Form Revision and Development Tiffany Hunt CCRP Rachael

































- Slides: 41

Form Revision and Development Tiffany Hunt, CCRP Rachael Latchana, MPH Stephanie Meyers February 20, 2019 The CIBMTR® (Center for International Blood and Marrow Transplant Research®) is a research collaboration between the National Marrow Donor Program® (NMDP)/Be The Match® and the Medical College of Wisconsin (MCW). TRAINING & DEVELOPMENT |.

Objectives Understand the Form Revision Process Review 2019 Revisions TRAINING & DEVELOPMENT | 2.

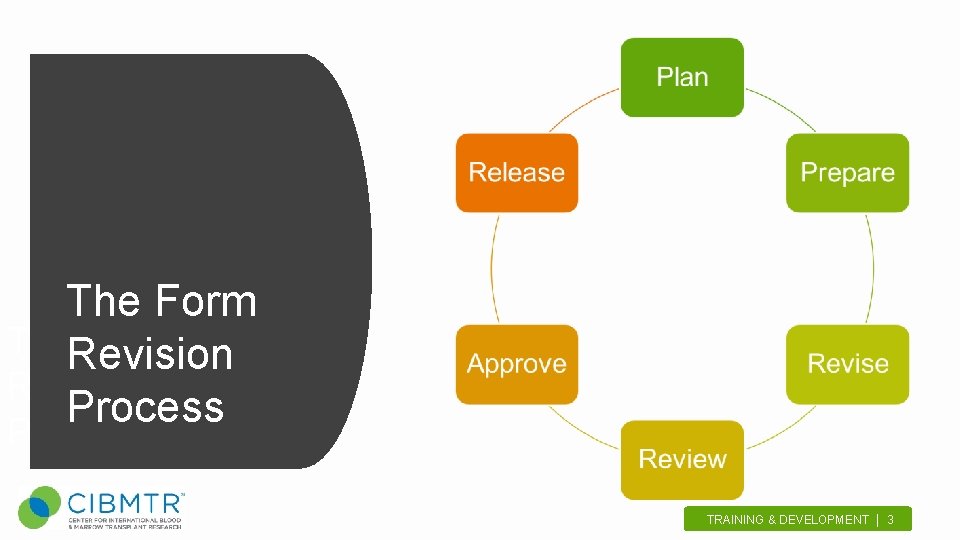
The Form The. Revision Form Revision Process TRAINING & DEVELOPMENT | 3.

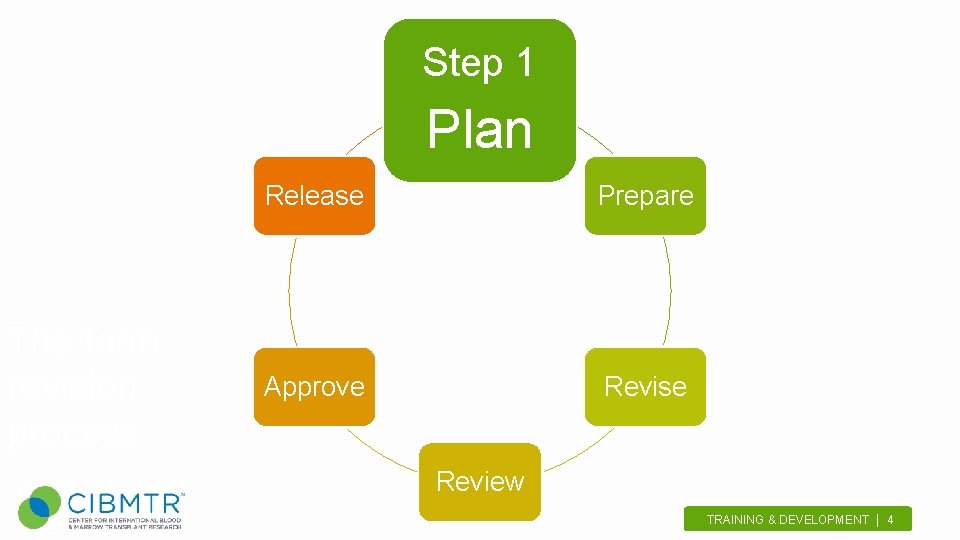
Step 1 Plan The form revision process Release Prepare Approve Revise Review TRAINING & DEVELOPMENT | 4.

