FOCUS ACTIVITY FILL IN THE FLOW CHART Matter































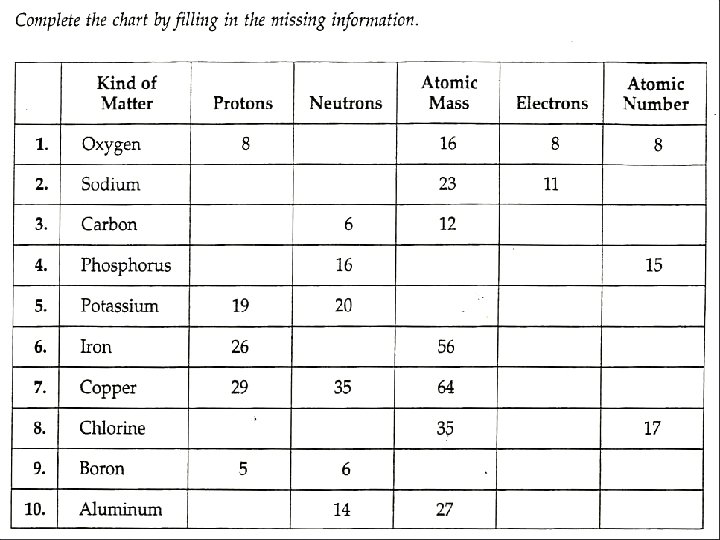








- Slides: 53

FOCUS ACTIVITY: FILL IN THE FLOW CHART Matter is made up of… ATOMS ____ The smallest particles. These particles help to make up… Elements, which help to make up. Compounds _____, a combination of elements in a fixed ratio.

FOCUS ACTIVITY: GRAB A WORKSHEET, 1 FOR YOU AND YOUR PARTNER TO SHARE. READ THE BACKGROUND INFORMATION AND PROCEDURE Homework: Work on your Adopt an Element Project and Study your Elements!

4. 1 The History of the Atoms make up the smallest particle of an element.

This model of the atom may look familiar to you. In this model, the nucleus is orbited by electrons, which are in different energy levels. A model uses familiar ideas to explain unfamiliar facts observed in nature. A model can be changed as new information is collected. Atomic Models

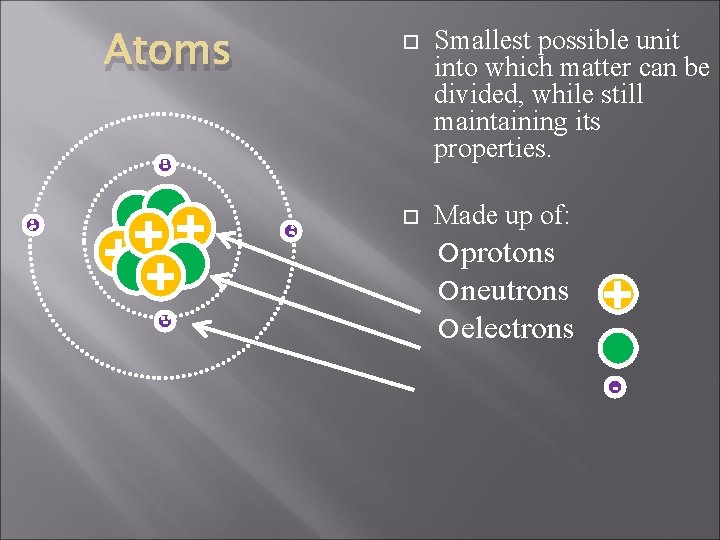
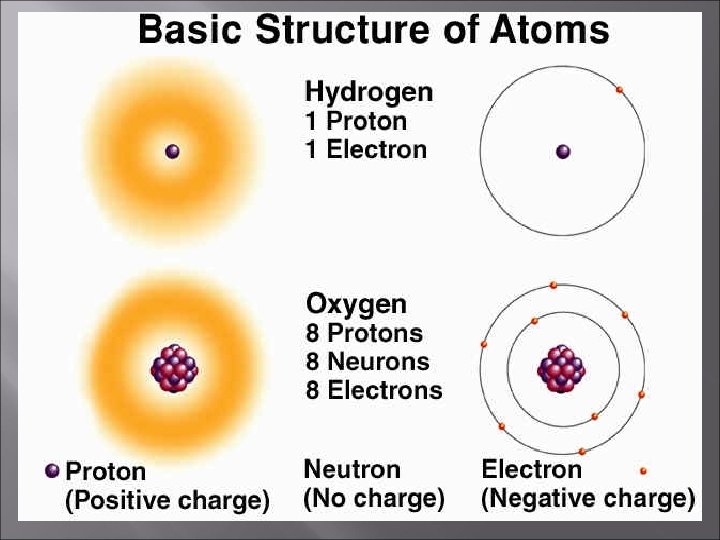
Atoms Smallest possible unit into which matter can be divided, while still maintaining its properties. Made up of: - + + ++ - - protons neutrons electrons + -



