Forces Between Molecules Intermolecular Forces They determine the

Forces Between Molecules Intermolecular Forces

They determine the phase of a substance at room temperature. (Competition with kinetic energy. ) Why are intermolecular forces important?

Strong Intermolecular Forces Solids

Weak Intermolecular Forces Gases

Dispersion or Van der Waals Dipole-dipole Hydrogen Bonds 3 Types of Intermolecular Forces

Hydrogen Bonds Strongest Intermolecular Force

Hydrogen Bonds

Between molecules containing F, O, N bonded to a H atom When do Hydrogen bonds occur?

Between molecules containing F-H bonds Strongest hydrogen bonds?

Between molecules containing N-H bonds Weakest hydrogen bonds?

Hydrogen bonding leads to substantial increases in the expected boiling point. What effect does H-bonding have on the properties of the substance?

Dispersion or Van der Waals forces Weakest Intermolecular Force

Occur between all molecules. Most important between nonpolar molecules When do dispersion forces occur?

No Poles! The molecule is symmetric! Cannot tell 1 end from the other. Nonpolar

Monatomic Gases (Column 18) (kickballs) & Diatomic Elements (H 2, N 2, O 2, F 2, Cl 2, Br 2, I 2) (footballs) 2 Easy categories of Nonpolar Molecules

Hydrocarbons (Cx. Hy) & Small symmetric molecules (CO 2, CS 2, CF 4, CBr 4, etc. ) 2 More Easy categories of Nonpolar Molecules

H 2, N 2, O 2, F 2, Br 2, Cl 2, & I 2 What are the diatomic elements?

The larger the molecules the stronger the dispersion forces! What can you say about dispersion forces as a function of molecular size?

Between polar molecules. Permanent separation of charge. 1 side - (electron rich) vs. 1 side + (electron poor) When do dipole-dipole forces occur?

Has Poles! The molecule is NOT symmetric! Can tell 1 end from the other. Polar

Gas phase of a substance that is normally a liquid at room temperature. Vapor

The partial pressure of a vapor above its liquid. Vapor Pressure

Vapor pressure depends on the temperature of the liquid. What does vapor pressure depend on? (Hint: see Table H)

The vapor pressure always increases with temperature. What happens to the vapor pressure as the temperature of the liquid is increased?

No. A thimbleful is as good as an ocean. Does the vapor pressure depend on the amount of liquid present?

Evaporation occurs at all temperatures. Boiling occurs at a definite temperature. Evaporation occurs at the surface. Boiling occurs throughout the liquid. What are some differences between evaporation & boiling?

Amount of energy required to convert 1 gram of a pure solid to the liquid phase at its melting point. Heat of fusion

Amount of energy required to convert 1 gram of a pure liquid to the gas phase at its boiling point. Heat of vaporization

The stronger the intermolecular forces, the higher the boiling point. What happens to the boiling point as the intermolecular forces increase?

The stronger the intermolecular forces, the higher the melting point. What happens to the melting point as the intermolecular forces increase?

The stronger the intermolecular forces, the higher the heat of fusion. What happens to the heat of fusion as the intermolecular forces increase?

The stronger the intermolecular forces, the higher the heat of vaporization. What happens to the heat of vaporization as the intermolecular forces increase?

The stronger the intermolecular forces, the lower the evaporation rate. What happens to the evaporation rate as the intermolecular forces increase?

The stronger the intermolecular forces, the lower the vapor pressure. What happens to the vapor pressure as the intermolecular forces increase?
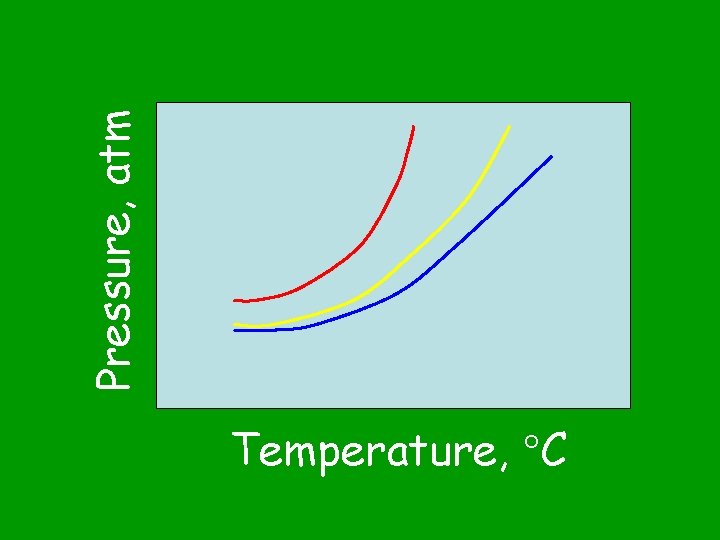
Pressure, atm Temperature, C

IMF BP MP Hf Hv Ev. Rt V. P. Ev. Rt VP Relationship between IMF and physical properties?

