Formation of Light Ground State Excited State Electron












- Slides: 39

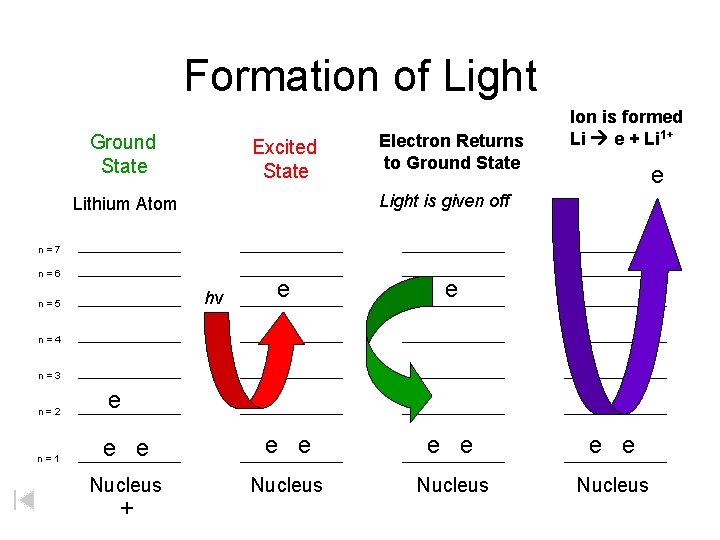
Formation of Light Ground State Excited State Electron Returns to Ground State Ion is formed Li e + Li 1+ e Light is given off Lithium Atom n=7 n=6 e e e e e Nucleus hv n=5 n=4 n=3 n=2 n=1 e +

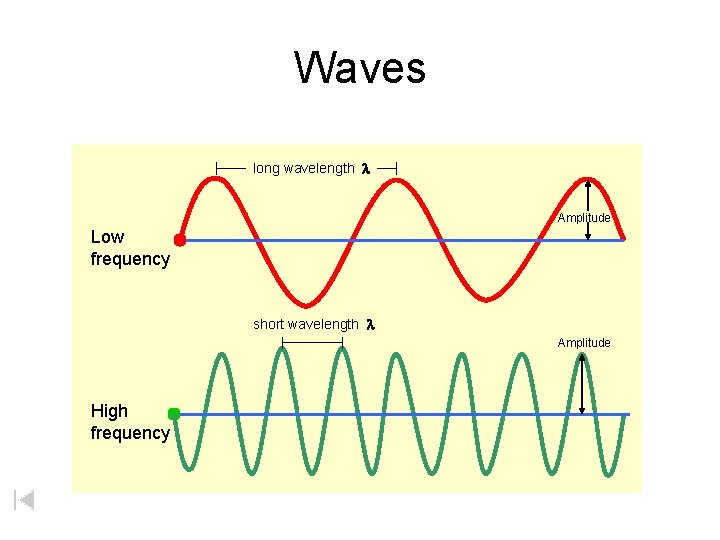
Waves • Wavelength ( ) - length of one complete wave • Frequency ( ) f - # of waves that pass a point during a certain time period – hertz (Hz) = 1/s • Amplitude (A) - distance from the origin to the trough or crest Courtesy Christy Johannesson www. nisd. net/communicationsarts/pages/chem

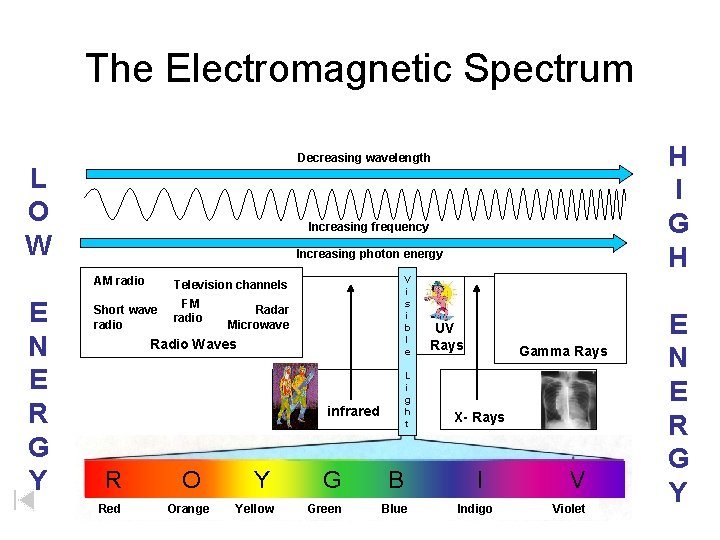
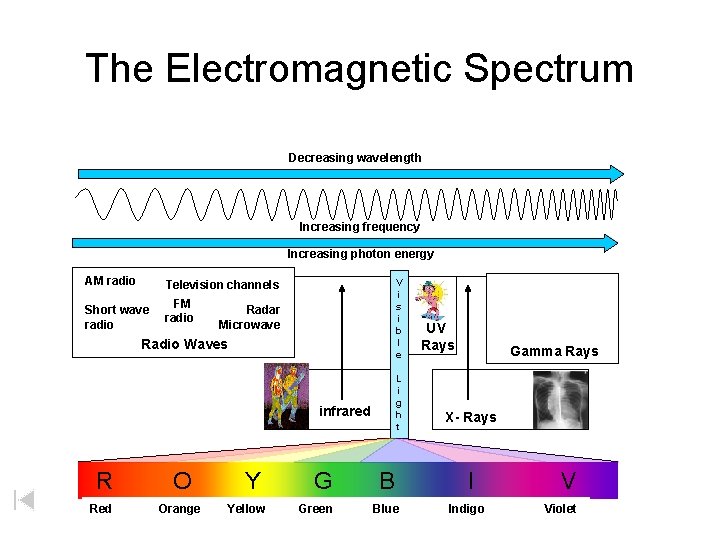
The Electromagnetic Spectrum L O W Increasing frequency Increasing photon energy AM radio E N E R G Y H I G H Decreasing wavelength V i s i b l e Television channels Short wave radio FM radio Radar Microwave Radio Waves L i g h t infrared R O Red Orange Y Yellow G Green B Blue UV Rays Gamma Rays X- Rays I Indigo V Violet E N E R G Y