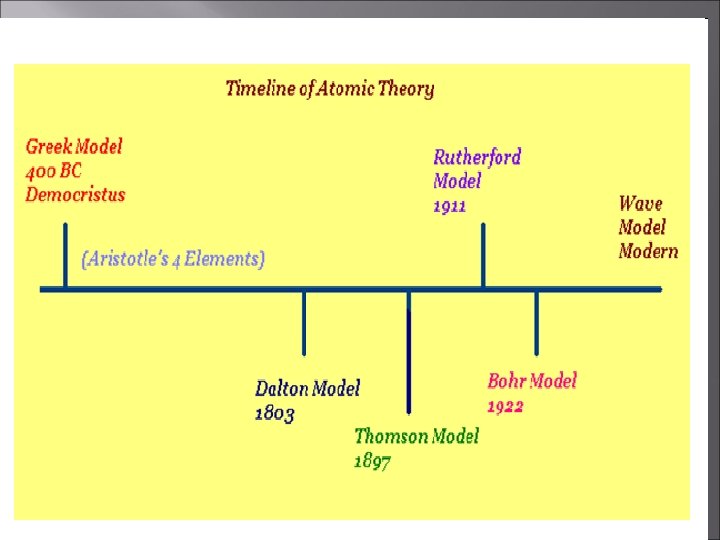
Atomic Theory The atomic theory has changed so much in the past starting in 400 B. C. when it was thought to look like a billiard ball.

Early Atomic Theory Democritus, Greek Philosopher, first proposed the idea that matter was made up of smaller pieces.


This theory was ignored and forgotten for more than 2000 years!!!!!!! Why? . . .

Aristotle A well respected philosopher suggested that matter was composed of 4 basic elements: water, fire, earth, and air. His theory was ultimately wrong, but believed for many years, delaying the pursuit of Atomos


John Dalton (early 1800 s) using the experimental observations of others, proposed his own atomic theory.

Dalton’s Atomic Theory 1. 2. 3. 4. 5. All elements are composed of tiny particles called atoms. Atoms of the same element are identical. The atoms of an element are different from those of another element. Atoms can not be changed into an atom of another element. Atoms can not be created or destroyed through chemical change, only rearranged. Every compound is composed of atoms of different elements, combined in a specific ratio.

J. J. Thomson British Scientist- 1897 Discovered negatively charged particles within the atom- (Electrons) through use of Cathode Ray Tube “Plum Pudding Model” -Atom = big positive charge with negative charges embedded in it. -looks like berries in a muffin

Cathode Rays and the Electron (DO NOT WRITE) J. J. Thomson investigated cathode rays using a device called a cathode ray tube. Currents of electricity were pumped into vacuum tubes. Thomson used magnets to determine the identity of the particles making up the rays. He found them to be tiny, negatively charged particles. http: //www. goalfinder. com/preview. asp? pro ductcode=SPAPRO 1&productid=66&produ ctname=Discovery of electron ( Science animation)

Robert Millikan (1909) - oil drop experiment – measured the charge and mass of electron http: //www. youtube. com/watch? v=XMf. YHag 7 Liw &feature=related He determined an electron has a mass of 1/1840 of an hydrogen atom and 1 unit of negative charge.

