Food Borne Illness Infections that require large infective

Food Borne Illness Infections that require large infective dosage

Infections that require large infective dosage Bacteria Salmonella spp. Yersinia enterocolitica and Yersinia pseudotuberculosis Vibrio parahaemolyticus and other vibrios Escherichia coli Enterotoxigenic (ETEC) Enterohemorrhagic HUS Campylobacter jejuni Listeria

Enterobacteriaceae w Classification – more than 15 different genera Lactoes Ferm +ve Lactoes Ferm –ve Escherichia Enterobacter Citrobacter Klebsiella Pectinobacterium Shigella Edwardsiella Salmonella Hafnia Serratia Proteus Providencia Morganella Yersinia Erwinia

Enterobacteriaceae w Morphology and General Characteristics w Gram-negative, non-sporing, rod shaped bacteria w Oxidase – w Ferment glucose and may or may not produce gas in the process (aerogenic vs anaerogenic) w Reduce nitrate to nitrite (there a few exceptions)

Enterobacteriaceae w Are facultative anaerobes w If motile, motility is by peritrichous flagella w Many are normal inhabitants of the intestinal tract of man and other animals w Some are enteric pathogens and others are urinary or respiratory tract pathogens w Differentiation is based on biochemical reactions and differences in antigenic structure

Enterobacteriaceae w Most grow well on a variety of lab media including a lot of selective and differential media originally developed for the selective isolation of enteric pathogens. w Most of this media is selective by incorporation of dyes and bile salts that inhibit G+ organisms and may suppress the growth of nonpathogenic species of Enterobacteriaceae. w Many are differential on the basis of whether or not the organisms ferment lactose and/or produce H 2 S.
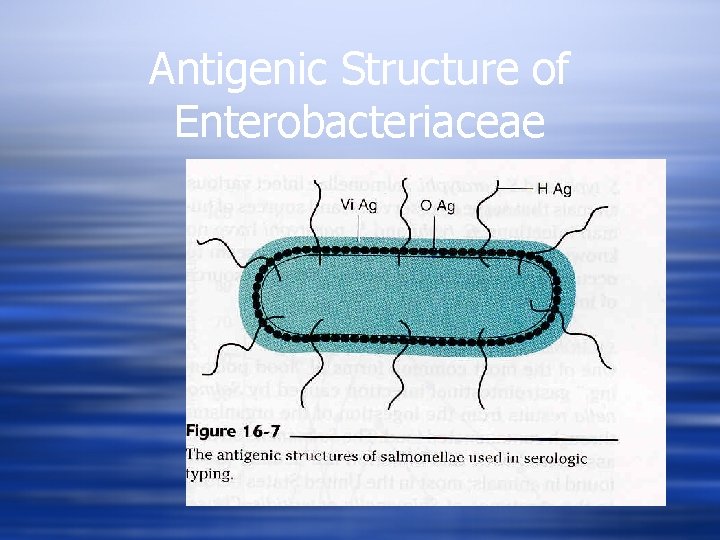
Antigenic Structure of Enterobacteriaceae

Escherichia coli w Normal inhabitant of the G. I. tract. w Some strains cause various forms of gastroenteritis. w Is a major cause of urinary tract infection and neonatal meningitis and septicemia.

E. coli w May be hemolytic on CBA – more common in pathogenic strains w KEY tests for the normal strain: w TSI is A/A + gas w LIA K/K w Urea – w Indole + w Citrate – w Motility + w There is an inactive biotype that is anaerogenic, lactose – , and nonmotile.

Escherichia coli w Virulence factors w Toxins w Enterotoxins and Shigella like Toxins w Enterotoxins causes a movement of water and ions from the tissues to the bowel resulting in watery diarrhea. w There are two types of enterotoxin: LT and ST

E. Coli Enterotoxins w LT – is heat labile and alters the activity of sodium and chloride transporters producing an ion imbalance that results in fluid transport into the bowel. w ST – is heat stable and binds to specific receptors with the same results as with LT.

