FORENSIC APPLICATIONS OF LAICPMS ELEMENTAL PROFILING AND EVALUATION






















- Slides: 31

FORENSIC APPLICATIONS OF LA-ICP-MS: ELEMENTAL PROFILING AND EVALUATION OF HOMOGENEITY IN SODALIME CONTAINER GLASS Karen J. Harrington

Inductively Coupled Plasma Mass Spectrometry (ICP-MS) Previous method of RI predominantly used l Elemental analysis research began three decades ago l l Allows classification of product type and use Shown to be a discriminating technique forensic analysis Drawbacks l l l Dangerous digestions Sample size requirements Destructive nature

Laser ablation (LA)-ICP-MS l. A viable method forensic glass comparisons l Creates aerosol of super-fine glass fragments – Advantages Decreased sample prep. time l Minimal sample consumption l Maintains good sensitivity for most elements l Less risk of contamination l

Homogeneity l Are elements consistent within a glass item (micro-homogeneity)? – Natural heterogeneity (result of manufacturing) Contamination, natural variation in raw material l How do elements vary between glass – Variation beyond natural fluctuation? items?

Previous Research on Homogeneity – LA-ICP-MS does reveal natural variations in glass – Understanding homogeneity within a unit of glass is a factor when: Comparing samples from different categorical sources l Comparing samples from different manufacturers l

Homogeneity l Glass items typically maintain some homogeneity overall l Glass of the same type can often be distinguished on an item-item basis l Glass bottles tend to be more variable and may not be distinguishable from each other within one manufacturer

Project Goals l How consistent are elemental profiles in glass bottles? l What are appropriate match-criteria to balance false positives and false negatives? l How variable are glass bottles within a manufacturer?

Background – Stage I l Verify valid set of match-criteria – Examine homogeneity of a single bottle – Determine what criteria allow for treatment of bottle as a whole unit

Background – Stage II l Determine normal/natural manufacturer variability – Hourly runs – Daily runs – Simultaneous/daily for multiple furnaces

Method – Sample Selection Gallo Glass Company, Modesto, CA l Typical soda-lime, with cullet (25% recycled) l Stage 1 l – 10 bottles from two furnaces, variable time intervals, colors, types (3 fragments each from neck, sidewall, base regions of each bottle) l Stage 2 – Hourly, daily simultaneous furnace

Method – Analyte Selection l Analytes were selected from an acid digestion of selected bottles from the population. l Ratios were determined based on %RSD and mass-to-charge values.

Method – Comparison Criteria l T-tests and ANOVA were found to be unacceptable for the purposes of this project. l Typical case-work match-criteria of ± 2 SD was used

Method – Instrumentation l Perkin-Elmer ELAN DRC II ICP-MS (a) l New Wave Research Nd: YAG LA (1 = 213 nm) unit (b) a, b Dodds, A. J. , Land, D. P. , Pollock, E. M. Determination of Elemental Homogeneity in Automotive Windshields by LAICP-MS. The CACNews. 4 th Quarter 2005. 17 – 20.