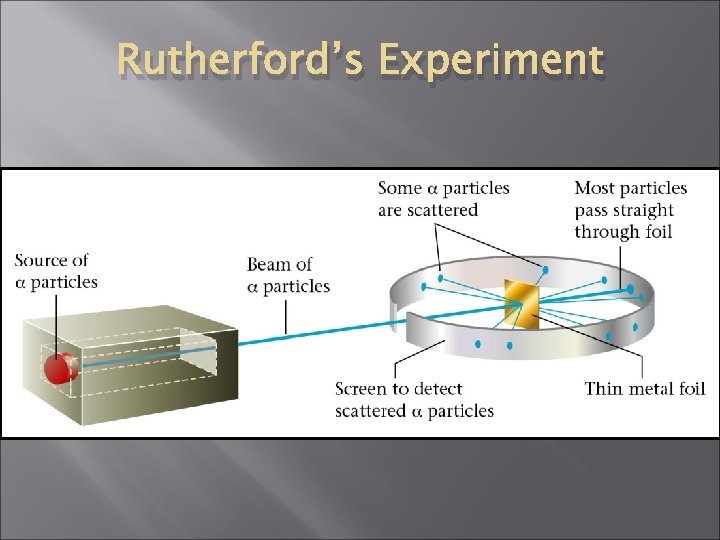
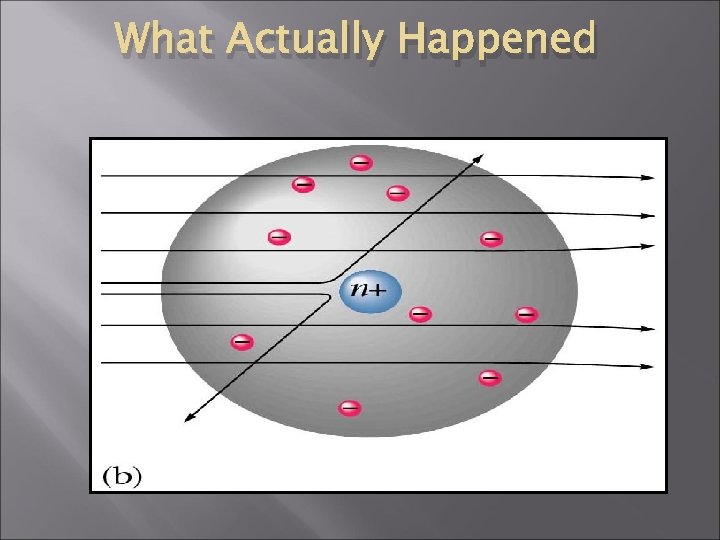
Ernest Rutherford - 1911 New ideas. Gold foil experiment Atom has a lot of empty space with Small, dense positively charged nucleus

Rutherford’s Experiment

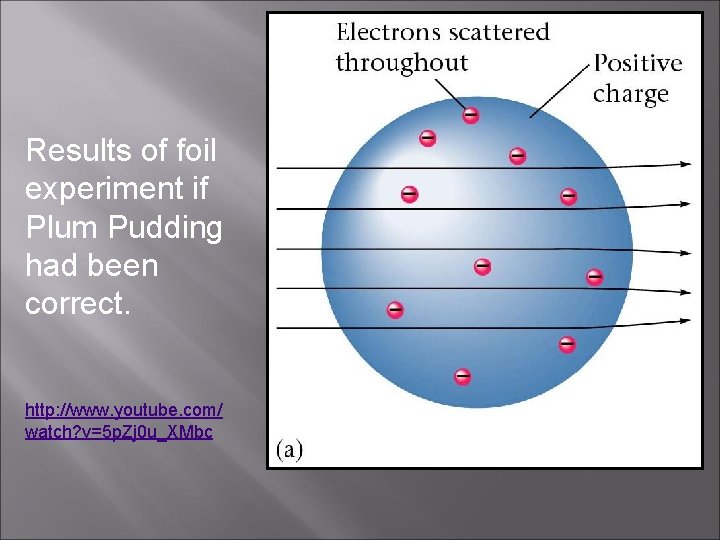
Results of foil experiment if Plum Pudding had been correct. http: //www. youtube. com/ watch? v=5 p. Zj 0 u_XMbc

What Actually Happened

Who Discovered the Proton? Goldstein suggested that since the atom is electrically neutral there must be a positively charged particle in the atom and tried to discover it. (1886) Rutherford's discovery of the nucleus demonstrated that these positive charges were concentrated in a very small fraction of the atoms' volume. (1918) Credit was given to Rutherford Canal Rays or positive Protons (red glow)

Recap! What is the positive particle that makes up part of the atom? The negative? Where are they located in the atom?

