 0. 14 Water 1. 00 118 90. 9 23. 5 Sty. Ox. (67%) 0. 14 Water 1. 00 70. 9 102 53. 5 Unknown* (22%) *Unknown present in Styrene Oxide Spectra Parafilm Seal Added
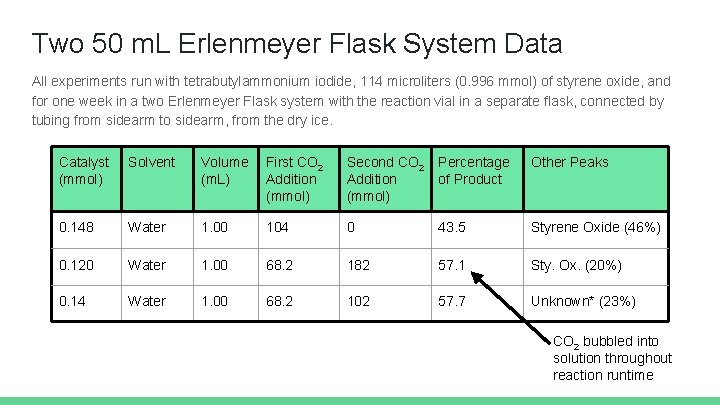
Two 50 m. L Erlenmeyer Flask System Data All experiments run with tetrabutylammonium iodide, 114 microliters (0. 996 mmol) of styrene oxide, and for one week in a two Erlenmeyer Flask system with the reaction vial in a separate flask, connected by tubing from sidearm to sidearm, from the dry ice. Catalyst (mmol) Solvent Volume (m. L) First CO 2 Addition (mmol) Second CO 2 Addition (mmol) Percentage of Product Other Peaks 0. 148 Water 1. 00 104 0 43. 5 Styrene Oxide (46%) 0. 120 Water 1. 00 68. 2 182 57. 1 Sty. Ox. (20%) 0. 14 Water 1. 00 68. 2 102 57. 7 Unknown* (23%) CO 2 bubbled into solution throughout reaction runtime
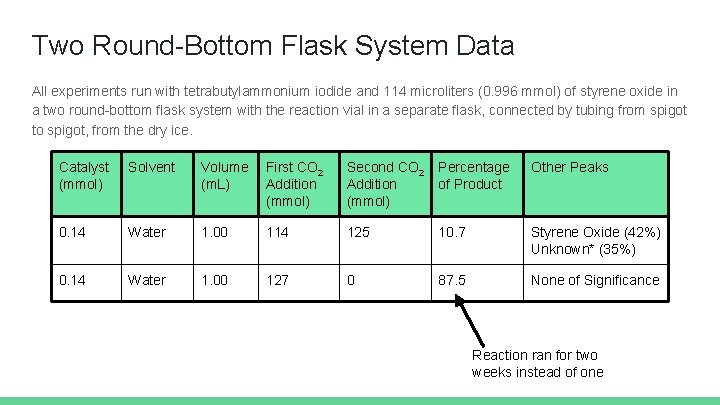
Two Round-Bottom Flask System Data All experiments run with tetrabutylammonium iodide and 114 microliters (0. 996 mmol) of styrene oxide in a two round-bottom flask system with the reaction vial in a separate flask, connected by tubing from spigot to spigot, from the dry ice. Catalyst (mmol) Solvent Volume (m. L) First CO 2 Addition (mmol) Second CO 2 Addition (mmol) Percentage of Product Other Peaks 0. 14 Water 1. 00 114 125 10. 7 Styrene Oxide (42%) Unknown* (35%) 0. 14 Water 1. 00 127 0 87. 5 None of Significance Reaction ran for two weeks instead of one

Conclusions ● Adding dry ice directly seems to be more effective. ● The length of time the reaction is run should be investigated further. ● The biggest problem was possible leaks in the set up. ● Bubbling carbon dioxide directly into the reactants could be further investigated.

