Formation and Dissipation of Trihalomethanes during Aquifer Storage



















- Slides: 21

Formation and Dissipation of Trihalomethanes during Aquifer Storage and Recovery Operations Jason Pulley City of Salem Public Works

System Overview • Slow sand filtration – 80 MGD firm treatment capacity – 66 MGD transmission capacity • • 183, 000 service population 30 MGD average 55 MGD peak day 136 MG system storage

ASR Operations • ASR used to supplement treatment plant production during high-use periods – Used in winter during high turbidity events • Injection typically from Nov-March – Inject at two of four wells ≈ 3. 5 mgd • Storage goal of 500 mg – Recovery from 3 of 4 wells ≈ 6. 7 mgd – 100% recovery with use of groundwater rights

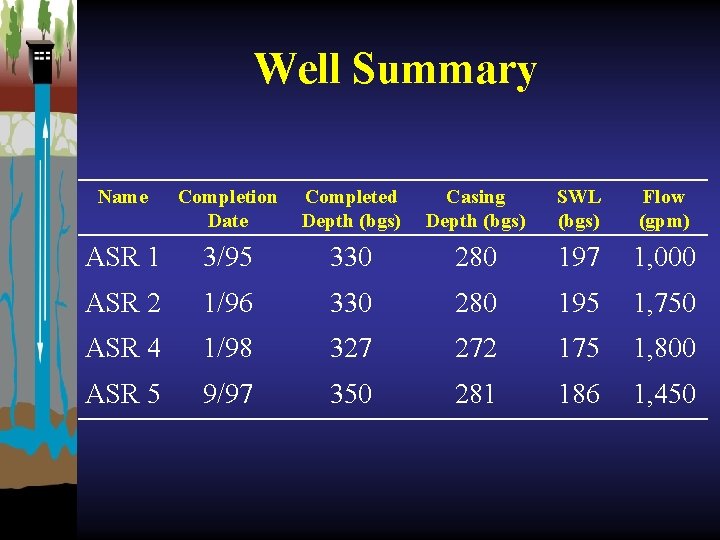
Well Summary Name Completion Date Completed Depth (bgs) Casing Depth (bgs) SWL (bgs) Flow (gpm) ASR 1 3/95 330 280 197 1, 000 ASR 2 1/96 330 280 195 1, 750 ASR 4 1/98 327 272 175 1, 800 ASR 5 9/97 350 281 186 1, 450

Distribution System Water Quality (Typical) p. H TOC TDS Alkalinity Calcium Magnesium Potassium TTHM 7. 2 0. 64 mg/L 30. 0 mg/L 20. 4 mg/L 3. 64 mg/L 0. 95 mg/l 0. 57 mg/L 17 -57 ug/L

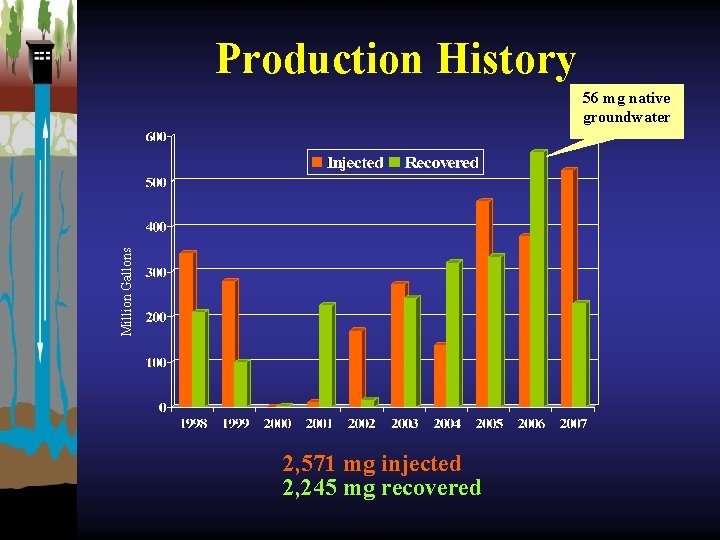
Production History Million Gallons 56 mg native groundwater 2, 571 mg injected 2, 245 mg recovered

