Formation and Nature of Ionic Bonds Ionic Bond

Formation and Nature of Ionic Bonds

Ionic Bond • Ionic bond – the electrostatic force that holds ions together in an ionic compound • Electrostatic force – like charges repel, opposite charges attract
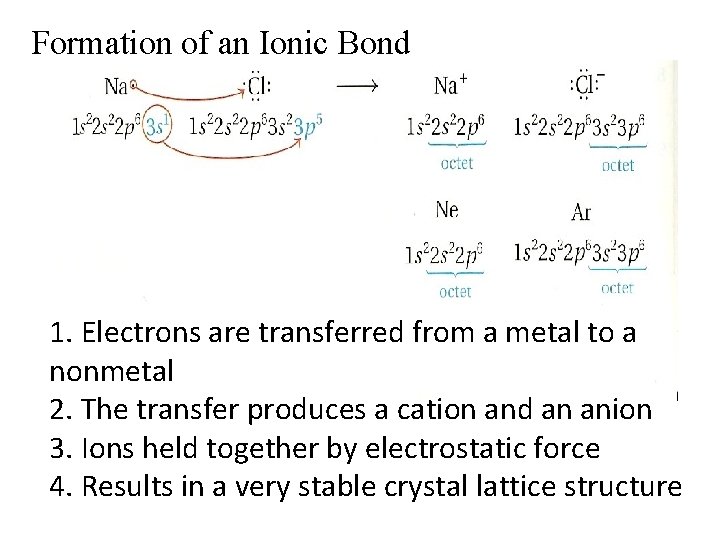
Formation of an Ionic Bond 1. Electrons are transferred from a metal to a nonmetal 2. The transfer produces a cation and an anion 3. Ions held together by electrostatic force 4. Results in a very stable crystal lattice structure

Ionic Compounds • • • Compound composed of cations and anions Usually a metal and nonmetal Overall neutral charge (zero) Held together by ionic bonds A. K. A. salts

Binary Compounds • Has just two different elements • A metallic cation combines with a nonmetallic anion • Example – Mg. O – Not Ca. SO 4, why?

Formula Units • A chemical formula shows the kinds and numbers of atoms in the smallest representative unit of a substance – Sodium chloride Na. Cl – Magnesium chloride Mg. Cl 2 • A formula unit is the lowest wholenumber ratio of ions in an ionic compound
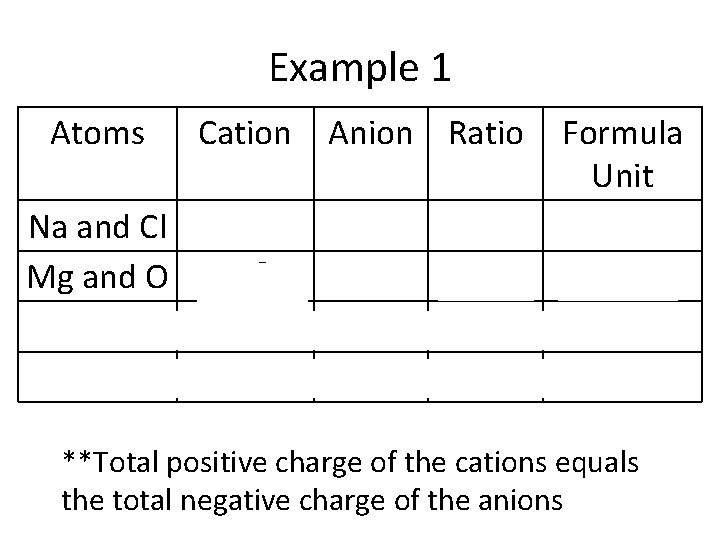
Example 1 Atoms Na and Cl Mg and O Na and O Al and Br Cation Anion Ratio Na+ Mg 2+ Na+ Al 3+ Cl− O 2− Br− 1: 1 2: 1 1: 3 Formula Unit Na. Cl Mg. O Na 2 O Al. Br 3 **Total positive charge of the cations equals the total negative charge of the anions

Example 2 Predict the compound formed when magnesium and nitrogen combine. 1. 2. 3. 4. 5. Start with the atoms Mg N Add Lewis dot diagram Mg 2+ N 3− Write their ions Crisscross charges to get neutral compound Write formula unit Mg 3 N 2

Properties of Ionic Compounds • • • Strong bonds High melting and boiling points Solid at room temperature Hard, rigid, and brittle When dissolved in water or melted it can conduct an electric current

Electrolyte • Definition: A substance that conducts an electric current when it is dissolved in water • When an ionic compound dissolves in solution it breaks up into its individual ions. This enables it to conduct a current because the ions move around. • Salt dissolving in water animation • Gatorade has electrolytes. It has salts dissolved in water for your body to conduct electrical currents.
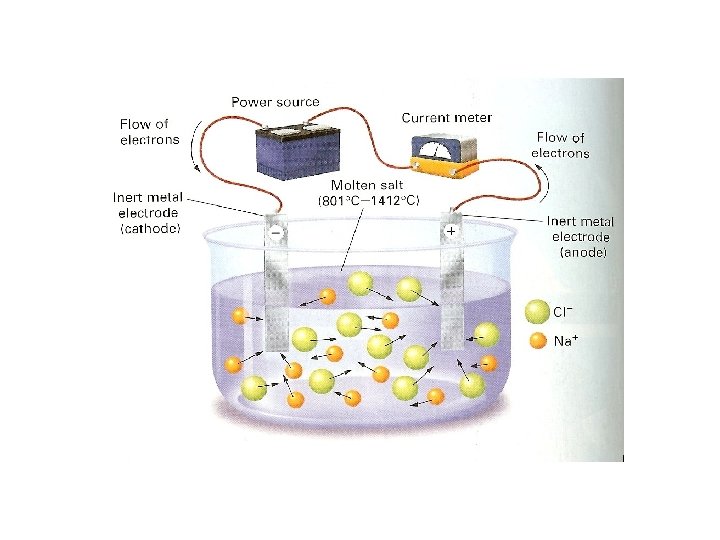
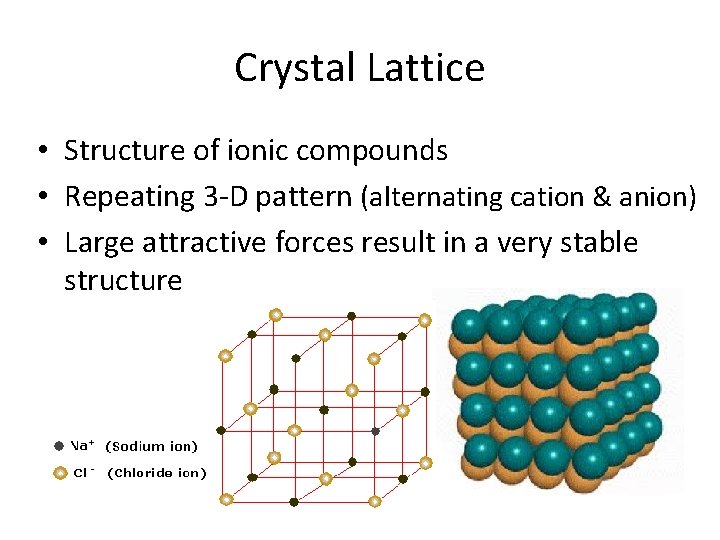
Crystal Lattice • Structure of ionic compounds • Repeating 3 -D pattern (alternating cation & anion) • Large attractive forces result in a very stable structure
- Slides: 12