Shiga-type toxins w Shiga-type toxin – also called the verotoxin produced by enterohemorrhagic strains of E. coli (EHEC) w – is cytotoxic, enterotoxic, neurotoxic, and may cause diarrhea and ulceration of the G. I. tract.

E. coli infections w Gastroenteritis – there are several distinct types of E. coli that are involved in different types of gastroenteritis: w w w enterotoxigenic E. coli (ETEC), enteroinvasive E. coli (EIEC), enteropathogenic E. coli (EPEC) , enteroaggregative E. coli (EAEC), and enterohemorrhagic E. coli (EHEC).

Shigella w Contains four species that differ antigenically and, to a lesser extent, biochemically.

Shigella species w w S. dysenteriae (Group A) S. flexneri (Group B) S. boydii (Group C) S. sonnei (Group D) w Biochemistry w w w TSI K/A with NO gas LIA K/A Urea – Motility All ferment mannitol except S. dysenteriae S. sonnei may show delayed lactose fermentation

Shigella species w Virulence factors w Shiga toxin – is produced by S. dysenteriae and in smaller amounts by S. flexneri and S. sonnei. w Acts to inhibit protein synthesis This plays a role in the ulceration of the intestinal mucosa.

Shigella w Clinical significance w Causes shigellosis or bacillary dysentery. w Transmission is via the fecal-oral route. w The infective dose required to cause infection is very low (10 -200 organisms). w There is an incubation of 1 -7 days followed by fever, cramping, abdominal pain, and watery diarrhea (due to the toxin)for 1 -3 days. w This may be followed by frequent, scant stools with blood, mucous, and pus (due to invasion of intestinal mucosa). w It is rare for the organism to disseminate. w The severity of the disease depends upon the species one is infected with. w S. dysenteria is the most pathogenic followed by S. flexneri, S. sonnei and S. boydii.

Salmonella w Classification has been changing in the last few years. w There is now 1 species: S. enteritica, and 7 subspecies: 1, 2 , 3 a , 3 b , 4 , 5, and 6. w Subgroup 1 causes most human infections w 2000 sub species

Salmonella w Clinically Salmonella isolates are often still reported out as serogroups or serotypes based on the Kauffman-White scheme of classification. w Based on O and H (flagella) antigens w The H antigens occur in two phases; 1 and 2 and only 1 phase is expressed at a given time. w Polyvalent antisera is used followed by group specific antisera (A, B, C 1, C 2, D, and E) w Salmonella typhi also has a Vi antigen which is a capsular antigen.

Salmonella w Biochemistry w TSI K/A + gas and H 2 S: S. typhi produces only a small amount of H 2 S and no gas , and S. paratyphi A produces no H 2 S w LIA K/K with H 2 S with S. paratyphi A giving K/A results w Urea – w Motility + w Citrate +/w Indole - w Virulence factors w Endotoxin – may play a role in intracellular survival w Capsule (for S. typhi and some strains of S. paratyphi) w Adhesions – both fimbrial and non-fimbrial

Salmonella w Clinical Significance – causes two different kinds of disease: enteric fevers and gastroenteritis. w Both types of disease begin in the same way, but with the gastroenteritis the bacteria remains restricted to the intestine and with the enteric fevers, the organism spreads w Transmission is via a fecal-oral route, i. e. , via ingestion of contaminated food or water.

Enterobacteriaceae w Proteus, Providencia, and Morganella w Are all part of the NF of the GI tract (except Providencia). w All motile, with Proteus swarming w PA + w Lysine deamination + (LIA R/A) w Urea + for most, strongly + for Proteus w TSI variable (know the reactions for each in the lab!) w Indole – only P. mirabilis is -

Proteus, Providencia, and Morganella w Virulence factors w Urease – the ammonia produced may damage the epithelial cells of the UT w Clinical Significance w UT infections, as well as pneumonia, septicemia, and wound infections

Enterobacter sakazakii w a Gram-negative rod-shaped pathogenic bacterium. w It is a rare cause of invasive infection with historically high case fatality rates (40– 80%) in infants. w From contaminated infant formulas w E. sakazakii as a now classified into a new genus, Cronobacter, comprising five species w For survivors, severe lasting complications can result including neurological disorders.