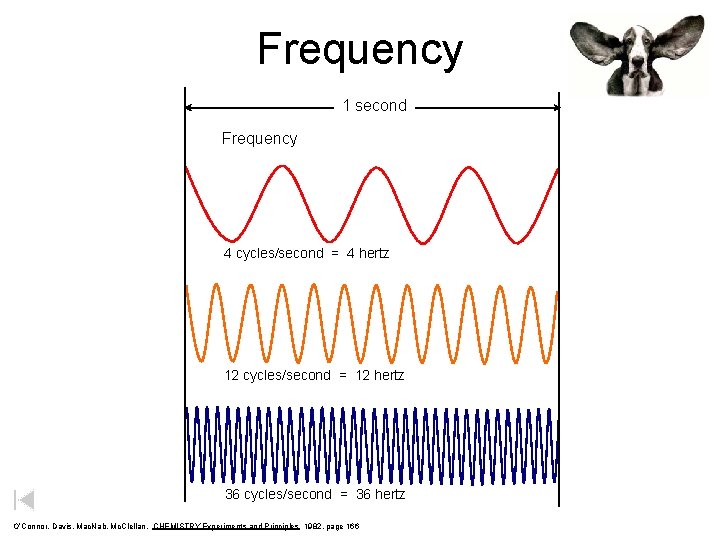
Frequency 1 second Frequency 4 cycles/second = 4 hertz 12 cycles/second = 12 hertz 36 cycles/second = 36 hertz O’Connor, Davis, Mac. Nab, Mc. Clellan, CHEMISTRY Experiments and Principles 1982, page 166

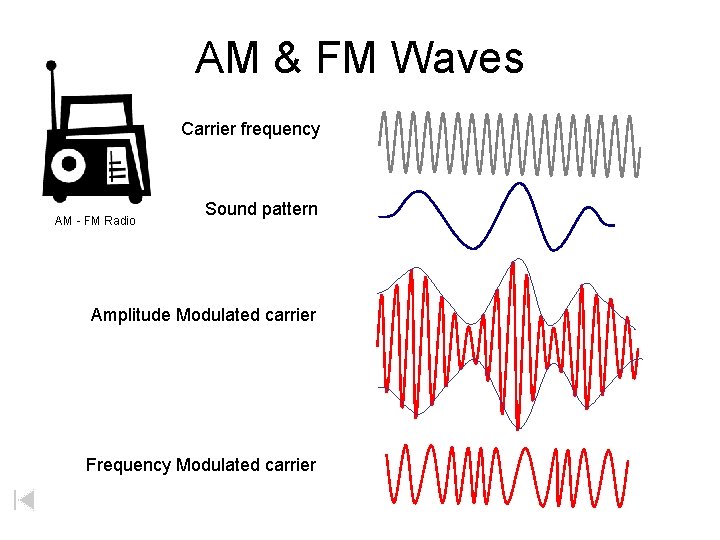
AM & FM Waves Carrier frequency AM - FM Radio Sound pattern Amplitude Modulated carrier Frequency Modulated carrier

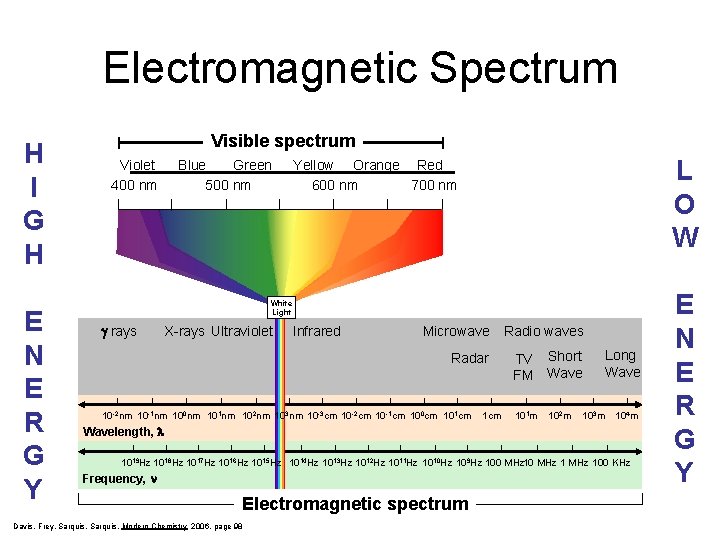
Electromagnetic Spectrum H I G H E N E R G Y Visible spectrum Violet 400 nm Blue Green 500 nm L O W Yellow Orange Red 600 nm 700 nm White Light g rays X-rays Ultraviolet Infrared Microwave Radio waves Radar TV Short FM Wave 10 -2 nm 10 -1 nm 100 nm 101 nm 102 nm 103 nm 10 -3 cm 10 -2 cm 10 -1 cm 100 cm 101 cm 101 m 102 m 103 m Long Wave 104 m Wavelength, l 1019 Hz 1018 Hz 1017 Hz 1016 Hz 1015 Hz 1014 Hz 1013 Hz 1012 Hz 1011 Hz 1010 Hz 109 Hz 100 MHz 100 KHz Frequency, Electromagnetic spectrum Davis, Frey, Sarquis, Modern Chemistry 2006, page 98 E N E R G Y