Niels Bohr 1913 ØElectrons have distinct orbits called ENERGY LEVELS. ØExcited – they jump levels ØCome back down – release light

Bohr Model Add energy to an electron = Excited State It will eventually drop back down to it’s GROUND STATE and give off energy in the form of light. (The lighted Pickle)

De Broglie and Schrodinger Louis De Broglie (1924): Electrons do not travel in perfect orbits, they travel in orbitals. Erwin Schrodinger (1930): The Probable location of an electron is based on how much energy the electron has. This model can be described as where the electron SHOULD be. The resulting image was an “electron cloud” Current Accepted Atomic Theory

Chadwick-1932 Chadwick -Discovered neutral particles of same mass as protons -Neutrons -Located in nucleus

Electrons are “wavicles”. Bohr model of the atom shows electrons orbiting a nucleus. Electrons are particles When we attempt to observe an electron, it changes position Electrons are waves

Current Atomic Theory 1. Center of Atom: Dense positively charged nucleus (protons and neutrons) 2. Negatively charged electrons with 1/5000 th of mass are moving around 3. Most of atom is empty space 4. Electrons have energy based on distance from nucleus. “Shell” 5. Electrons emit light or absorb energy when moving from one energy level to another 6. No exact known location for electrons, but probable placement- “Electron Cloud”

Copy this Chart into the LEFT side. Particle Charge Relative Location mass 1/1840 ? Electron -1 Proton +1 1 ? 0 1 ? Neutron :

The Atom An atom consists of a nucleus (of protons and neutrons) electrons in space about the nucleus. Electron cloud Nucleus


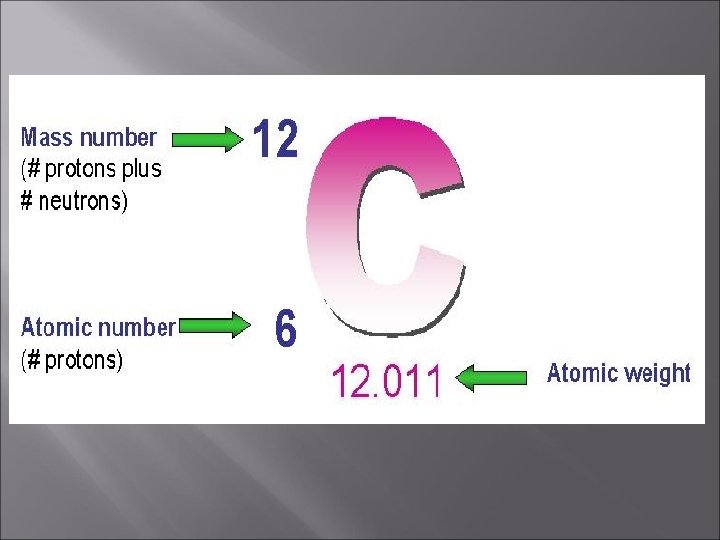
The atomic number is the number of protons in an atom. This number is unique for all elements and the atomic number is used to identify each element. Since atoms are electrically neutral, THE NUMBER OF PROTONS EQUALS THE NUMBER OF ELECTRONS.

The mass number is the total number of protons and neutrons in an atom. Mass Number = protons + neutrons Mass number – protons = Number of Neutrons

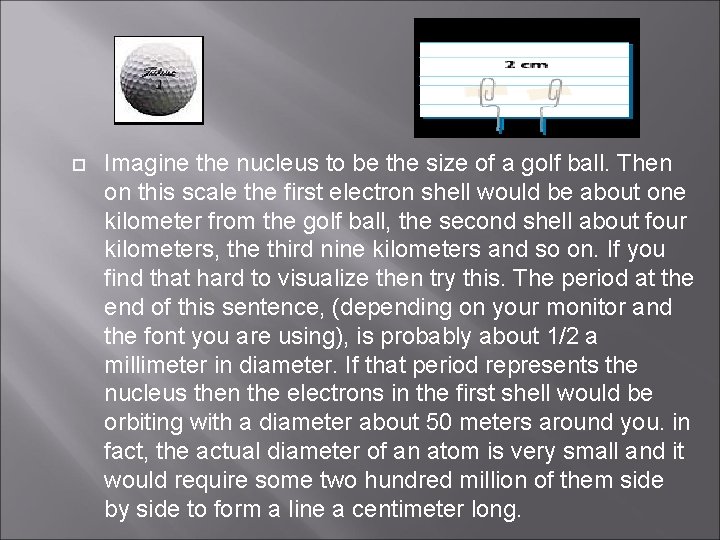
Imagine the nucleus to be the size of a golf ball. Then on this scale the first electron shell would be about one kilometer from the golf ball, the second shell about four kilometers, the third nine kilometers and so on. If you find that hard to visualize then try this. The period at the end of this sentence, (depending on your monitor and the font you are using), is probably about 1/2 a millimeter in diameter. If that period represents the nucleus then the electrons in the first shell would be orbiting with a diameter about 50 meters around you. in fact, the actual diameter of an atom is very small and it would require some two hundred million of them side by side to form a line a centimeter long.