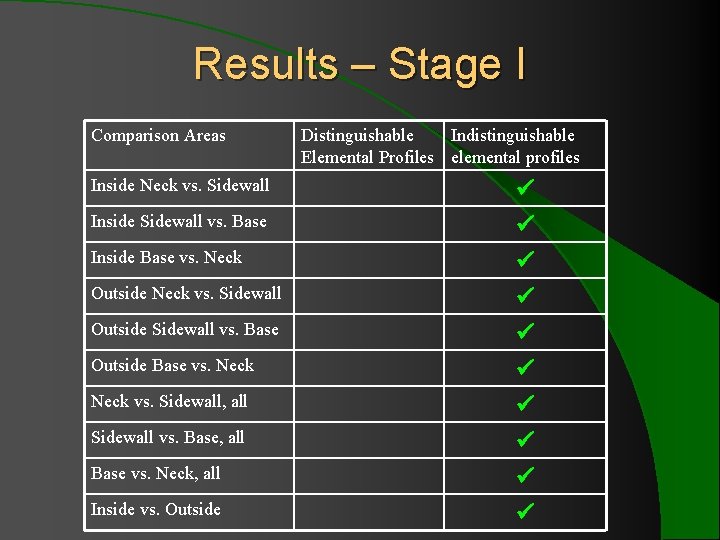
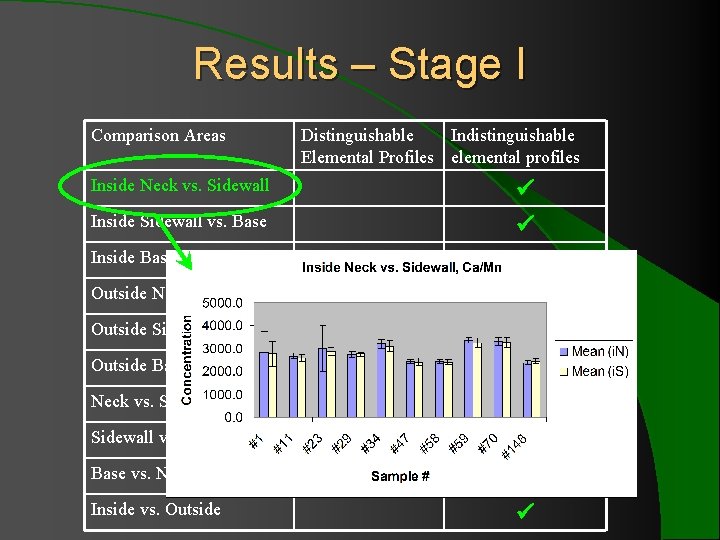
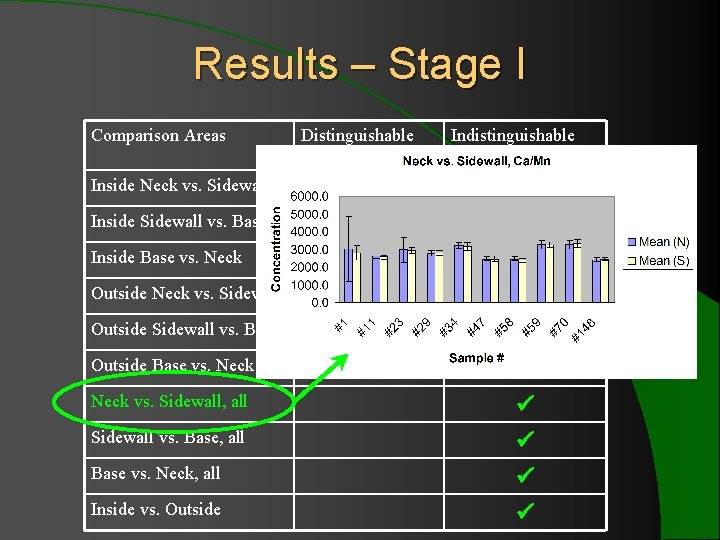
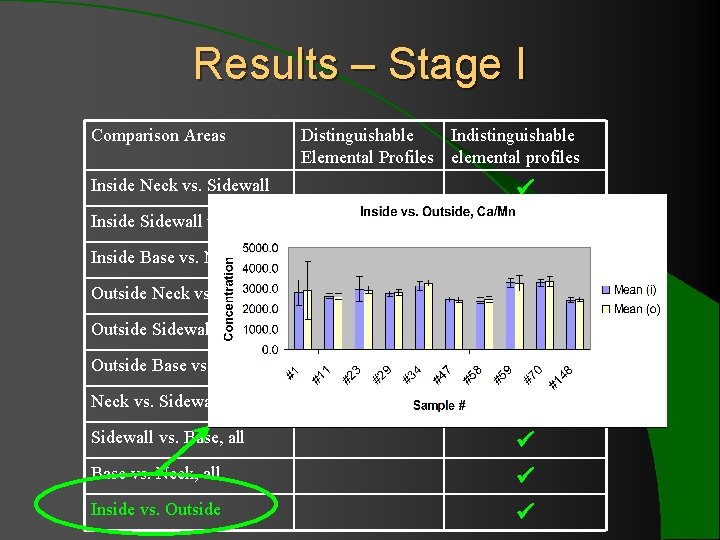
Results – Stage I l 10 comparison of multiple regions showed indistinguishable elemental profiles for 13 ratios with ± 2 SD. l When ± 3 SD was applied to Sr/Zr and Fe/Cr (the two consistently problematic ratios), all 10 bottles showed indistinguishable elemental profiles for 15 elements.