Waves long wavelength l Amplitude Low frequency short wavelength l Amplitude High frequency

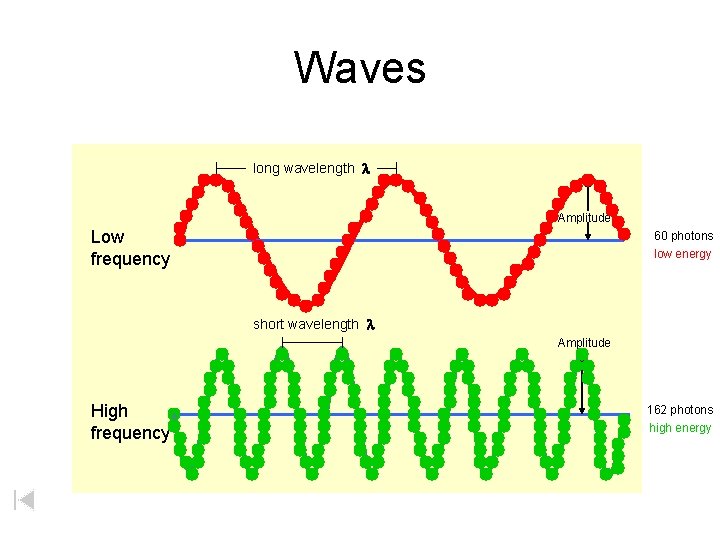
Waves long wavelength l Amplitude Low frequency 60 photons low energy short wavelength l Amplitude High frequency 162 photons high energy

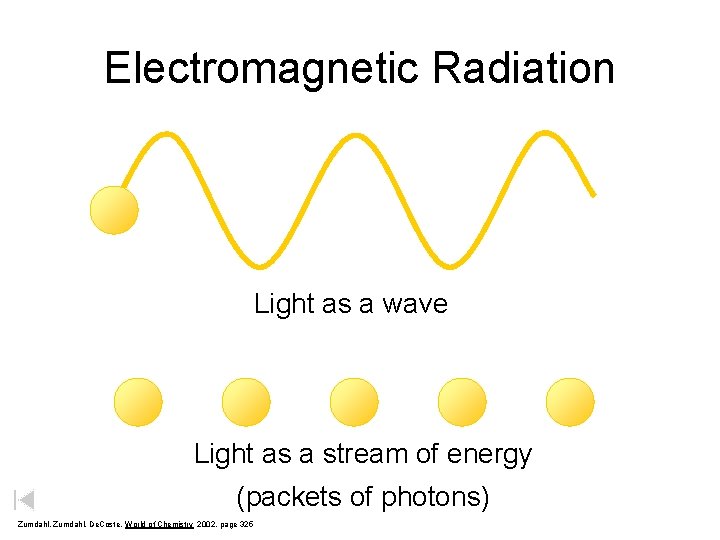
Electromagnetic Radiation Light as a wave Light as a stream of energy (packets of photons) Zumdahl, De. Coste, World of Chemistry 2002, page 325

Wavelength and Frequency “nu” “lambda” c = f E = h f c = speed of light (3 x 108 m/s) f = frequency (s-1) = wavelength (m) E = energy (Joules or J) h = Planck’s constant (6. 6 x 10 -34 J/s) f = frequency (s-1)

Electromagnetic Spectrum • EX: Find the frequency of a photon with a wavelength of 434 nm. GIVEN: f=? = 434 nm WORK: 1 m 1 x 109 nm = 4. 34 10 -7 m c = 3. 00 108 m/s f = 3. 00 108 m/s 4. 34 10 -7 m f = 6. 91 1014 Hz Courtesy Christy Johannesson www. nisd. net/communicationsarts/pages/chem

Coins are Quantified Do we have a coin for 37 cents? Light is also quantified and only comes in certain bundles called photons.

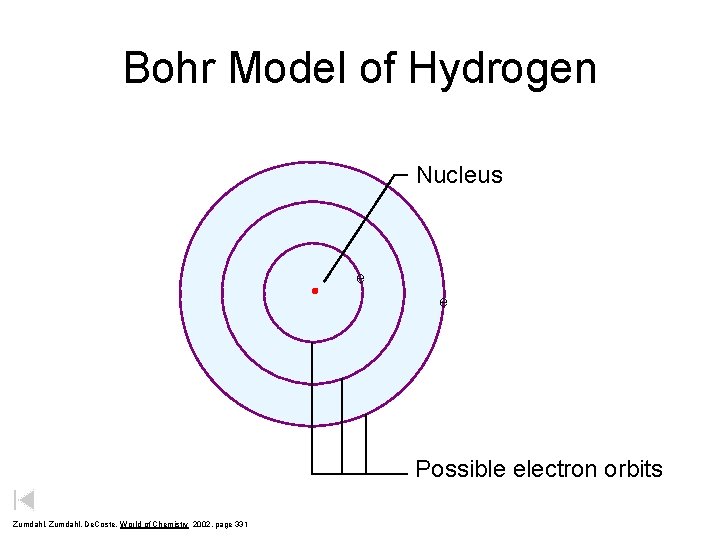
Bohr Model of Hydrogen Nucleus e e Possible electron orbits Zumdahl, De. Coste, World of Chemistry 2002, page 331