Energy Levels In Atoms Electrons Level 1 - 2 Level 2 - 8 Level 3 - 18 Level 4 - 32 http: //www. youtube. com/watch? v=WK 7 wu Tw. Ai. BU

Focus Activity: Grab a textbook and a blue, yellow, and green colored pencil or crayon for you and your partner to share. Then complete this problem: In a class of 10 students; 4 weigh 50 kg, 3 weigh 70 kg, 2 weigh 30, and 1 weighs 100 kg. What percentage of students weigh under 100 kg? 70 kg? 50 kg? What is the average weight for this class? 9 out of 10 weigh under 100 kg= 90% 6 out of 10 weigh under 70 kg = 60% 2 out of 10 weigh under 50 kg = 20% Average weight for the class: (4 X 50 kg) + (3 X 70 kg) + (2 X 30 kg) + (1 x 100 kg) 570 kg / 10 = 57 kg

EXAMPLE How many protons, neutrons and electrons are found in an atom of 133 55 Cs Atomic number = protons and electrons There are 55 protons and 55 electrons Mass number = sum of protons and neutrons 133 – 55 = 78 There are 78 neutrons


The elements of aluminum, Iron, Oxygen, and Silicon make up about 88 percent of the earth's solid surface. Water - made up of hydrogen and oxygen. Air is 99 percent nitrogen and oxygen. Hydrogen, oxygen, and carbon make up 97 percent of a person. Almost everything you see in this picture us made up of just 6 elements.




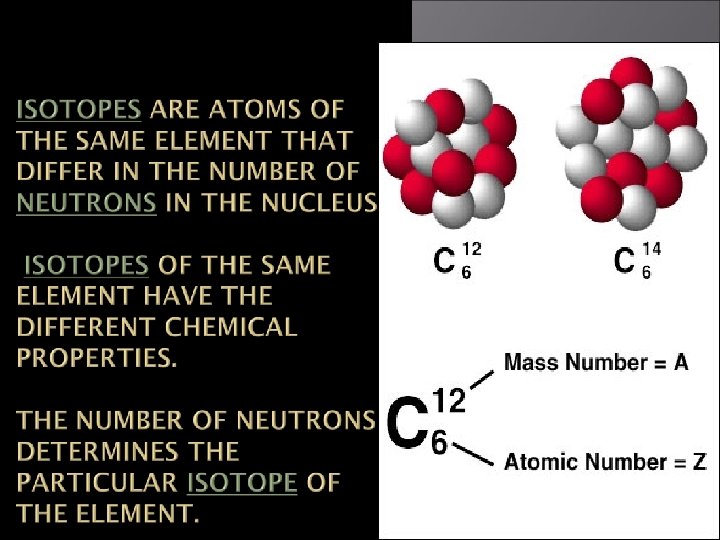
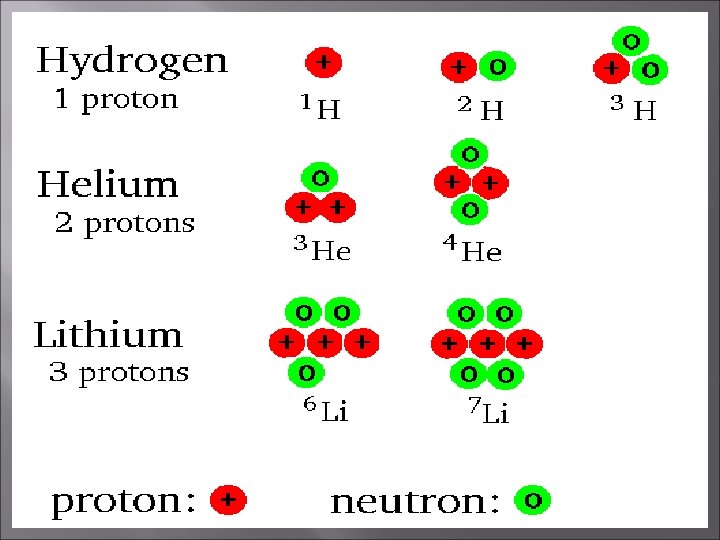
Isotopes Atoms of the same element but different mass number. Boron-10 (10 B) has 5 p and 5 n Boron-11 (11 B) has 5 p and 6 n 11 B 10 B