Observations • Elevated THM concentrations noticed in 2005 – Isolated at ASR 5 – Quickly dissipated during recovery operations • Observed again in 2006 at ASR 5 and ASR 4 – Possibly related to storage volume – 350 mg in 2005; 400 mg in 2006 – Had only stored 350 mg once (2000) and never 400 mg – Historic levels had fluctuated but never at the levels observed

Previous Studies • Singer et al. (1993) J. AWWA – THMs and HAAs are removed from chlorinated water during storage – Precursors are also removed to a significant degree – Biological mechanisms are suspected • Mc. Quarrie et al. (2003) J. Env. Eng. – Acquifer storage of chlorinated water resulted in a 44% reduction in THM formation – THM removal accelerated under anoxic conditions – Significant DOC reduction during aquifer storage

Previous Studies • Pyne et al. (1996) AWWARF – Focused on five sites with injected treated drinking water – Storage periods from 36 -127 d – THM reductions of 25 -100% – Some loss attributed to dilution/mixing; biodegradation plays a significant role – Also reported reduction in THM precursors

Previous Studies • Landmeyer et al. (2000) J. AWRA – Las Vegas Valley Water District ASR – Observed increases in THM concentrations during recovery – Conc. decreased with continued pumping • Adsorption • Mixing • Microbial degradation – Lab studies show no significant CHCl 3 biodegradation (aerobic or anaerobic) • Low organic carbon content restricts microbial attenuation – CHCL 3 entrained in water or formed in situ will tend to persist

Initial Investigations • Monitor THM concentrations over a 30 -day storage period – Weekly measurements of THM at each of four wells • Collect samples from each well and finished water from TP – 7 -d THMFP, DOC, SUVA on all samples – 30 -d THMFP on finished water – 7 & 30 -d SDS on finished water • Time series analysis after 30 -d storage (every 10 min for 1 h)

Analytical Methodology • EPA Method 524. 2 – Purgeable organic compounds by capillary column GC/MS – Alternate methods 551. 1 & 552. 2 (liquid extraction with ECD) – All provide full speciation • Hach Procedure 10132 – Colorimetric read on spectrophotometer – All results reported as chloroform (CHCl 3) – Estimated detection limit of 6 g L-1

Hach Procedure 10132 • Provides “screening level” data – Low-cost quantitative data – Internal comparisons within +/- 10% – Prep and analysis time < 30 min • Hach validation – vs 524. 2, 551. 1, 552. 2 – R 2 values of 0. 906, 0. 938, and 0. 959, respectively

NOM & SUVA • NOM – a mixture of humic and nonhumic organic substances – Contributes to DBP precursor levels and speciation • Humic substances have higher SUVA and formation potential than nonhumic – SUVA = UV @ 254 / DOC • SUVA provides an indicator for DBP formation • SUVA > 2 L/mg-m generally considered high formation potential

ASR THM Formation Potential 7 d THMFP DOC SUVA FCl* --- g/L--- --mg/L-- --L/mg-m-- ---mg/L--- ASR 1 82 0. 95 2. 9 4. 2 ASR 2 90 0. 85 3. 6 ASR 4 87 0. 65 3. 1 4. 6 ASR 5 95 0. 58 2. 6 4. 7 * Initial chlorine dose of 6. 0 mg/L Treatment Plant Effluent DOC: 0. 67 mg/L 7 d FP: 41 g/L 7 d SDS: 41 g/L SUVA: 1. 9 L/mg-m 30 d FP: 36 g/L 30 d SDS: 62 g/L

Historic Storage Period THM Concentrations

THM and Storage Volume

Geochemistry of Stored Water

Time-Series THM Concentrations During Recovery

Findings • THM concentration appears to be related to storage volume • Formation occurs rapidly during storage period • Rate of dissipation indicates that THMs are not evenly distributed in stored water – Lack of observation in ASR 1 & 2 suggests a localized phenom • Dissipation of THMs does not appear to be a function of mixing/dilution (based on geochem data) • TOC probably the limiting factor in formation • ∆ SUVA indicates potential source of additional TOC/DOC

Next Steps • Continue to monitor during current storage period to see if levels increase above existing concentrations – More frequent analysis during recovery operations • Further analysis of SUVA during injection and recovery cycles at varying water elevations • Examine HAA formation characteristics • Pursue dechlorination of injection water if increasing concentration are not manageable