Three ways infant formula get contaminated with Cronobacter sp. ? w a) Through the raw material used for producing the formula; w b) through contamination of the formula or other dry ingredients after pasteurization; and w c) through contamination of the formula as it is being reconstituted by the caregiver just prior to feeding. Can other foods also be contaminated? w Cronobacter sp. has been detected in other types of food, but only powdered infant formula has been linked to outbreaks of disease.

Campylobacteriosis Most frequent notified enteric disease w The organism – G-ve, motile, spiral rod w C. jejuni & C. coli w Thermophile (25 to 43 deg. C)
Campylobacter Slender, curved shaped morphology Exponential growth

Campylobacter w Characteristics - thermophile, survives > 1 hour on hands & moist surfaces - survives refrigeration - can revert to VBNC

Campylobacter w The illness - incubation – 2 to 5 days - febrile prodrome - watery/bloody diarrhoea, abdominal pain & nausea

Campylobacter w The illness, continued - infective dose – 1 000 to 10 000 cells - any age group; infants < 1 year & young adults; males up to 45 years - Rx – fluids, ? erythromycin (resistance)

Campylobacter w Sources - human – faecal-oral person-to-person - animal – ruminants, cats, dogs, flies, birds
Campylobacter jejuni

Campylobacter w Sources - food – raw poultry, raw milk, offal, red meat - environment – faeces from infected animals contaminate soil & water

Yersinia enterocolitica Yersiniosis w The organism – small G-ve rods of family Enterobacteriaceae w Geographical variation in pathogenic serotypes w Serotype O: 3 in NZ

Yersinia enterocolitica w Characteristics - grows at wide range of temperatures (0 to 44 0 C), aerobically & anaerobically - withstands freezing & survives in damp soil

Yersinia enterocolitica w The illness - incubation – 7 days - abdominal pain (confused with appendicitis) - headache, fever, diarrhoea, nausea & vomiting (children – watery, mucoid diarrhoea)

Yersinia enterocolitica w The illness, continued - infective dose – unknown - children < 4 years & adults 20 -34 years - Rx – antibiotics when serious

Yersinia enterocolitica w Sources - human – person-to-person, hospitals - animal – primary source is pigs (tongue & tonsils), companion animals, rats & insects - food – pork & pork products - environment – drinking & surface water, sewage sludge
Yersinia enterocolitica

Listeria monocytogenes Listeriosis (invasive disease & non-invasive enteritis) w The organism – G+ve ovoid to rod w Widespread in environment -shaped bacterium
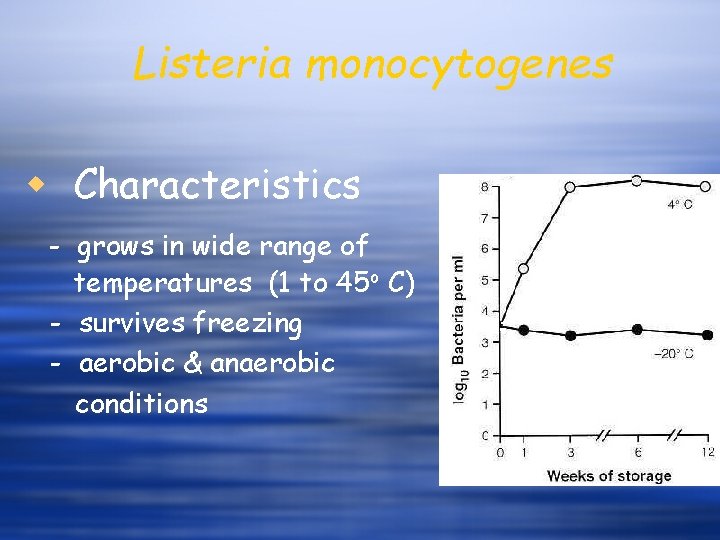
Listeria monocytogenes w Characteristics - grows in wide range of temperatures (1 to 45 o C) - survives freezing - aerobic & anaerobic conditions