Temperature at which the vapor pressure of a liquid is equal to the external pressure. Boiling Point

Temperature at which the vapor pressure of a liquid is equal to 1 atm or 101. 3 k. Pa or 760 torr. Normal Boiling Point

The boiling point is reduced. What happens to the boiling point when the external pressure is reduced?

Solid to gas Sublimation

Gas to solid Deposition

Liquid to Gas Vaporization

Gas to liquid Condensation

Solid to liquid Or Melting Fusion

Liquid to solid Crystallization Solidification Freezing

Solid to liquid Liquid to Gas Solid to Gas G L S Which phase changes are endothermic?
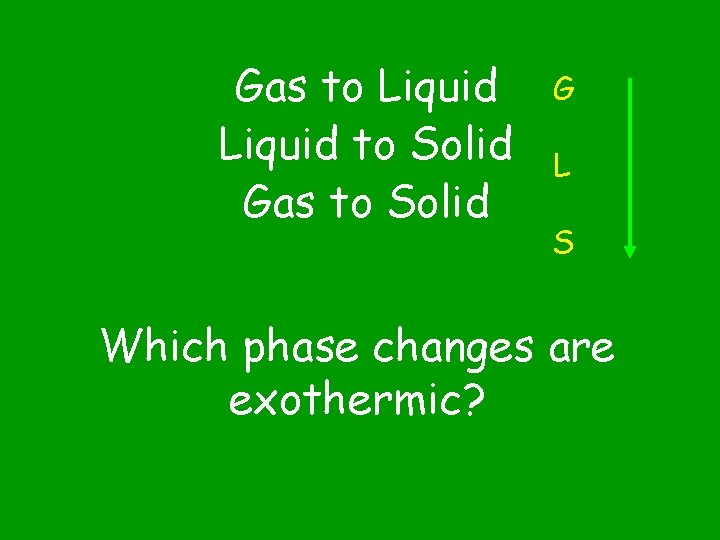
Gas to Liquid to Solid Gas to Solid G L S Which phase changes are exothermic?

Solid > Liquid > Gas Given 3 substances at room temperature: 1 solid, 1 liquid, & 1 gas, which has the strongest intermolecular forces?

Low! Describe the mp & bp of gases.

Nothing, until the phase change is complete. The heat energy is going into the potential energy of the system. What happens to the temperature as heat is added to a substance at its melting or boiling point?
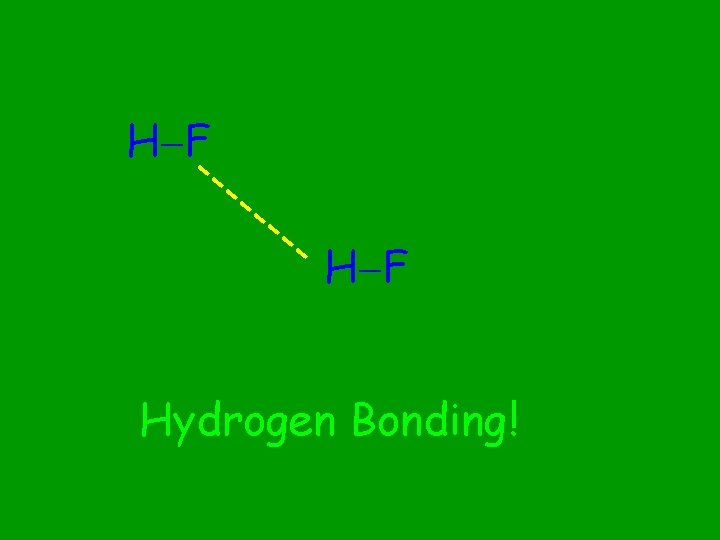
H F --- -- -- H F Hydrogen Bonding!