Focus Activity: How many electrons should an atom have? Why are isotopes still the same element?

Calculating Atomic Mass is the weighted average of the masses of the isotopes of that element. A weighted average takes into consideration both the mass and the abundance of each isotope. The correct unit for atomic mass is amu.

Calculating Atomic Mass To calculate relative atomic mass, multiply the mass number of each isotope by its percent abundance changed to a decimal and total. Boron is 20% 10 B and 80% 11 B. That is, 11 B is 80 percent abundant on earth. = 0. 20 (10 amu) + 0. 80 (11 amu) = 10. 8 amu Hint – decimals will always add up to 1 or 100%

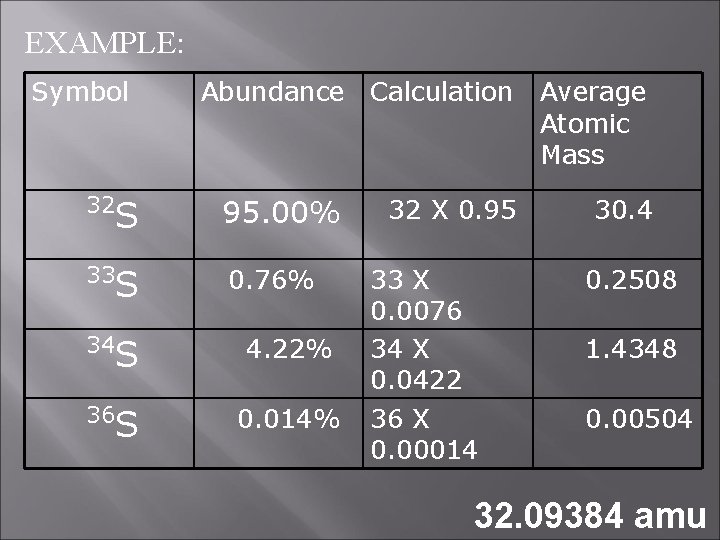
EXAMPLE: Symbol Abundance Calculation 32 S 95. 00% 33 S 0. 76% 34 S 4. 22% 36 S 0. 014% 32 X 0. 95 33 X 0. 0076 34 X 0. 0422 36 X 0. 00014 Average Atomic Mass 30. 4 0. 2508 1. 4348 0. 00504 32. 09384 amu

Isotopes & Average Atomic Mass Because of the existence of isotopes, the mass of a collection of atoms has an average value. 6 Li = 7. 5% abundant and 7 Li = 92. 5% 6. 925 amu Avg. Atomic mass of Li = _______ 28 Si = 92. 23%, 29 Si = 4. 67%, 30 Si = 3. 10% 28. 1087 amu Avg. Atomic mass of Si = _______

Focus Activity: Example 2: Neon has 2 isotopes, Ne-20 with an abundance of 90% and Ne-22 with an abundance of 10%. What are the 2 isotopes? Calculate the average atomic mass of neon. Ne-20 Ne-22. 90 X 20 +. 10 X 22 = 20. 2 amu Example 3: Carbon occurs in nature as a mixture of atoms of which 98. 89% have a mass of 12. 00 u and 1. 11% have a mass of 13. 00335 u. Calculate the atomic mass of carbon. . 9889 X 12 +. 011 X 13. 00335 = 12. 01113719 amu

Truck tires 29 27 Temp increases: Explain condensation using Kinetic Theory 30 28 Water vapor (gas) hits the Pressure increases cold glass and the particles (Gay-Lussac’s Law) lose energy turning into (liquid) water. This is condensation on the outside of the glass.

Focus Activity: Work on worksheet H/w: 4. 2 questions and reading, Finish Worksheets Finish Project