Listeria monocytogenes w The illness – invasive form - incubation – 30 days - flu’-like symptoms, diarrhoea, vomiting, meningitis, septicaemia, spontaneous abortion
Listeria monocytogenes w The illness – invasive form, continued - infective dose – 100 to 1 000 cells - pregnant women, newborn babies, the elderly & AIDS patients - Rx – penicillin, ampicillin +/gentamicin

Listeria monocytogenes w The illness – non-invasive - incubation – 18 hours - diarrhoea, fever, muscle pain, headache, abdominal cramps & vomiting

Listeria monocytogenes w The illness – non-invasive - infective dose – > 100 thou. cells/gm - all individuals susceptible - Rx - penicillin, ampicillin +/gentamicin

Listeria monocytogenes w Sources - human – person-to-person rare - animal – diseased animals shed in faeces, contamination of red meat; silage - food – ready-to-eat cooked food with long shelf-life - raw foods - environment – widespread in soil, water & sewage (Hospitals & occupational exposure)
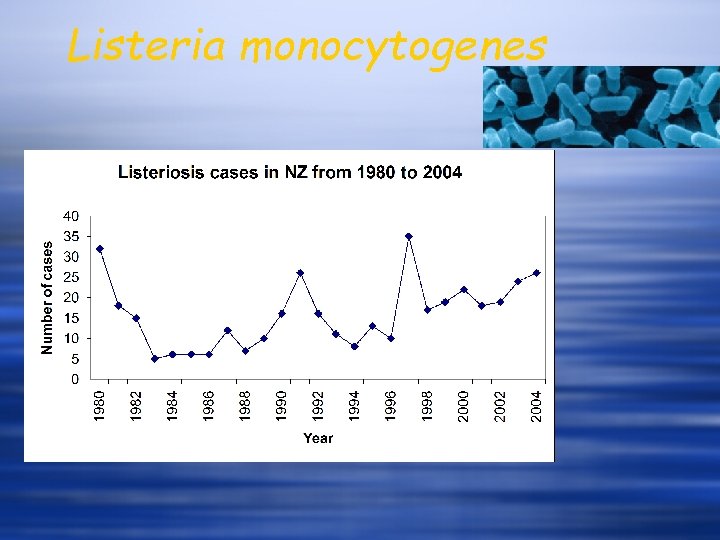
Listeria monocytogenes

Vibrio, Aeromonas & Plesiomonas

General Characteristics of Vibrio, Aeromonas and Plesiomonas Ø Similarities to Enterobacteriaceae · Gram-negative · Facultative anaerobes · Fermentative bacilli Ø Differences from Enterobacteriaceae · Polar flagella · Oxidase positive Ø Formerly classified together as Vibrionaceae · Primarily found in water sources · Cause gastrointestinal disease · Shown not closely related by molecular methods

Morphology & Physiology of Vibrio Ø Comma-shaped (vibrioid) bacilli Ø V. cholerae, V. parahaemolyticus, V. vulnificus are most significant human pathogens Ø Broad temperature & p. H range for growth on media · 18 -37 C · p. H 7. 0 - 9. 0 (useful for enrichment) Ø Grow on variety of simple media including: · Mac. Conkey’s agar · TCBS (Thiosulfate Citrate Bile salts Sucrose) agar Ø V. cholerae grow without salt · Most other vibrios are halophilic

Vibrio spp. (Family Vibrionaceae) Associated with Human Disease

Epidemiology of Vibrio spp. Ø Vibrio spp. (including V. cholerae) grow in estuarine and marine environments worldwide Ø All Vibrio spp. can survive and replicate in contaminated waters with increased salinity and at temperatures of 10 -30 o. C Ø Pathogenic Vibrio spp. appear to form symbiotic (? ) associations with chitinous shellfish which serve as an important and only recently recognized reservoir Ø Asymptomatically infected humans also serve as an important reservoir in regions where cholera is endemic

