FMEA Applied to the Phenomenon of Retained Objects

FMEA Applied to the Phenomenon of Retained Objects After Surgery Project Managers Dr. Joan Burtner and Dr. Laura Moody Mercer University School of Engineering

Presentation Overview n n n Introduction Motivation for the Study Healthcare Failure Modes and Effects Analysis Case Study Typical Results Select Recommendations Questions/Comments IIE/ASQ 'Quality Drives Lean' Conference Oct 2006 Dr. Joan Burtner, Presenter Slide 2

Introduction n To Err is Human – Call for action with respect to reducing medical errors Case study courses at Mercer University School of Engineering emphasize realworld projects Clients: MD and RN responsible for administering Quality programs at a hospital in the southeast IIE/ASQ 'Quality Drives Lean' Conference Oct 2006 Dr. Joan Burtner, Presenter Slide 3

What is an FMEA? n n n Failure Modes and Effects Analysis “FMEA is a team-based problem-solving tool intended to help users identify and eliminate, or reduce the negative effects of, potential failures before they occur in systems, subsystems, product or process design, or the delivery of a service. ” The Certified Quality Engineer Handbook, page 233 CQE Body of Knowledge (Reliability and Risk Management) IIE/ASQ 'Quality Drives Lean' Conference Oct 2006 Dr. Joan Burtner, Presenter Slide 4

What is a Healthcare FMEA? n Motivation for the HFMEA n n Joint Commission on the Accreditation of Healthcare Organizations (JCAHO) Standard LD. 5. 2 requires facilities to select at least one high-risk process for proactive risk assessment annually FMEA vs HFMEA n n n HFMEA combines the detectability and criticality steps of a traditional FMEA HFMEA uses a hazard score in place of the risk priority number (RPN) that is associated with a traditional FMEA Hazard Score obtained from the Hazard Matrix Table developed by the Department of Veteran’s Affairs National Center for Patient Safety IIE/ASQ 'Quality Drives Lean' Conference Oct 2006 Dr. Joan Burtner, Presenter Slide 5

Project Timeline 2005 -2006 n Preliminary research n n Operating room observations Process flow and documentation n n Journal articles and books Materials provided by southeastern hospital High-Level Detailed counting procedures Healthcare Failure Modes and Effects Analysis Consultation with MD and RN IIE/ASQ 'Quality Drives Lean' Conference Oct 2006 Dr. Joan Burtner, Presenter Slide 6

Project Team n Clients: Upper level administrators at a southeastern hospital n n n Faculty at Mercer University n n n Chief Quality Officer (Physician) Performance Improvement Coordinator of Surgical Services (Registered Nurse) Dr. Joan Burtner – Certified Quality Engineer Dr. Laura Moody – Human Factors Engineer Students enrolled at Mercer University n n Industrial Engineering Seniors Industrial Management Seniors IIE/ASQ 'Quality Drives Lean' Conference Oct 2006 Dr. Joan Burtner, Presenter Slide 7

Preliminary Research n n Factors associated with retained objects n Emergency surgery n Unplanned change in surgical procedure n Patient obesity (higher mean body-massindex) Most likely causes for discrepancies in counts n Intensity/complexity of the environment n Non-standardized methods for performing counts n Poor communication among the Operating Room (OR) team members IIE/ASQ 'Quality Drives Lean' Conference Oct 2006 Dr. Joan Burtner, Presenter Slide 8

Site-Specific Observations n n Forms used by southeastern hospital Qualitative assessment of process Two people have to witness the count for it to be valid Lap sponges are mainly lost in cases with obese people and/or abdominal surgeries Sponges will do more damage to the patient than instruments if left inside the body, due to decomposition IIE/ASQ 'Quality Drives Lean' Conference Oct 2006 Dr. Joan Burtner, Presenter Slide 9

Healthcare FMEA Step #1 n Define the process that will be examined and define the scope Process - Counting of surgical tools and sponges prior to, during and after operations n Goal - Provide client with possible recommendations for performing this task that will attempt to prevent surgical tools and sponges from being left inside patients n IIE/ASQ 'Quality Drives Lean' Conference Oct 2006 Dr. Joan Burtner, Presenter Slide 10

Healthcare FMEA Step #2 n Assemble the Team n n n ISE Students ISE Professors IDM Students MD RN Expertise n n Subject-matter Process Improvement IIE/ASQ 'Quality Drives Lean' Conference Oct 2006 Dr. Joan Burtner, Presenter Slide 11

Healthcare FMEA Step #3 n n Graphically represent the process Two flows generated High-Level process flow n Detailed counting procedure process flow n n Only partial graphics will be presented due to proprietary reasons IIE/ASQ 'Quality Drives Lean' Conference Oct 2006 Dr. Joan Burtner, Presenter Slide 12