Effects of Bromide Catalysts on Styrene Carbonate Formation Sam Blizzard and Jumee Park

Setup Except where noted, all reactions: Used 13. 75 mmol (250 micro. L) of water as solvent Used 1 mmol (. 44 g) o f CO 2 Used 1 mmol (114 micro. L) of styrene oxide Conducted at room temperature (293 K) Conducted in steel vial Conducted for one week
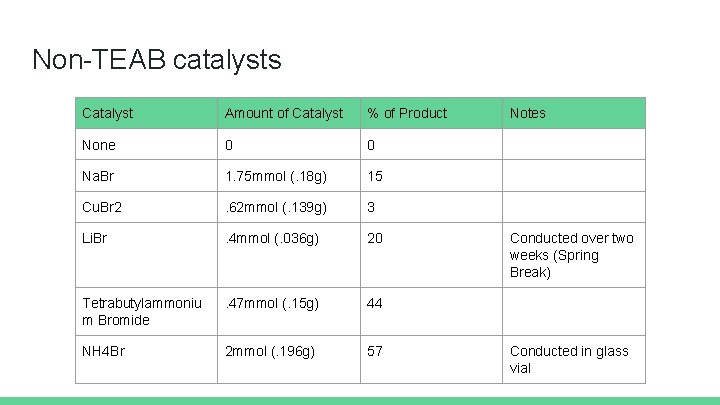
Non-TEAB catalysts Catalyst Amount of Catalyst % of Product None 0 0 Na. Br 1. 75 mmol (. 18 g) 15 Cu. Br 2 . 62 mmol (. 139 g) 3 Li. Br . 4 mmol (. 036 g) 20 Tetrabutylammoniu m Bromide . 47 mmol (. 15 g) 44 NH 4 Br 2 mmol (. 196 g) 57 Notes Conducted over two weeks (Spring Break) Conducted in glass vial
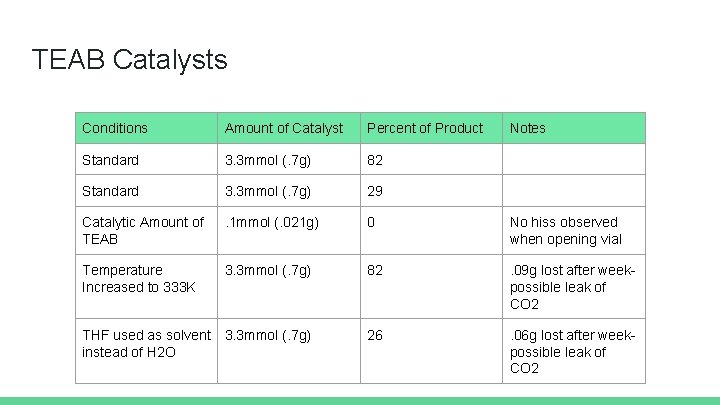
TEAB Catalysts Conditions Amount of Catalyst Percent of Product Notes Standard 3. 3 mmol (. 7 g) 82 Standard 3. 3 mmol (. 7 g) 29 Catalytic Amount of TEAB . 1 mmol (. 021 g) 0 No hiss observed when opening vial Temperature Increased to 333 K 3. 3 mmol (. 7 g) 82 . 09 g lost after weekpossible leak of CO 2 THF used as solvent instead of H 2 O 3. 3 mmol (. 7 g) 26 . 06 g lost after weekpossible leak of CO 2
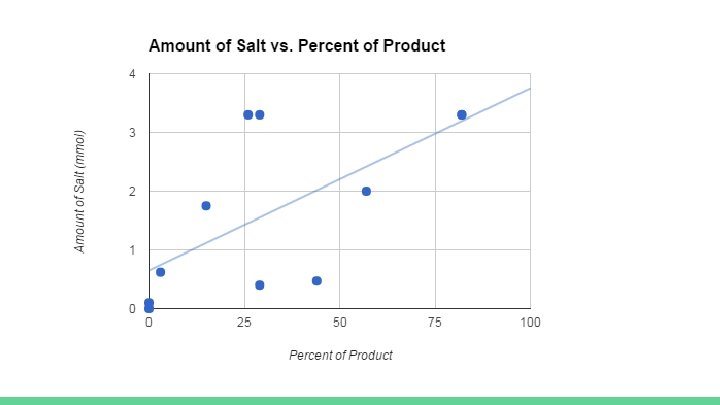

Conclusions Amount of salt Positive linear trend noticed, so standardize across experiments and use catalytic amounts for practical purposes Loss of CO 2 noticed on some trials Look for avenues of experimentation that guarantee tighter seals TEAB generally stronger than other catalysts More experimentation with catalytic amounts Examine role of solubility of salts in water vs. styrene oxide Temperature could have effect