Taxonomy of Vibrio cholerae Ø >200 serogroups based on somatic O-antigen Ø O 1 and O 139 serogroups are responsible for classic epidemic cholera Ø O 1 serogroup subdivided into · Two biotypes: El Tor and classical (or cholerae) · Three serotypes: ogawa, inaba, hikojima Ø Some O 1 strains do not produce cholera enterotoxin (atypical or nontoxigenic O 1 V. cholerae) Ø Other strains are identical to O 1 strains but do not agglutinate in O 1 antiserum (non-cholera (NCV) or non-agglutinating(NAG) vibrios) (non-O 1 V. cholerae) Ø Several phage types

Epidemiology of Vibrio cholerae Ø Cholera recognized for more than two millennia with sporadic disease and epidemics Ø Endemic in regions of Southern and Southeastern Asia; origin of pandemic cholera outbreaks Ø Generally in communities with poor sanitation Ø Seven pandemics (possible beginning of 8 th) since 1817 attributable to increased world travel Ø Cholera spread by contaminated water and food Ø Human carriers and environmental reservoirs

Recent Cholera Pandemics Ø 7 th pandemic: · · V. cholerae O 1 biotype El Tor Began in Asia in 1961 Spread to other continents in 1970 s and 1980 s Spread to Peru in 1991 and then to most of South & Central America and to U. S. & Canada · By 1995 in the Americas, >106 cases; 104 dead Ø 8 th pandemic (? ? ) · V. cholerae O 139 Bengal is first non-O 1 strain capable of causing epidemic cholera · Began in India in 1992 and spread to Asia, Europe and U. S. · Disease in humans previously infected with O 1 strain, thus no cross-protective immunity

Pathogenesis of V. cholerae Ø Incubation period: 2 -3 days Ø High infectious dose: >108 CFU · 103 -105 CFU with achlorhydria or hypochlorhydria (lack of or reduced stomach acid) Ø Abrupt onset of vomiting and life-threatening watery diarrhea (15 -20 liters/day) Ø As more fluid is lost, feces-streaked stool changes to rice-water stools: · Colorless · Odorless · No protein · Speckled with mucus

Pathogenesis of V. cholerae (cont. ) Ø Cholera toxin leads to profuse loss of fluids and electrolytes (sodium, potassium, bicarbonate) · Hypokalemia (low levels of K in blood) · Cardiac arrhythmia and renal failure Ø Cholera toxin blocks uptake of sodium & chloride from lumen of small intestine Ø Death attributable to: · Hypovolemic shock (due to abnormally low volume of circulating fluid (plasma) in the body) · Metabolic acidosis (p. H shifts toward acid side due to loss of bicarbonate buffering capacity)

Treatment & Prevention of V. cholerae Ø Untreated: 60% fatality Ø Treated: <1% fatality Ø Rehydration & supportive therapy · Oral Sodium chloride (3. 5 g/L) + Potassium chloride (1. 5 g/L) + Rice flour (30 -80 g/L) + Trisodium citrate (2. 9 g/L) · Intravenous (IV) Ø Doxycycline or tetracycline (Tet resistance may be developing) of secondary value Ø Water purification, sanitation & sewage treatment Ø Vaccines

Laboratory Identification of Vibrios Ø Transport medium - Cary-Blair semi-solid agar Ø Enrichment medium - alkaline peptone broth • Vibrios survive and replicate at high p. H • Other organisms are killed or do not multiply Ø Selective/differential culture medium - TCBS agar • V. cholerae grow as yellow colonies Ø Biochemical and serological tests

Characteristics and Epidemiology of Aeromonas (Family Aeromonadaceae) Ø Gram-negative facultatively anaerobic bacillus resembling members of the Enterobacteriaceae Ø Motile species have single polar flagellum (nonmotile species apparently not associated with human disease) Ø 16 phenospecies: Most significant human pathogens A. hydrophila, A. caviae, A. veronii biovar sobria Ø Ubiquitous in fresh and brackish water Ø Acquired by ingestion of or exposure to contaminated water or food