Ne - -- -- -- Ne Van der Waals or dispersion forces
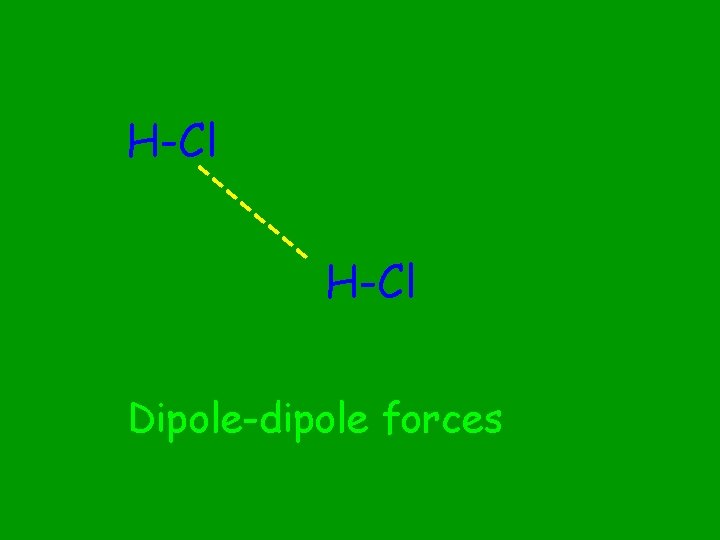
H-Cl --- -- -- H-Cl Dipole-dipole forces
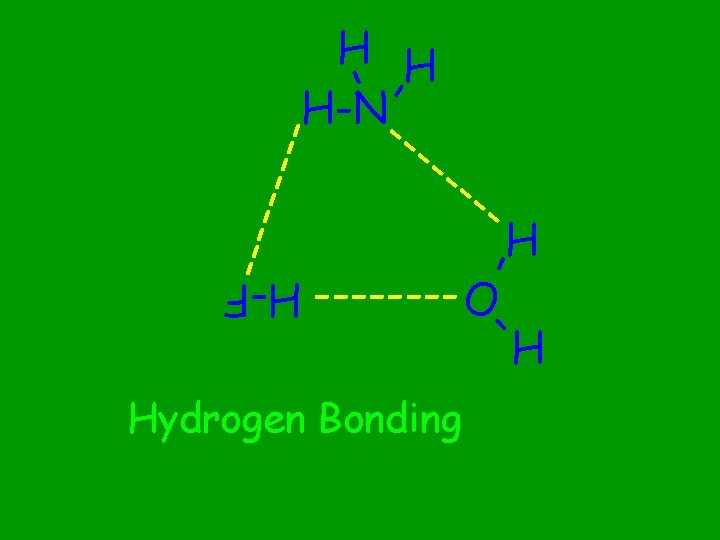
------- - - H H H-N--- ---- O H - H-F Hydrogen Bonding H
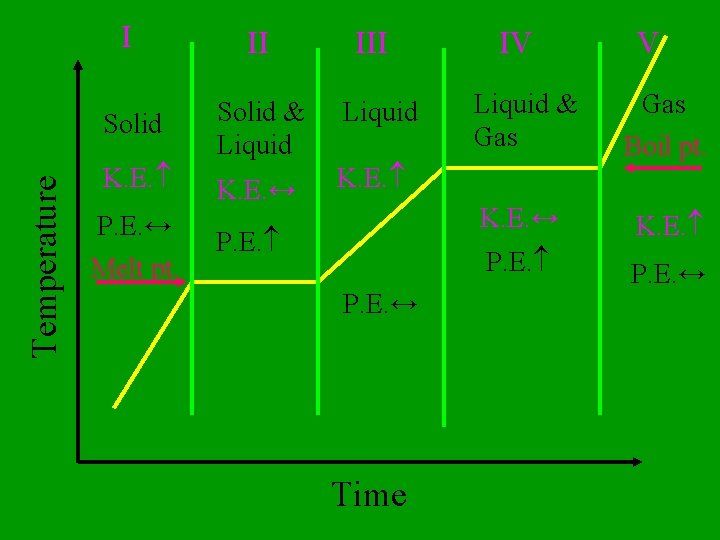
Temperature I II Solid & Liquid K. E. ↔ K. E. P. E. ↔ Melt pt. III P. E. ↔ Time IV V Liquid & Gas Boil pt. K. E. ↔ K. E. P. E. ↔

Q = m. C T Q = energy in Joules m = mass in grams C = specific heat in J/g T = temperature change Equation for a pure substance in a single phase being warmed up or cooled down.

Q = m. Hf Q = energy in Joules m = mass in grams Hf = heat of fusion Equation for a substance melting or freezing at the freezing point.

Q = m. Hv Q = energy in Joules m = mass in grams Hv = heat of vaporization Equation for a substance boiling or condensing at the boiling point.

0 C or 273 K Freezing point Melting point Temperature of the ice-water equilibrium.

100 C or 373 K Boiling point Condensation point Temperature of the steam-water equilibrium.
- Slides: 60