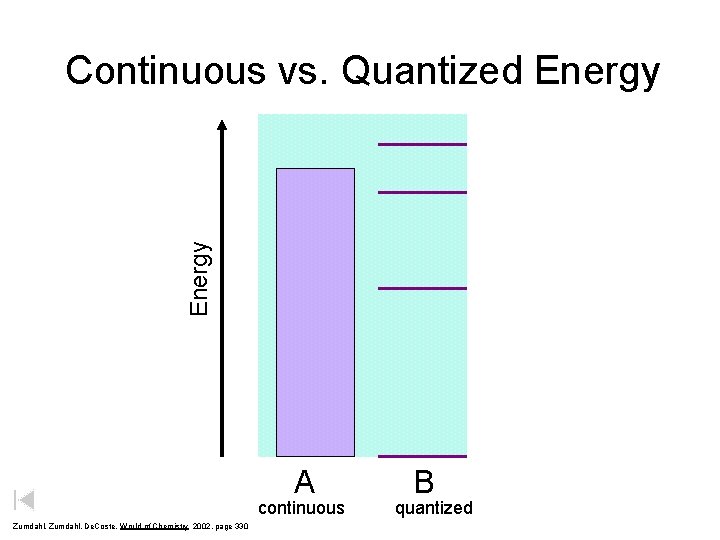
Energy Continuous vs. Quantized Energy A continuous Zumdahl, De. Coste, World of Chemistry 2002, page 330 B quantized

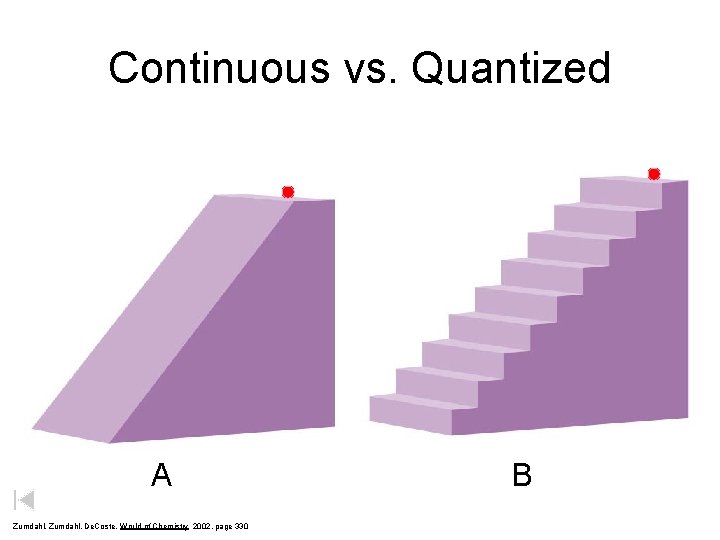
Continuous vs. Quantized A Zumdahl, De. Coste, World of Chemistry 2002, page 330 B

Wavelength and Frequency “nu” “lambda” c = f E = h f c = speed of light (3 x 108 m/s) f = frequency (s-1) = wavelength (m) E = energy (Joules or J) h = Planck’s constant (6. 6 x 10 -34 J/s) f = frequency (s-1)

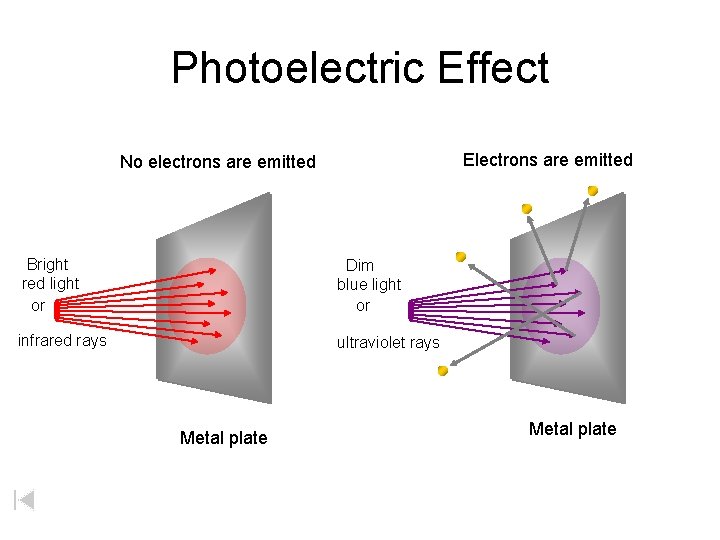
Photoelectric Effect Electrons are emitted No electrons are emitted Bright red light or Dim blue light or infrared rays ultraviolet rays Metal plate

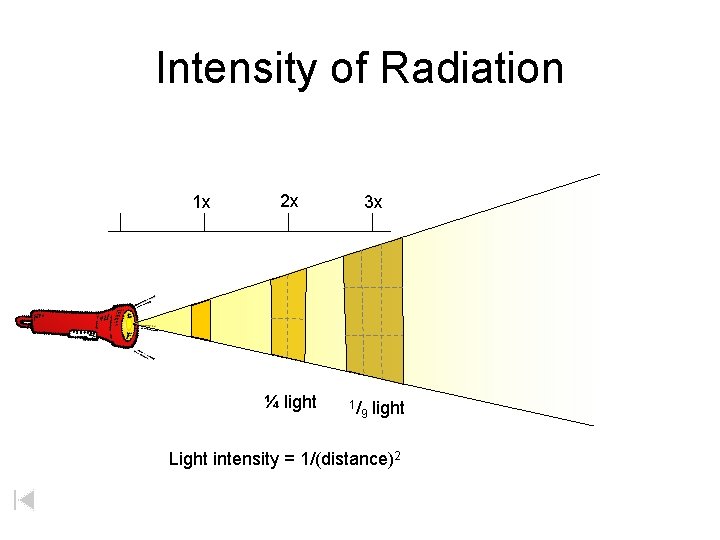
Intensity of Radiation 1 x 2 x ¼ light 3 x 1/ 9 light Light intensity = 1/(distance)2