Clinical Syndromes of Aeromonas Ø Associated with gastrointestinal disease · Chronic diarrhea in adults · Self-limited acute, severe disease in children resembling shigellosis with blood and leukocytes in the stool · 3% carriage rate Ø Wound infections Ø Opportunistic systemic disease in immunocompromised Ø Putative virulence factors include: endotoxin; hemolysins; eneterotoxin; proteases; siderophores; adhesins
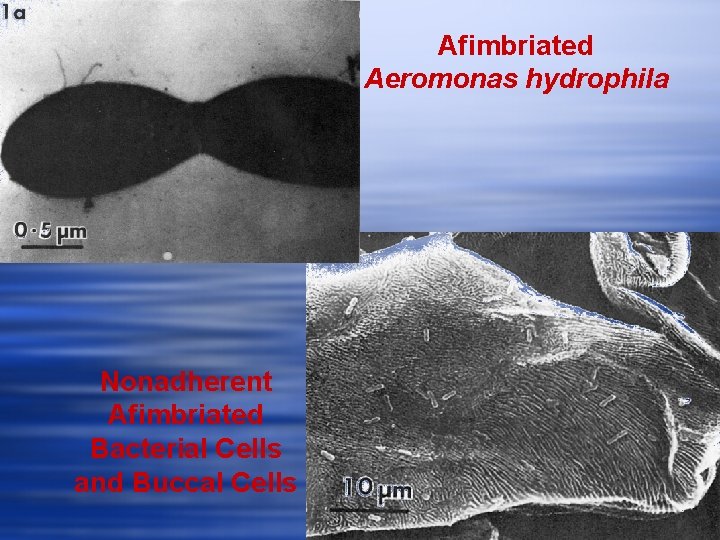
Afimbriated Aeromonas hydrophila Nonadherent Afimbriated Bacterial Cells and Buccal Cells
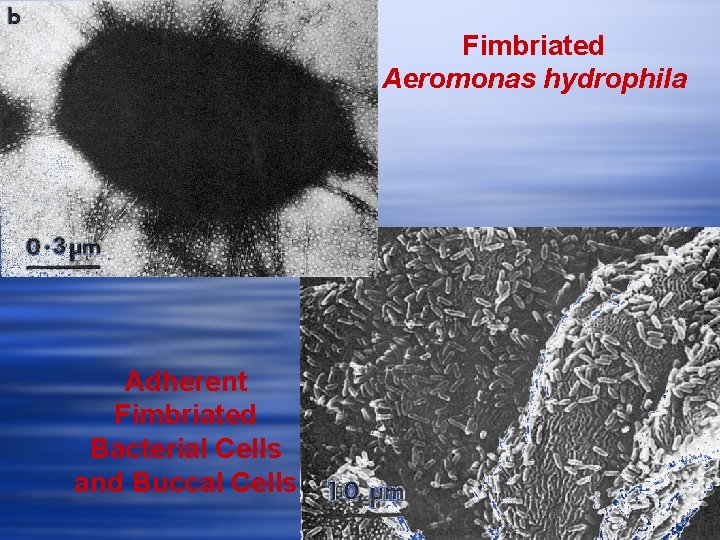
Fimbriated Aeromonas hydrophila Adherent Fimbriated Bacterial Cells and Buccal Cells

Characteristics of Plesiomonas Ø Formerly Plesiomonadaceae Ø Closely related to Proteus & now classified as Enterobacteriaceae despite differences: · Oxidase positive · Multiple polar flagella (lophotrichous) Ø Single species: Plesiomonas shigelloides Ø Isolated from aquatic environment (fresh or estuarine) Ø Acquired by ingestion of or exposure to contaminated water or seafood or by exposure to amphibians or reptiles Ø Self-limited gastroenteritis: secretory, colitis or chronic forms Ø Variety of uncommon extra-intestinal infections

Characteristics of Aeromonas and Plesiomonas Gastroenteritis Epidemiological Features Natural Habitat Source of Infection Aeromonas Plesiomonas Fresh or brackish water Contaminated water or food Blood/WBCs in Stool Present Present Absent Present Pathogenesis Enterotoxin (? ? ) Invasiveness Clinical Features Diarrhea Vomiting Abdominal Cramps Fever
- Slides: 65