Plan Why revise a form? • • • Time since last revision Upcoming studies Updates or resolving issues Updates from WHO Requests from staff or centers TRAINING & DEVELOPMENT | 5.

Plan Create a Roadmap • Form Revision Core Team • Draft proposed schedule • Obtain approval • Scientific directors and senior leaders review and approve • Be flexible! • allow wiggle room for critical revisions, or new priority forms TRAINING & DEVELOPMENT | 6.

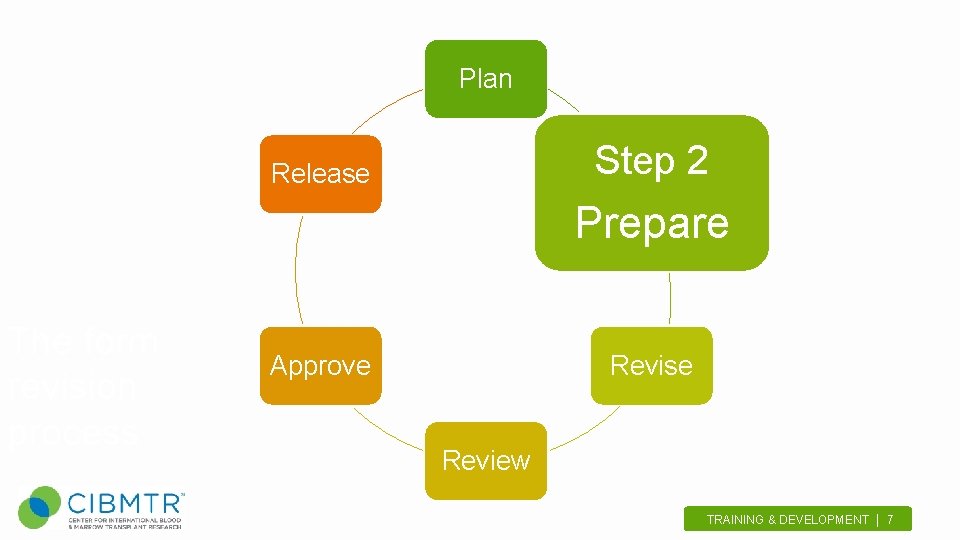
Plan Step 2 Release Prepare The form revision process Approve Revise Review TRAINING & DEVELOPMENT | 7.

Prepare • Announce revisions to the network – Includes scientific directors and centers • Volunteer! – Send form change suggestions • Meet with scientific director of working committee TRAINING & DEVELOPMENT | 8.

Plan Release The form revision process Prepare Step 3 Approve Revise Review TRAINING & DEVELOPMENT | 9.

Revise Initial Review Committee (IRC) • Consists of field expert physicians, subject matter experts (SME), data managers, CIBMTR staff and other individuals • Weekly Meetings (4 weeks, 2 hours) – review the form(s) top to bottom, question by question – remove outdated questions, add new questions based on new technology/treatments, clarify problematic sections • Q&A Meeting – allow for data managers to pilot the forms – ensure source documents exist for requested data TRAINING & DEVELOPMENT | 10.

Plan The form revision process Release Prepare Approve Revise Step 4 Review TRAINING & DEVELOPMENT | 11.

Review Internal Review – CIT review – Statistical review – Database review • Develop validations, identify potential problem areas, and discuss form development TRAINING & DEVELOPMENT | 12.

Plan Release Prepare Step 5 Revise Approve Review TRAINING & DEVELOPMENT | 13.

Approve • Scientific Director(s) – Review form drafts and change summaries – Review occurs at standing weekly meeting • Request volunteers for time studies – Assess form completion burdens TRAINING & DEVELOPMENT | 14.