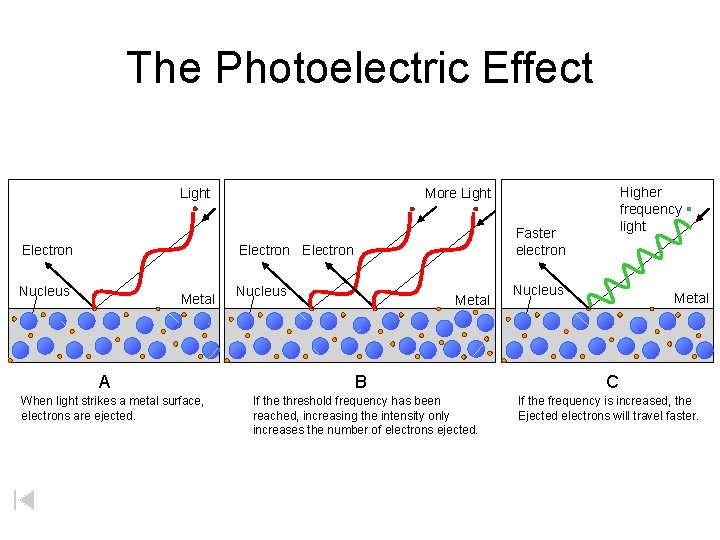
The Photoelectric Effect Light Electron Faster electron Electron Nucleus Metal A When light strikes a metal surface, electrons are ejected. Higher frequency light More Light Nucleus Metal B If the threshold frequency has been reached, increasing the intensity only increases the number of electrons ejected. Nucleus Metal C If the frequency is increased, the Ejected electrons will travel faster.

Photoelectric Effect Some key results For low frequency light (low energy, below threshold) - electrons are not ejected regardless of the light’s intensity (number of light waves) For high frequency light (high energy, above threshold) - same number of electrons are ejected regardless of the frequency (energy of light waves) - Increasing the light’s intensity increases the number of electrons ejected (increases the current) Einstein considered light as mass-less, energetic particles (photons) to help explain this effect – energy follows Planck’s equation.

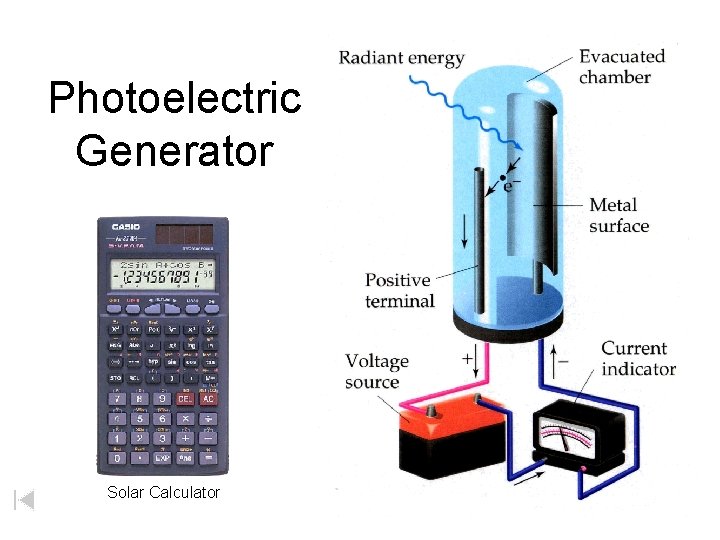
Photoelectric Generator Solar Calculator

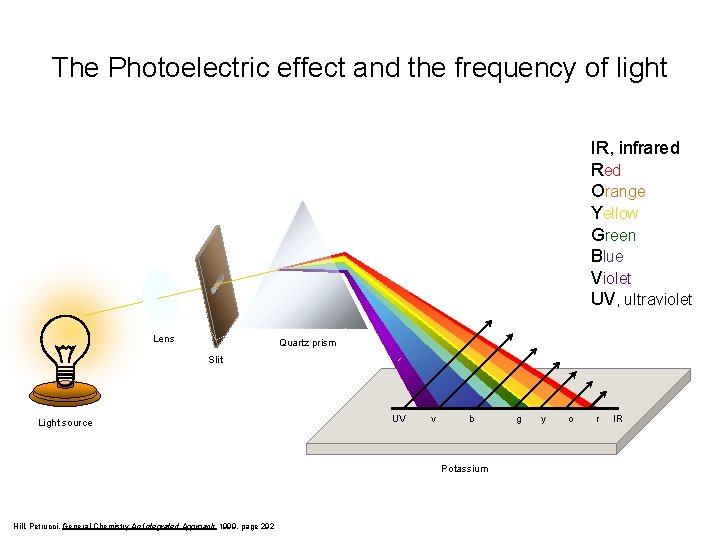
The Photoelectric effect and the frequency of light IR, infrared Red Orange Yellow Green Blue Violet UV, ultraviolet Lens Quartz prism Slit Light source UV v b Potassium Hill, Petrucci, General Chemistry An Integrated Approach 1999, page 292 g y o r IR

Quantum Theory • The energy of a photon is proportional to its frequency. E = h E: energy (J, joules) h: Planck’s constant (6. 6262 10 -34 J·s) : frequency (Hz) Courtesy Christy Johannesson www. nisd. net/communicationsarts/pages/chem