Results – Stage I Comparison Areas Inside Neck vs. Sidewall Inside Sidewall vs. Base Inside Base vs. Neck Outside Neck vs. Sidewall Outside Sidewall vs. Base Outside Base vs. Neck vs. Sidewall, all Sidewall vs. Base, all Base vs. Neck, all Inside vs. Outside Distinguishable Indistinguishable Elemental Profiles elemental profiles

Results – Stage I Comparison Areas Inside Neck vs. Sidewall Inside Sidewall vs. Base Inside Base vs. Neck Outside Neck vs. Sidewall Outside Sidewall vs. Base Outside Base vs. Neck vs. Sidewall, all Sidewall vs. Base, all Base vs. Neck, all Inside vs. Outside Distinguishable Indistinguishable Elemental Profiles elemental profiles

Results – Stage I Comparison Areas Inside Neck vs. Sidewall Inside Sidewall vs. Base Inside Base vs. Neck Outside Neck vs. Sidewall Outside Sidewall vs. Base Outside Base vs. Neck vs. Sidewall, all Sidewall vs. Base, all Base vs. Neck, all Inside vs. Outside Distinguishable Indistinguishable Elemental Profiles elemental profiles

Results – Stage I Comparison Areas Inside Neck vs. Sidewall Inside Sidewall vs. Base Inside Base vs. Neck Outside Neck vs. Sidewall Outside Sidewall vs. Base Outside Base vs. Neck vs. Sidewall, all Sidewall vs. Base, all Base vs. Neck, all Inside vs. Outside Distinguishable Indistinguishable Elemental Profiles elemental profiles

Results – Stage 2 (hourly) Comparison Areas Distinguishable Elemental Profiles Group 1 – furnace 3 * * Group 2 – furnace 2 Group 1 vs. Group 2 Indistinguishable elemental profiles * Group 1 (one bottle distinguishable for 1 element) and Group 2 (two bottles distinguishable for 3 elements) show some distinguishable elemental profiles from other bottles.

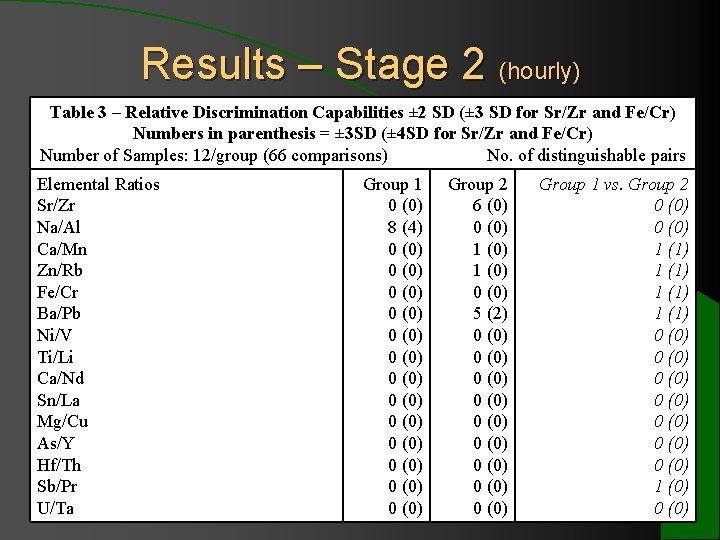
Results – Stage 2 (hourly) Table 3 – Relative Discrimination Capabilities ± 2 SD (± 3 SD for Sr/Zr and Fe/Cr) Numbers in parenthesis = ± 3 SD (± 4 SD for Sr/Zr and Fe/Cr) Number of Samples: 12/group (66 comparisons) No. of distinguishable pairs Elemental Ratios Group 1 Group 2 Group 1 vs. Group 2 Sr/Zr 0 (0) 6 (0) 0 (0) Na/Al 8 (4) 0 Indistinguishable (0) 0 (0) Comparison Areas Distinguishable Ca/Mn 0 (0) 1 elemental (0) 1 (1) Elemental Profiles profiles Zn/Rb 0 (0) 1 (1) Group 1 – furnace 3 * Fe/Cr 0 (0) 1 (1) Ba/Pb 0 (0) 5 (2) 1 (1) Group 2 – furnace 2 * Ni/V 0 (0) Ti/Li 0 (0) Group 1 vs. Group 2 0 (0) Ca/Nd 0 (0) Sn/La 0 (0) Mg/Cu 0 (0) As/Y 0 (0) 2 (bottles #48 0 (0) * Group 1 (bottle # 38 for 1 element)0 (0) and Group Hf/Th 0 (0) and #50 for 3 elements) show some distinguishable elemental Sb/Pr 0 (0) 1 (0) profiles from other bottles. U/Ta 0 (0)