Approve OMB Approval • Form 2400, 2402, 2004, 2005, 2006, and 2450 • Long process (6+ months) • Post to the Federal Register: 60 day notice, 30 day notice • OMB review and approval TRAINING & DEVELOPMENT | 15.

Post- Approval • Forms. Net 3 Process – Build new forms – Write/test validations – Define Events and Actions (logic for how forms come due) – Metadata work – AGNIS – Mappings TRAINING & DEVELOPMENT | 16.

Plan Step 5 Release Prepare Approval Revise Review TRAINING & DEVELOPMENT | 17.

Release • New Forms – Announce to Network – Release forms prior to “Go Live” date • Forms Instruction Manual – Announce to Network – Release updated manuals prior to “Go Live” date • Update / develop e. Learnings TRAINING & DEVELOPMENT | 18.

Finally…How can YOU help? • Submit form change suggestions – Submit at any time – Send to your CRC – Suggestions reviewed during the next revision • Volunteer – Participate on a form revision committee! – Important to have data managers involved TRAINING & DEVELOPMENT | 19.

Finally…How can YOU help? • Time studies – We need your input on the length of time it takes to complete the forms – Important information for OMB process but also any revised forms TRAINING & DEVELOPMENT | 20.

Upcoming Revisions 2 1 TRAINING & DEVELOPMENT | 21.

The Core Forms • 2400, 2402, 2000, 2004, 2005, 2006, 2450, 2804 – Revision committees met May – July 2018 for 2400/2000, 2006 – Internal review July – October 2018 – Requires OMB approval (in process) • Scheduled for release in Summer 2019 – Will be posted 2 months in advance for preview TRAINING & DEVELOPMENT | 2222.

Pre-TED (Form 2400) Revision Highlights *not a complete list Fun Fact: Decreased total number of questions (357 to 142) • ‘Check all that apply’ capability: • Race, race detail, country of residence, comorbidities, planned post-HCT therapy • Recipient Information section – Ethnicity, race detail • Collected on CRID assignment (Form 2804) • Auto-populated on Pre-TED TRAINING & DEVELOPMENT | 2323.

Pre-TED (Form 2400) Revision Highlights (cont. ) • Hematopoietic Cellular Transplant (HCT) section – New cellular therapy (CT) history questions • • Previous history of CT All prior CT reported to CIBMTR Place of prior CT Does not require completion of a F 4000 TRAINING & DEVELOPMENT | 2424.

Pre-TED (Form 2400) Revision Highlights (cont. ) • Donor Information section – Donor ID • Collect only on Pre-TED • Auto populate Forms 2004/2005/2006 – Specify donor list condensed • Autologous, allogeneic related / unrelated • Product • NMDP – Genetically modified HCT products – Consolidated all donor-related questions from this and other forms TRAINING & DEVELOPMENT | 25.

Pre-TED (Form 2400) Revision Highlights (cont. ) • Pre-HCT Preparative Regimen (Conditioning) section – Chemotherapy Drugs • Added ‘multiple’ capability to eliminate Yes/No for each drug • Updated drug list • Additional Drugs Given in Peri-Transplant Period section – NEW SECTION TO FORM • Used to report drugs given for both the preparative regimen and GVHD prophylaxis TRAINING & DEVELOPMENT | 2626.

Disease classification (F 2402) Revision Highlights • New indications *not a complete list – “Tolerance induction associated with solid organ transplant” – “Recessive Dystrophic Epidermolysis Bullosa” • Cytogenetic results – Added a place holder question to allow for future submission of results – Cytogenetic abnormality list has been made ‘check all that apply’ (MDS/MPN, PCD) TRAINING & DEVELOPMENT | 2727.

Disease classification (F 2402) Revision Highlights cont. • Inherited Abnormalities of Erythrocyte Differentiation or Function section – New questions added to capture beta thalassemia and sickle cell TRAINING & DEVELOPMENT | 2828.