Quantum Theory • Example: Find the energy of a red photon with a frequency of 4. 57 1014 Hz. GIVEN: E=? = 4. 57 1014 Hz h = 6. 6262 10 -34 J· s WORK: E = h E = (6. 6262 10 -34 J· s) (4. 57 1014 Hz) E = 3. 03 10 -19 J Courtesy Christy Johannesson www. nisd. net/communicationsarts/pages/chem

Color = Energy of Photons Zumdahl, De. Coste, World of Chemistry 2002, page 329

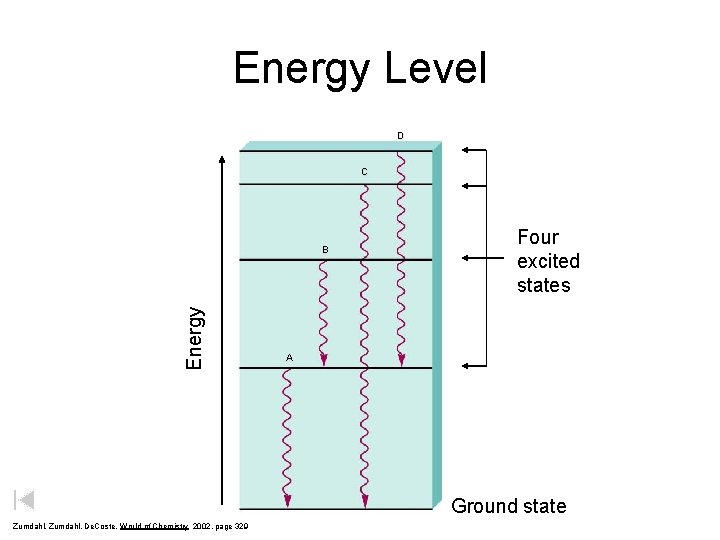
Energy Level D C Energy B Four excited states A Ground state Zumdahl, De. Coste, World of Chemistry 2002, page 329

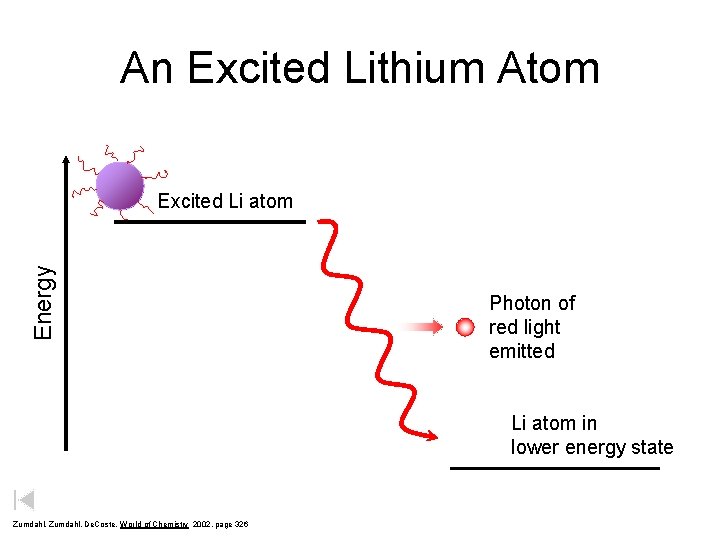
An Excited Lithium Atom Energy Excited Li atom Photon of red light emitted Li atom in lower energy state Zumdahl, De. Coste, World of Chemistry 2002, page 326

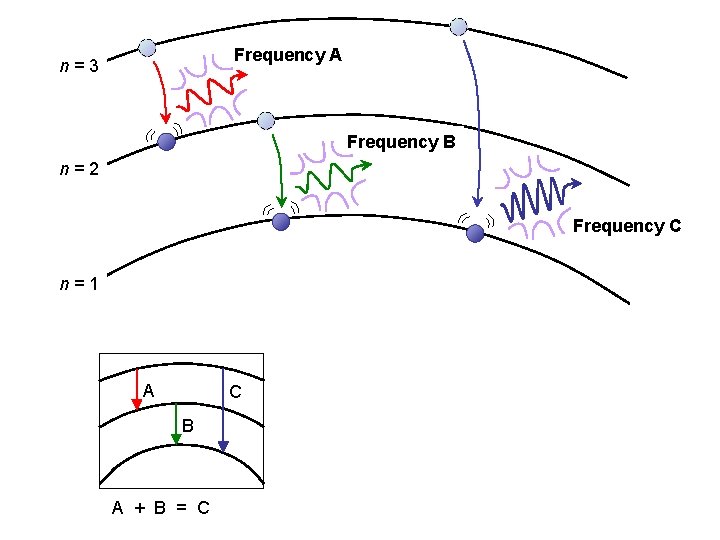
Frequency A n=3 Frequency B n=2 Frequency C n=1 A C B A + B = C