Effects of Temperature on Styrene Carbonate Formation Dean Hayes and Jordan Hill

Background Question: Does temperature of the system significantly alter the amount of product obtained in a styrene oxide to styrene carbonate reaction? Experimental set-up Salt: TBAI (0. 27 mmol) CO 2: 1. 2 grams (27 mmol, excess) Starting material: 100 u. L (0. 87 mmol) Solvent: water (55 mmol, 1 m. L)
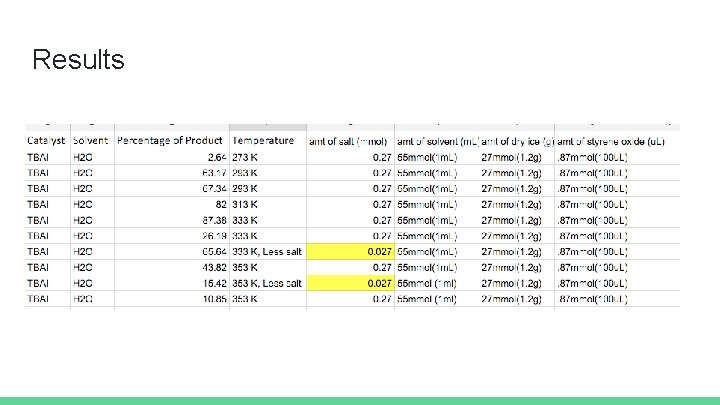
Results
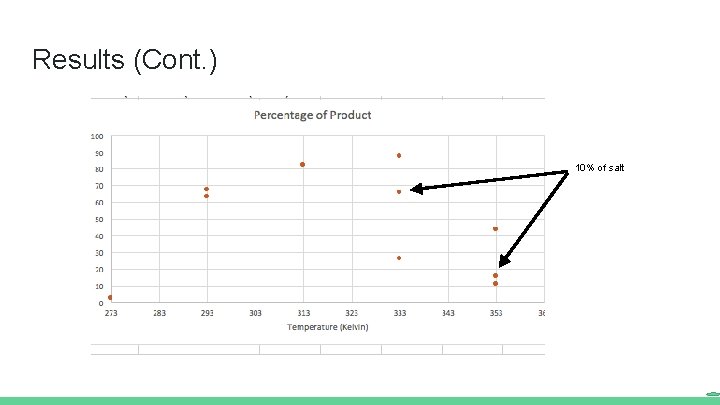
Results (Cont. ) 10% of salt
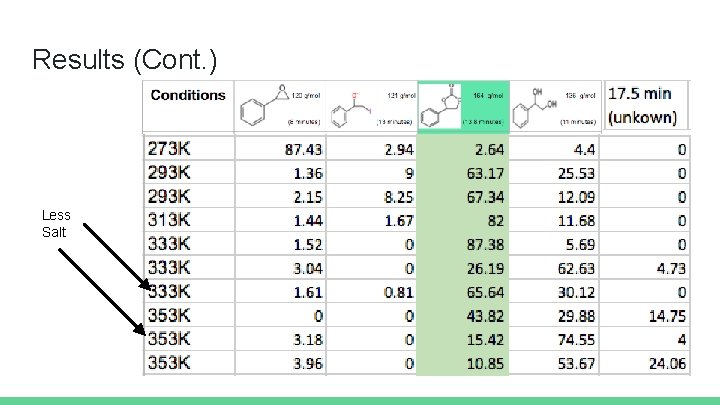
Results (Cont. ) Less Salt

Conclusions Temperature has a significant effect on the amount of product obtained Increasing temperature increased desired product up to 333 K At 333 K and above, results were inconsistent and undesired products increased in prevalence Parabolic pattern of product formation possible with temperature change Decreasing the amount of salt appeared to inhibit product formation In order to obtain more concrete conclusions, all the experiments should be repeated several times

Overall Conclusions Concentration of CO 2 used is not linearly correlated (the more you use does not mean the more yield you will get), but an optimal concentration does exist for this reaction. Water is a more effective solvent than THF Varying results for no solvent - could be due to different quantities of salt or CO 2 TBAI was frequently found to be the most effective catalyst The procedure used led to inconsistent results in some cases Same exact conditions led to different product yield

Future Directions Determine optimal amount of CO 2 and catalyst to be used for maximum yields. Further investigate relationship between solubility of catalyst in styrene oxide/solvent and product yield Standardize reaction preparation amounts for all materials amongst groups participating in study. Quantify product yields beyond GCMS percentages. Test how reaction runtime affects yield. Modify experiment methods to increase the reproducibility of trials.
- Slides: 50