Healthcare FMEA Step #4 n Conduct a hazard analysis n n n Define potential failures at each step in each process Define causes for failures at each step in process Assign severity rating: n n n catastrophic, major, moderate, minor Determine probability score Determine hazard score Eliminate, control, or accept failure mode Actions for eliminate or control Who is responsible? IIE/ASQ 'Quality Drives Lean' Conference Oct 2006 Dr. Joan Burtner, Presenter Slide 13

Process Flow and Documentation n High-Level Process Highlights overall operating room procedures n Reviewed and approved by RN n n Detailed Counting Procedures Highlights the specific counting procedures for sponges, sharps, and instruments n Reviewed and approved by RN n IIE/ASQ 'Quality Drives Lean' Conference Oct 2006 Dr. Joan Burtner, Presenter Slide 14
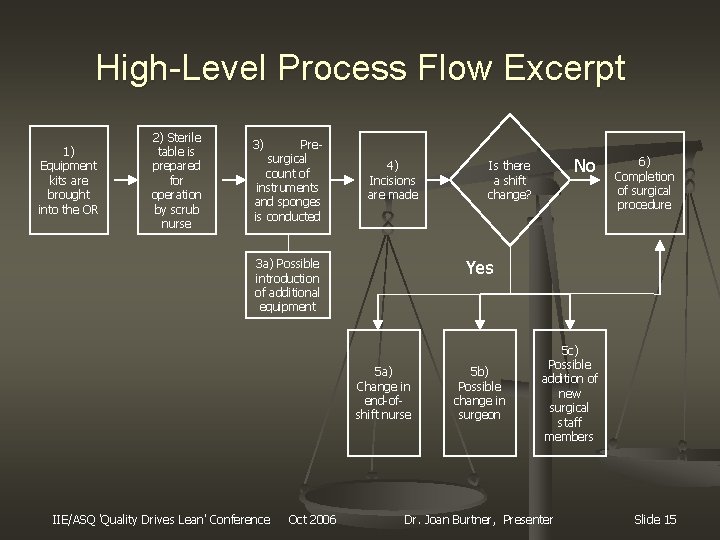
High-Level Process Flow Excerpt 1) Equipment kits are brought into the OR 2) Sterile table is prepared for operation by scrub nurse 3) Presurgical count of instruments and sponges is conducted 4) Incisions are made 5 a) Change in end-ofshift nurse Oct 2006 6) Completion of surgical procedure Yes 3 a) Possible introduction of additional equipment IIE/ASQ 'Quality Drives Lean' Conference No Is there a shift change? 5 b) Possible change in surgeon 5 c) Possible addition of new surgical staff members Dr. Joan Burtner, Presenter Slide 15
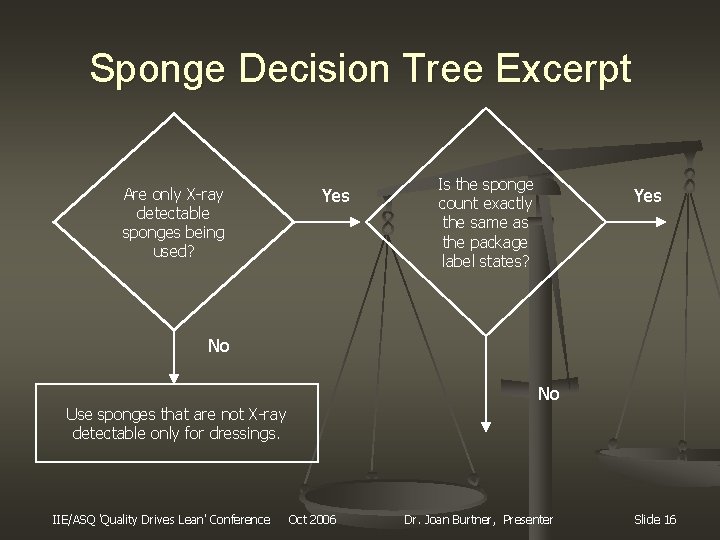
Sponge Decision Tree Excerpt Are only X-ray detectable sponges being used? Yes Is the sponge count exactly the same as the package label states? Yes No No Use sponges that are not X-ray detectable only for dressings. IIE/ASQ 'Quality Drives Lean' Conference Oct 2006 Dr. Joan Burtner, Presenter Slide 16
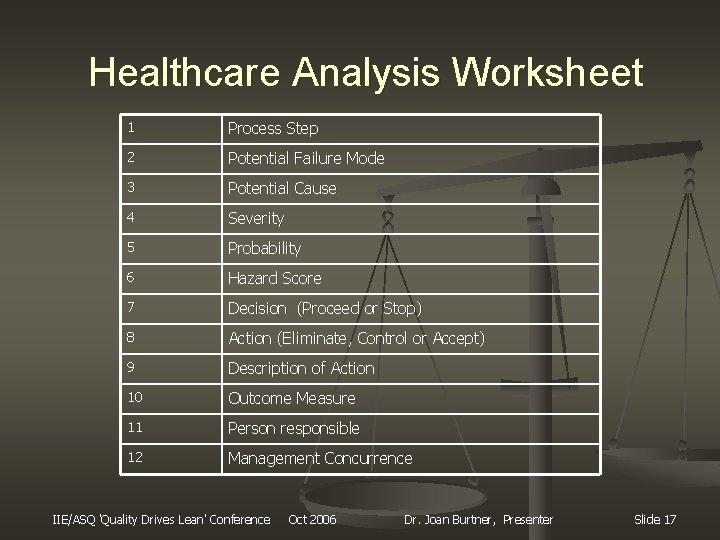
Healthcare Analysis Worksheet 1 Process Step 2 Potential Failure Mode 3 Potential Cause 4 Severity 5 Probability 6 Hazard Score 7 Decision (Proceed or Stop) 8 Action (Eliminate, Control or Accept) 9 Description of Action 10 Outcome Measure 11 Person responsible 12 Management Concurrence IIE/ASQ 'Quality Drives Lean' Conference Oct 2006 Dr. Joan Burtner, Presenter Slide 17
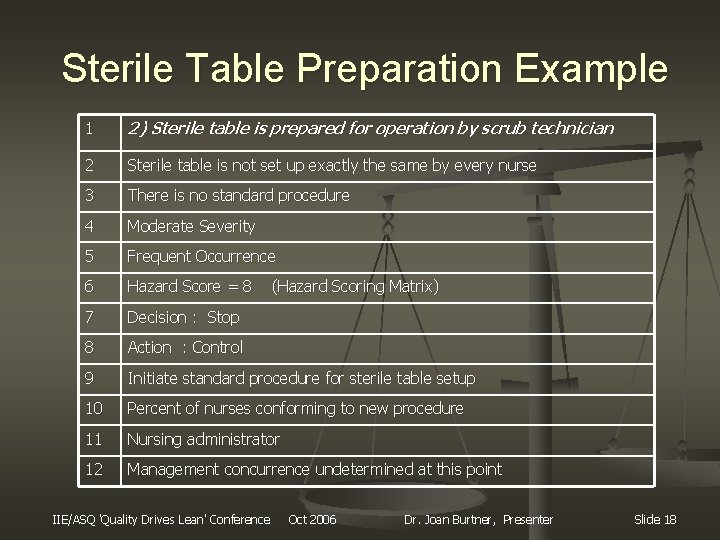
Sterile Table Preparation Example 1 2) Sterile table is prepared for operation by scrub technician 2 Sterile table is not set up exactly the same by every nurse 3 There is no standard procedure 4 Moderate Severity 5 Frequent Occurrence 6 Hazard Score = 8 7 Decision : Stop 8 Action : Control 9 Initiate standard procedure for sterile table setup 10 Percent of nurses conforming to new procedure 11 Nursing administrator 12 Management concurrence undetermined at this point IIE/ASQ 'Quality Drives Lean' Conference (Hazard Scoring Matrix) Oct 2006 Dr. Joan Burtner, Presenter Slide 18
n Accept n n Step 1 - Equipment is brought into the operating room Control n n Results (High-Level) Step 3 - Pre-surgical count of the sponges and instruments Eliminate n Step 6 - Completion of surgical process n Instruments, sponges, or sharps are left inside of a patient IIE/ASQ 'Quality Drives Lean' Conference Oct 2006 Dr. Joan Burtner, Presenter Slide 19