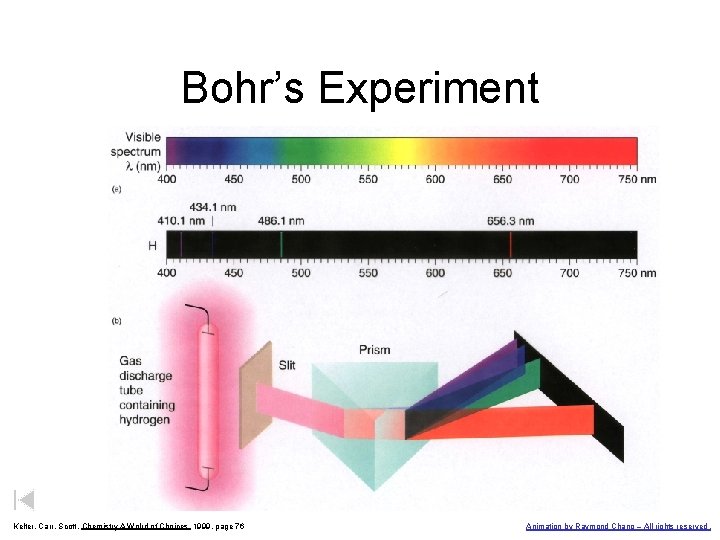
Bohr’s Experiment Kelter, Carr, Scott, Chemistry A Wolrd of Choices 1999, page 76 Animation by Raymond Chang – All rights reserved.

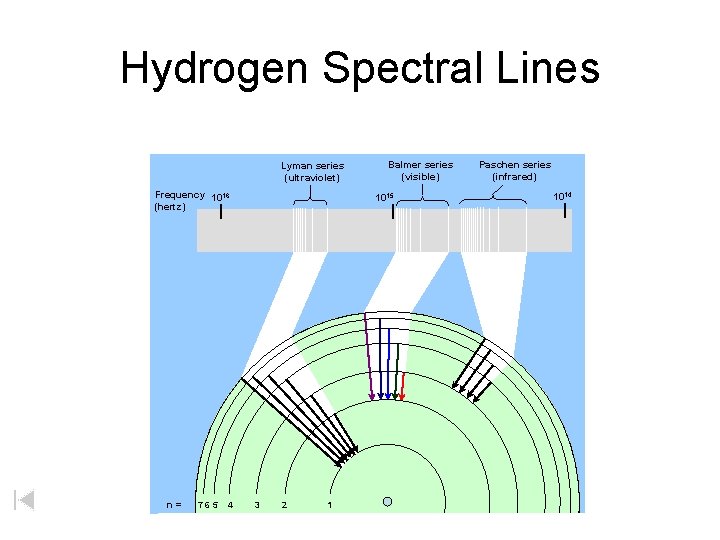
Hydrogen Spectral Lines Lyman series (ultraviolet) Frequency 1016 (hertz) n= 765 4 Balmer series (visible) 1015 3 2 1 Paschen series (infrared) 1014

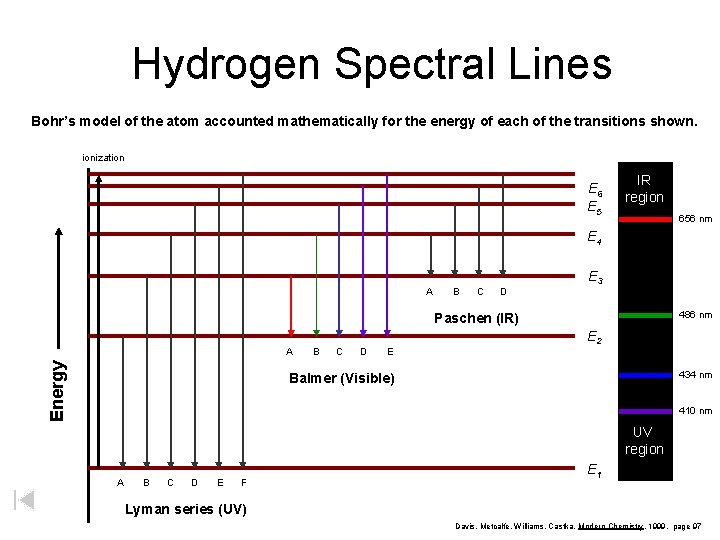
Hydrogen Spectral Lines Bohr’s model of the atom accounted mathematically for the energy of each of the transitions shown. ionization E 6 E 5 IR region 656 nm E 4 A B C D E 3 486 nm Paschen (IR) Energy A B C D E E 2 434 nm Balmer (Visible) 410 nm UV region A B C D E F E 1 Lyman series (UV) Davis, Metcalfe, Williams, Castka, Modern Chemistry, 1999, page 97

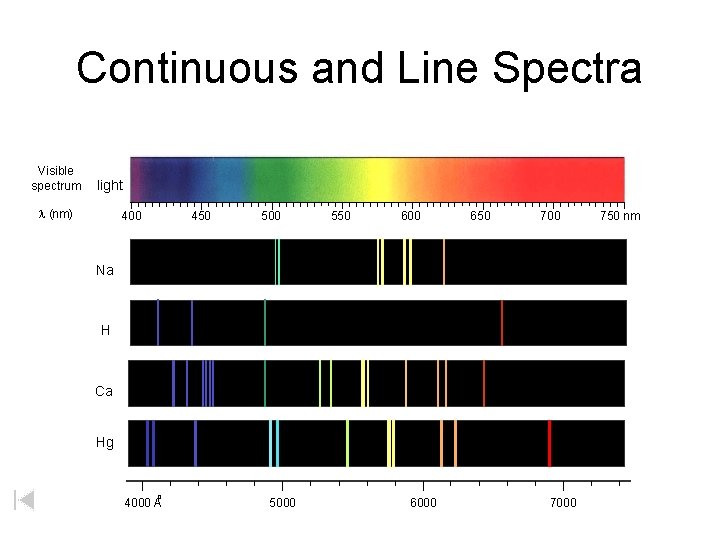
Continuous and Line Spectra Visible spectrum light (nm) 400 450 500 550 600 650 700 Na H Ca Hg o 4000 A 5000 6000 750 nm