Results – Stage 2 (daily) Comparison Areas Distinguishable Elemental Profiles Group 3 – 7/19/05 – 7/30/05 (after 5 days) Group 4 – 7/31/05 – 8/15/05 (after 8 days) Group 5 – 8/9/05 – 8/21/05 (after 4 days) Indistinguishable elemental profiles * * 1 bottle was indistinguishable from another that was 11 days apart

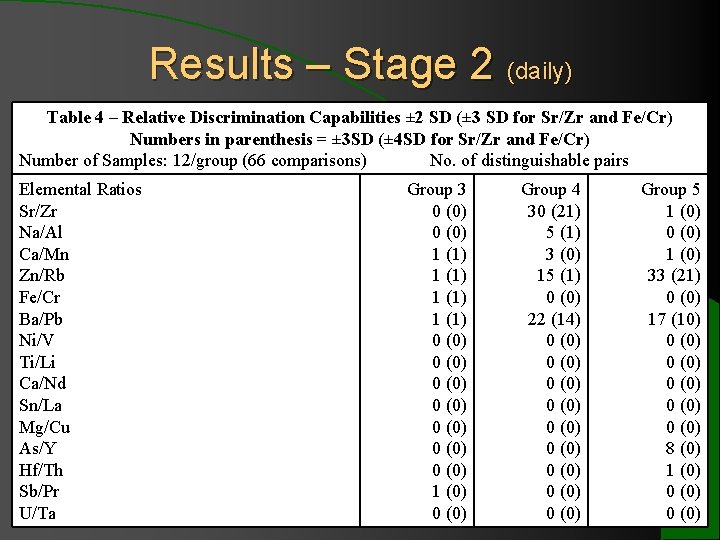
Results – Stage 2 (daily) Table 4 – Relative Discrimination Capabilities ± 2 SD (± 3 SD for Sr/Zr and Fe/Cr) Numbers in parenthesis = ± 3 SD (± 4 SD for Sr/Zr and Fe/Cr) Number of Samples: 12/group (66 comparisons) No. of distinguishable pairs Comparison Distingui Indistingui Elemental Group 3 Group 4 Group 5 Areas. Ratios shable Sr/Zr 0 (0) 30 (21) 1 (0) Elemental elemental Na/Al 0 (0) 5 (1) 0 (0) Profiles profiles Ca/Mn 1 (1) 3 (0) 1 (0) Zn/Rb. Group 3 – 1 (1) 15 (1) 33 (21) (5 Fe/Cr 7/19/05 – 1 (1) 0 (0) days*) Ba/Pb 7/30/05 1 (1) 22 (14) 17 (10) Ni/V 0 (0) Ti/Li Group 4 – 0 (0) (8 Ca/Nd 7/31/05 – 0 (0) days) Sn/La 8/15/05 0 (0) Mg/Cu. Group 5 – 0 (0) (4 * 8/9/05 1 bottle that were As/Y (0) 0 (0) 11 days 8 (0) – was only distinguishable from 0 others Hf/Th 0 (0) 1 (0) or 8/21/05 more apart. days) Sb/Pr 1 (0) 0 (0) U/Ta 0 (0)

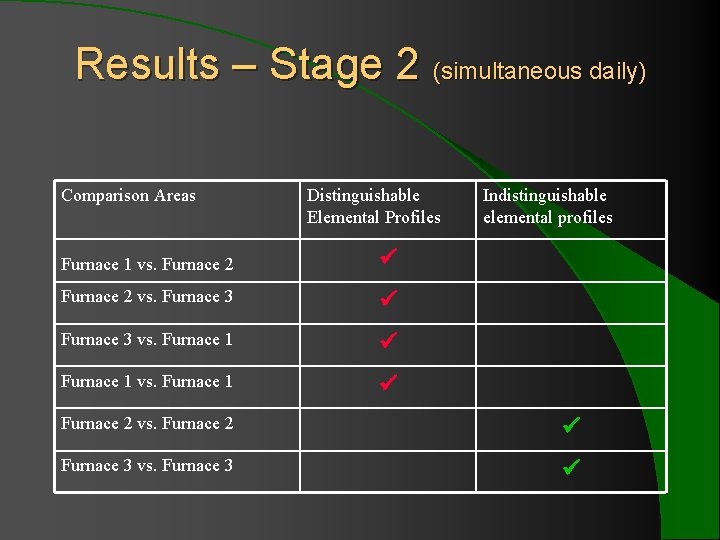
Results – Stage 2 (simultaneous daily) Comparison Areas Distinguishable Elemental Profiles Furnace 1 vs. Furnace 2 vs. Furnace 3 vs. Furnace 1 Indistinguishable elemental profiles Furnace 2 vs. Furnace 2 Furnace 3 vs. Furnace 3