Results (Detailed Counting Procedures) n Accept n n Control n n Step 2 a - Sponges are not completely separated during the count Step 11 - Object has left sterile field, circulator must retrieve and verify with the scrub nurse Eliminate n Step 10 - The scrub nurse continually counts needles during the procedure IIE/ASQ 'Quality Drives Lean' Conference Oct 2006 Dr. Joan Burtner, Presenter Slide 20

Recommendations for Future n n Review FMEA worksheets Institute recommendations and test Continue to monitor process flow periodically Revise as necessary IIE/ASQ 'Quality Drives Lean' Conference Oct 2006 Dr. Joan Burtner, Presenter Slide 21

Acknowledgements The project managers would like to acknowledge the exceptional efforts of the members of the student team as well as the professionalism of our community partners at a hospital in the southeast. IIE/ASQ 'Quality Drives Lean' Conference Oct 2006 Dr. Joan Burtner, Presenter Slide 22

Questions or Comments? Dr. Joan Burtner ASQ Certified Quality Engineer Associate Professor of Industrial Engineering Mercer University Macon, GA (478) 301 -4127 Burtner_J@Mercer. edu IIE/ASQ 'Quality Drives Lean' Conference Oct 2006 Dr. Joan Burtner, Presenter Slide 23
- Slides: 23