Baseline (F 2000) Revision Highlights*not a complete list Fun Fact: Decreased total number of questions (264 to 118) • Recipient Demographics / Race / Clinical status – Moved country of primary residence, ethnicity, race detail, recipient blood type and Rh factor to pre-TED • Infection – Collect infections in 6 months prior to infusion • Pre-HCT Preparative Regimen (Conditioning) section – Chemotherapy Drugs • Added ‘multiple’ capability to eliminate Yes/No for each drug • Updated drug list TRAINING & DEVELOPMENT | 2929.

Baseline (F 2000) Revision Highlights (cont. ) • Additional Drugs Given in Peri-Transplant Period section – NEW SECTION TO FORM • Used to report drugs given for both the preparative regimen and GVHD prophylaxis • Socioeconomic Information – Will collect most recent works status – Updated insurance list TRAINING & DEVELOPMENT | 3030.

Infusion (F 2006) Revision Highlights*not a complete list Fun Fact: Decreased total number of questions (285 to 170) • Donor/cord blood unit identification – Donor Identification to be auto-populated on form in key fields • Product processing and manipulation – Consolidated questions from pre-TED to here • Removed whole section for Autologous products TRAINING & DEVELOPMENT | 3131.

Infusion (F 2006) Revision Highlights (cont. ) • Product analysis – Consolidated timepoints to “pre-cryopreservation” and “post thaw” – Expanded cell types (TNC, CD 34, CD 3/4/8) and • collect viability per cell type instead of overall viability • Product infusion – Clarified question “was the entire volume of received product infused” TRAINING & DEVELOPMENT | 3232.

Infusion (F 2006) Revision Highlights (cont. ) • Donor/Infant demographic information – Donor blood type/Rh factor moved to pre-TED – Biological relationship of donor to recipient • Removed and consolidated to the pre-TED – Transferable genetic or clonal abnormalities • Expanded option list TRAINING & DEVELOPMENT | 33.

IDM and HLA (F 2004 / F 2005) Revision Highlights • Donor/cord blood unit identification *not a complete list – Donor Identification to be auto-populated on form in key fields • Form 2004 – New question added to capture NAT testing for HBV – NAT testing for HIV-1 and HCV split – Removed questions for Anti-HTLV 1, syphyllis, CMV, WNV, toxoplasmosis • Form 2005 – Form will be collected for recipient and final donor only TRAINING & DEVELOPMENT | 3434.

Post-TED (F 2450) Revision Highlights*not a complete list • ‘Check all that apply’ capability: • Liver toxicity prophylaxis, post-HCT therapy, relapse or progression post-HCT therapy • GVHD – Added questions to capture organ staging since the date of the last report • Chimerism studies – Collecting chimerism data for beta thalassemia and sickle cell recipients TRAINING & DEVELOPMENT | 3535.

Post-TED (F 2450) Revision Highlights (cont. ) • Relapse or progression post-HCT – The intent of the F 2450 has always been to capture the first relapse only – Updated question text to say “was the date of the first clinical/hematologic relapse or progression previously reported” – Removed “decreased chimerism” from the intervention given for relapse question TRAINING & DEVELOPMENT | 36.

CRID Assignment Form (F 2804) Revision Highlights • Demographics – Added recipient ethnicity, race and race detail – Will be auto-populated on pre-TED – Race will be ‘check all that apply’ – Race detail list will automatically filter based on race selection TRAINING & DEVELOPMENT | 37.

Plasma Cell Disorder Revision Highlights • Revising the forms 2016/2116 and the PCD section of the 2402 • Capturing biclonal/triclonal scenarios • Capturing information on monoclonal gammopathy of renal significance (MRGS) and POEMS • Updated Amyloidosis sections • Fall release TRAINING & DEVELOPMENT | 38.

Contact form (F 2820) • Form will collect recipient contact details for use in: – CIBMTR observational research database: permission to contact for future CIBMTR research studies – BMT CTN studies for direct patient follow up – Additional future studies • Includes: – name – address – phone number – email TRAINING & DEVELOPMENT | 3939.

TRAINING & DEVELOPMENT | 40. 40

Questions TRAINING & DEVELOPMENT | 41.