Flame Emission Spectra Photographs of flame tests of burning wooden splints soaked in different salts. methane gas wooden splint sodium ion calcium ion copper ion strontium ion Include link to web page http: //www. unit 5. org/christjs/flame%20 tests. htm

The Electromagnetic Spectrum Decreasing wavelength Increasing frequency Increasing photon energy AM radio V i s i b l e Television channels Short wave radio FM radio Radar Microwave Radio Waves L i g h t infrared R O Red Orange Y Yellow G Green B Blue UV Rays Gamma Rays X- Rays I Indigo V Violet

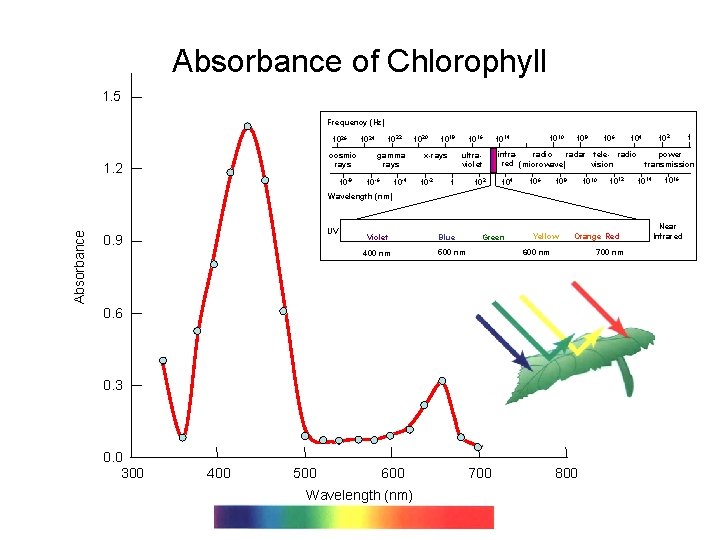
Absorbance of Chlorophyll 1. 5 Frequency (Hz) 1026 cosmic rays 1. 2 10 -8 1022 1024 gamma rays 10 -4 10 -6 1020 1018 ultraviolet x-rays 10 -2 1016 1 102 1010 1014 106 108 infraradio radar tele- radio red (microwave) vision 104 106 108 1010 1012 104 power transmission 1014 1016 Absorbance Wavelength (nm) UV 0. 9 Violet 400 nm Blue Green Yellow Orange Red 700 nm 600 nm 500 nm 0. 6 0. 3 0. 0 300 400 500 600 Wavelength (nm) 700 800 1 Near Infrared

Wave Interference

Wave Interference Bright Bright

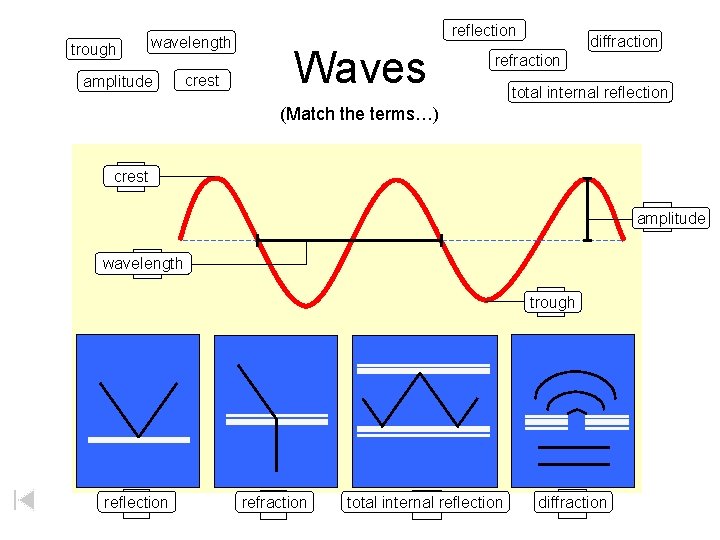
trough wavelength amplitude crest reflection Waves diffraction refraction total internal reflection (Match the terms…) crest A D amplitude wavelength C trough B reflection E refraction F total internal G reflection diffraction H

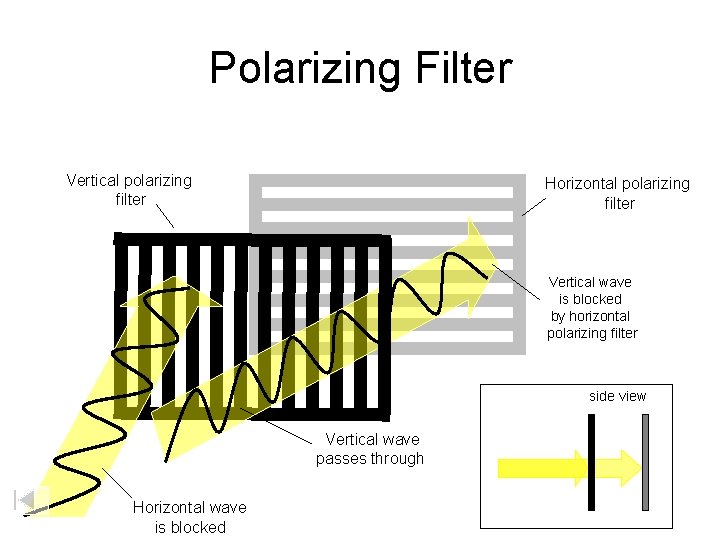
Polarizing Filter Vertical polarizing filter Horizontal polarizing filter Vertical wave is blocked by horizontal polarizing filter side view Vertical wave passes through Horizontal wave is blocked