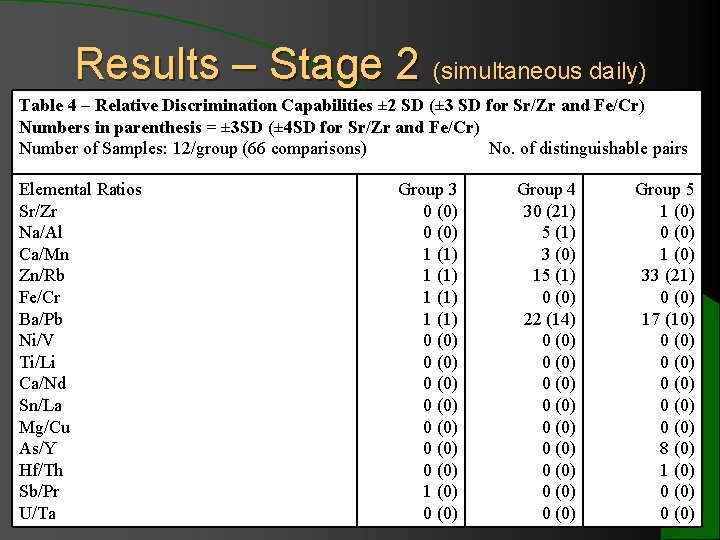
Results – Stage 2 (simultaneous daily) Table 4 – Relative Discrimination Capabilities ± 2 SD (± 3 SD for Sr/Zr and Fe/Cr) Numbers in parenthesis = ± 3 SD (± 4 SD for Sr/Zr and Fe/Cr) Number of Samples: 12/group (66 comparisons) No. of distinguishable pairs Elemental Ratios Sr/Zr Na/Al Ca/Mn Zn/Rb Fe/Cr Ba/Pb Ni/V Ti/Li Ca/Nd Sn/La Mg/Cu As/Y Hf/Th Sb/Pr U/Ta Group 3 0 (0) 1 (1) 0 (0) 0 (0) 1 (0) 0 (0) Group 4 30 (21) 5 (1) 3 (0) 15 (1) 0 (0) 22 (14) 0 (0) 0 (0) 0 (0) Group 5 1 (0) 0 (0) 1 (0) 33 (21) 0 (0) 17 (10) 0 (0) 0 (0) 8 (0) 1 (0) 0 (0)

Conclusion - hourly l Bottles manufactured from the same furnace are typically not distinguishable when compared hourly l 80 – 90% of bottles manufactured within a daily production lot from the same furnace share indistinguishable elemental profiles

Conclusion - daily l Bottles manufactured from the same furnace between 4 and 8 days apart (11 maximum) are typically distinguishable.

Conclusion – simultaneous daily l Bottles manufactured at the same time from different furnaces are distinguishable. l Additionally, some bottles manufactured at the same time on two consecutive days from the same furnace may be distinguishable.

Conclusion – significant information l Supports previous research – Same case/six-pack are indistinguishable 1 – Indistinguishable profiles are typically from same manufacturer 2 l Gallo Glass Company – Produces 800 cases/hour – 1 billion bottles per year 1 2 Trejos, T and Almirall, J. R. Sampling strategies for the analysis of glass fragments by LA-ICP-MS Part I. Microhomogeneity study of glass and its application to the interpretation of forensic evidence. Talanta. 67(2) 388 – 395 (August 2005). J. R. Almirall. Glass as evidence of association. In Mute Witness; When Trace Evidence Makes the Case, M. Houck (Ed. ), Academic Press, San Diego, CA, USE, 139 -155 (2001).

Final Conclusions l Still important to collect/examine multiple fragments from question sample 1 l Provides evidentiary value to forensic examinations – Can distinguish between furnaces of same manufacturer – Can distinguish between bottles from the same furnace after approximately 8 -11 days 1 Trejos, T and Almirall, J. R. Sampling strategies for the analysis of glass fragments by LA-ICP-MS Part I. Microhomogeneity study of glass and its application to the interpretation of forensic evidence. Talanta. 67(2) 388 – 395 (August 2005).

Future Research l Multiple l Bottle l More manufacturer studies distribution (cases, vendors) effective statistical studies

Acknowledgments l Sacramento County District Attorney Laboratory of Forensic Services and Staff l Gallo Glass Company l University of California, Davis: Forensic Science Masters program l California Criminalistics Institute